Abstract
Objective
To review the efficacy and safety of tannin albuminate and ethacridine lactate monohydrate in the treatment and prevention of diarrhea.
Design
Narrative review.
Results
Of 366 titles identified, a total of 13 studies were included in this review. Five randomized controlled trials and four noninterventional trials demonstrate the efficacy and safety of the fixed combination of tannin albuminate and ethacridine lactate monohydrate in treating acute diarrhea and preventing traveler's diarrhea. The fixed combination reduced acute diarrhea duration by improving stool frequency, consistency, and weight and reduced the risk of traveler's diarrhea. Two pilot studies indicate efficacy in chronic diarrhea. Safety and tolerability were very good in all included studies. Two meta-analyses investigated the efficacy of gelatin tannate in children with acute gastroenteritis.
Conclusion
The efficacy and safety of tannin albuminate and ethacridine lactate monohydrate for the treatment of acute diarrhea and the prevention of traveler's diarrhea are proven in randomized controlled trials. Several noninterventional studies substantiate the real-world effectiveness and tolerability of the fixed combination. Both active ingredients combine antidiarrheal, antimicrobial, mucosal protection, and spasmolytic properties. However, existing clinical data should be updated with larger, well-designed, placebo-controlled trials in adult and pediatric patient populations.
Keywords
Introduction
Diarrhea affects approximately two billion people worldwide each year. Although acute diarrhea is mostly self-limiting and well-treatable in European regions, it causes high numbers of doctor's visits and hospital admissions. 1 Diarrhea is characterized by an increased stool frequency combined with reduced stool consistency. The World Health Organization defines diarrhea as “the passage of three or more loose or liquid stools per day (or more frequent passage than is normal for the individual)”. 2 German guidelines define diarrhea more precisely as ≥3 defecations per day and a daily stool weight >250 g, combined with a decreased stool consistency.3 However, exact definitions vary between different clinical guidelines.
Diarrhea is a symptom of various diseases. Bacterial or viral gastrointestinal infections are the most common cause of acute diarrhea. These infections are generally associated with further clinical symptoms such as nausea, vomiting, abdominal pain and cramps, bloating, flatulence, or fever. 4 Other forms of acute diarrheal diseases include antibiotic-associated diarrhea, parasitic diarrhea, and traveler's diarrhea. It is estimated that more than 60% of tourists who visit developing countries may experience traveler's diarrhea. 5 Acute diarrhea is usually a self-limiting, short-term condition. Diarrhea lasting more than 4 weeks is defined as chronic. Common causes of chronic diarrhea include inflammatory bowel disease (IBD), irritable bowel syndrome, bowel resection, and long-term medication.
Maintaining adequate hydration is the main objective during diarrheal illnesses. Oral rehydration solutions should be the basic treatment for moderate to severe diarrhea, especially for populations particularly susceptible to dehydration, such as young children and older adults. Since diarrhea can be associated with substantial impairments in general wellbeing and quality of life, symptomatic treatment is often used in addition to oral rehydration. Symptomatic treatment with antidiarrheals such as antisecretory, antimotility, and adsorbent drugs helps reduce fluid loss, improve stool frequency and consistency, and shorten the duration of diarrhea. 6
The combination of tannins and ethacridine has been used for the prevention and symptomatic treatment of acute diarrhea for several decades. Tannins are polyphenolic compounds that are naturally occurring secondary metabolites found in a wide range of plants. For example, Quercus and Rhus species produce especially large amounts of tannins in galls after contact with specific insects. 7 Tannins can be divided into condensed and hydrolysable tannins.
Tannin Albuminate
Tannin albuminate is a stable, heat-formed complex of tannin and ovalbumin. The tannin used in tannin albuminate mainly contains hydrolysable tannins, such as gallotannins, which in turn mainly consist of esterified gallic acids (GA). First insights and experimental data on tannin albuminate as an antidiarrheal substance were published in the 1890s.8,9 The primary mode of action of tannins is based on a protein precipitation effect, which is mediated through the binding of proteins and macromolecules via hydrogen bonding and hydrophobic interactions.10,11 This effect leads to a condensation of the colloidal structure of the inflammatory altered intestinal mucosa by flocculation or gelling of the dissolved proteins - a so-called “astringent effect”. As the precipitated proteins impair the resorption of toxic substances, the intestinal mucosa is protected against further stress factors or other irritants. 12 Hence, hypersecretion of water decreases. Tannin albuminate has low solubility in acidic solutions. In the more neutral conditions of the duodenum, tannin is slowly released from the tannin-albumin complex. Therefore, tannin unfolds its astringent effect throughout the entire intestinal tract, from the small to the large intestine. 13
GA, a degradation product of gallotannin, may also play a crucial role in the mode of action of tannins. Some commensal bacteria of the intestinal microbiome produce tannase, a glycoprotein esterase, which can hydrolyze gallotannins to GA. 14 Several recent studies showed antibacterial, antifungal, anti-inflammatory, and antiviral properties of GA. According to in vitro studies, GA inhibits biofilm formation by regulating gene expression in various bacterial strains, including e.g. Escherichia coli, Staphylococcus aureus, and Shigella flexneri. Furthermore, antimicrobial activity was shown against Pseudomonas spp, Salmonella, and Enterobacteriaceae, as well as various dermatophytes.7,15 The mechanisms behind the antimicrobial effects are not clear yet. However, some studies postulate that a weakening of cell membranes, regulation of virulence factors’ gene expression or iron deprivation might contribute to the observed antimicrobial effects. First insights into anti-inflammatory properties have been gained by preclinical studies with Quercus infectoria galls and GA: GA was able to significantly reduce the expression of proinflammatory cytokines, such as tumor necrosis factor-α (TNF-α) and interleukin-6.15,16 Antiviral effects have been shown against influenza A (H1N1) and hepatitis C virus (HPC).17,18
Recent studies indicate that GA might also have prebiotic effects. Commensal bacteria of the gut microbiome are able to use GA or its degradation products as fermentation substrates. Thereby, GA might promote favorable changes in the intestinal microbiome, thereby leading to an increased production of short-chain fatty acids (SCFA). 14 Prebiotic effects of GA were shown in several recent studies. For example, in vitro digestion and fermentation assays using gut microbiota of healthy subjects showed a decrease of Bacteroides and an increase of Akkermansia and SCFA concentration after adding tannin to gut microbiota. 19 The intake of mango pulp (gallotannins and GA) increased the abundance of several Lactobacillus species and the levels of butyrate in the fecal microbiome of IBD patients. 20 These preliminary data suggest that GA might have positive effects on the microbiome and antimicrobial effects against several pathogens.
Ethacridine
Ethacridine lactate monohydrate has antiseptic-bacteriostatic properties against a range of pathogenic bacterial strains, including Staphylococcus aureus, Escherichia coli, several Salmonella strains, and Shigella. However, there is virtually no influence on the natural intestinal microbiome.21,22 Ethacridine lactate monohydrate also acts as an acetylcholine antagonist, resulting in spasmolytic effects on the smooth muscles of the intestine. 23 Thereby, ethacridine lactate monohydrate reduces abdominal cramps and slows the intestinal transit, supporting the resorption of water. Although of limited interest with regard to diarrhea, it should be noted that ethacridine was recently shown to have in vitro antiviral activity against SARS-CoV-2. Ethacridine impaired SARS-CoV-2 binding to human cells, mainly by inactivating viral particles. 24
The complementary mode of action of tannin albuminate and ethacridine lactate monohydrate is shown in Figure 1. Other complexations of tannin, such as gelatin tannate, are also used for the treatment of diarrhea. This narrative review provides a comprehensive overview of published efficacy/effectiveness and safety/tolerability data of tannin and ethacridine for the treatment of acute and chronic diarrhea, as well as the prevention of traveler's diarrhea.
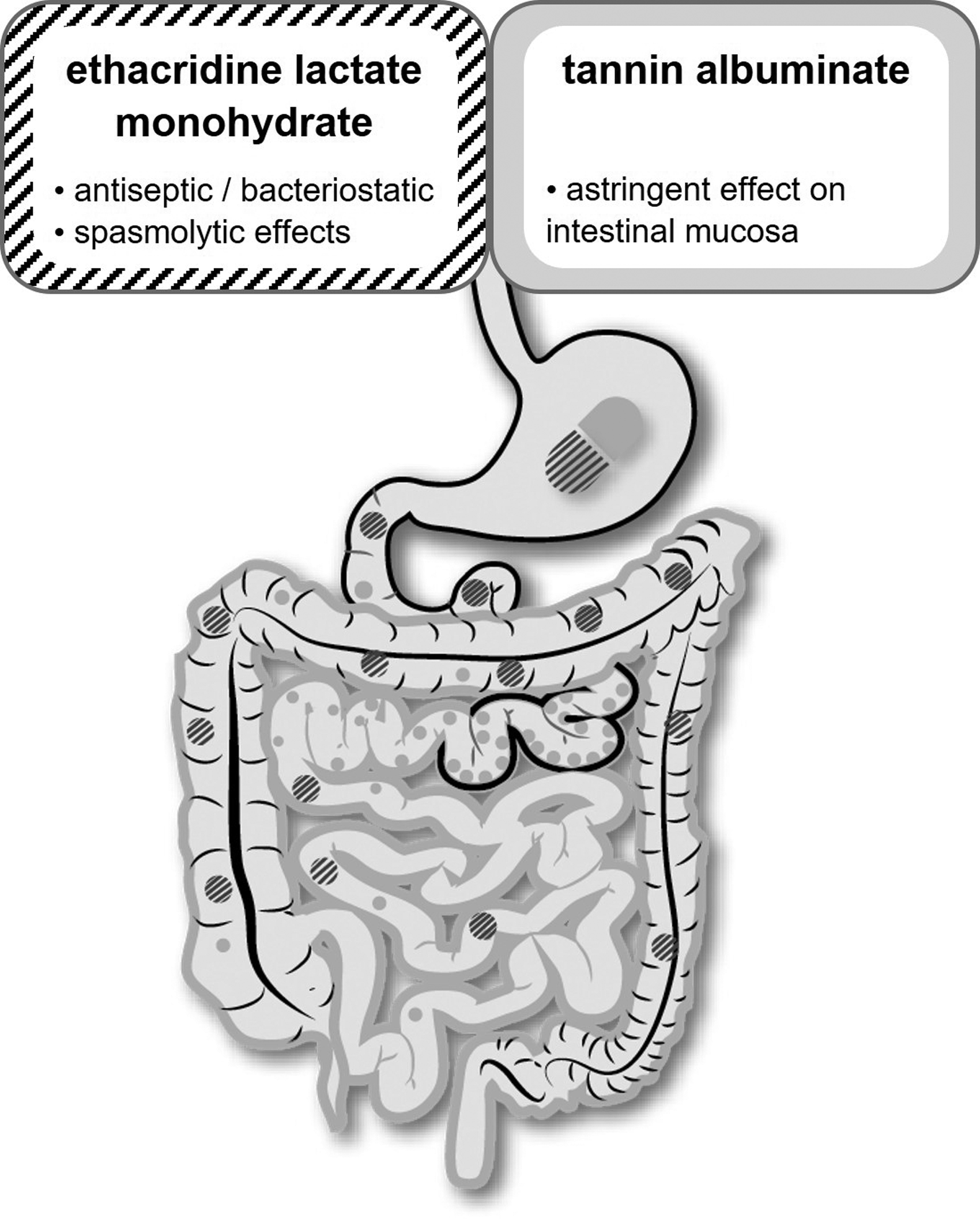
Schematic illustration of the mode of action of tannin albuminate and ethacridine lactate monohydrate in the gut. Grey: tannin albuminate; black-striped: ethacridine lactate monohydrate.
Methods
Search Strategy and Eligibility Criteria
We performed a literature search in MEDLINE and Embase using ProQuest Dialog from inception to September 2022. The search strategy is provided in Supplementary Appendix 1 and included all types of human clinical studies and reviews assessing tannins or ethacridine in the fields of diarrhea or other gastrointestinal diseases. Language was restricted to English and German. In this review, we included randomized controlled trials (RCT; placebo-controlled, reference-controlled, or untreated as control), noninterventional studies (NIS), and meta-analyses assessing the fixed combination of tannin albuminate and ethacridine lactate monohydrate (TA/ET) (1) for the treatment of acute diarrhea, (2) for the treatment of chronic diarrhea, and (3) for the prevention of traveler's diarrhea. Moreover, we included (4) meta-analyses assessing tannin in other complexations for the treatment of diarrhea. No exclusion criteria were applied.
For RCTs and meta-analyses evaluating the treatment of diarrhea, the primary outcome was defined as the improvement of diarrhea (improvement in stool frequency, stool consistency, or duration of diarrhea). Secondary outcomes included abdominal pain and adverse events. For NIS, the duration of diarrhea (number of days or reduction in stool frequency) was defined as the primary outcome. Global evaluation of effectiveness and tolerability were defined as secondary outcomes. For studies assessing the prevention of diarrhea, we defined the occurrence of diarrhea as the primary outcome.
Study Selection and Data Extraction
NR screened titles, abstracts, and full-text articles for study eligibility. The following information was extracted from each included study: study characteristics (design, year, sample size), patient characteristics (age group, in- or out-patients, etiology of diarrhea), details of interventions (active ingredients and doses) and the above-defined primary and secondary outcome results.
Results
The search identified 366 records, of which 13 were assessed as eligible for inclusion in this review (see Figure 2). Study characteristics are shown in Tables 1-4. Four reference-controlled studies and three NIS analyzed the effect of TA/ET on acute diarrhea (see Table 1). Two studies (1 RCT and 1 NIS) examined the effect of TA/ET on the prevention of traveler's diarrhea (see Table 3), and two further controlled studies investigated TA/ET in the treatment of chronic diarrhea due to IBD or radiation-induced colitis (see Table 2). All RCTs and controlled studies conducted with TA/ET included only adults. Children (1-18 years) were included in three NIS studying TA/ET in acute diarrhea. For gelatin tannate, two meta-analyses described the effects in children aged 0-18 years (see Table 4).

Flow chart of study selection.
Characteristics of Included Studies - Tannin Albuminate/Ethacridine Lactate Monohydrate (TA/ET) for the Treatment of Acute Diarrhea.

a Fisher's exact test.
b Five-point scale from “very good” to “unsatisfactory”.
c Diarrhea defined as more than three stools per day or watery stool consistency.
d Diarrhea defined as more than three liquid stools within 24 h.
Characteristics of Included Studies - Tannin Albuminate/Ethacridine Lactate Monohydrate (TA/ET) for the Treatment of Chronic Diarrhea.

* patient status (in/outpatient) not known
Characteristics of Included Studies - Tannin Albuminate/Ethacridine Lactate Monohydrate (TA/ET) for the Prevention of Travelers’ Diarrhea.

a More than three loose or watery stools within 24 h accompanied by another clinical symptom of enteric disease.
Characteristics of Included Studies—Meta-analyses for Gelatin Tannate (GT) in the Treatment of Diarrhea.

GT: gelatin tannate.
MD: mean difference.
CI: confidence interval.
Clinical Efficacy and Effectiveness
The antidiarrheal efficacy of TA/ET has been investigated in RCTs and NIS, including a total of more than 3 000 patients.
Treatment of Acute Diarrhea with TA/ET
Four RCTs investigated the efficacy of TA/ET in the treatment of acute diarrhea (see Table 1). In a reference-controlled, single-blind, multicenter study by Vorberg et al, 1978, 25 51 adult outpatients with gastroenteritis, enterocolitis, or summer diarrhea were treated with TA/ET (500 mg/50 mg; 2 tablets qid) or tannin albuminate (500 mg; 2 tablets qid) for 5 days. 25 The primary endpoints were improvements in stool frequency and consistency within the first 24 h of treatment. Stool frequency within the first 7-24 h was significantly reduced in the TA/ET group compared to the tannin albuminate group (2.7 vs 3.9). After 24 h, 23.1% of patients on TA/ET still had loose/liquid stools, compared to 81.8% of patients on tannin albuminate. Furthermore, TA/ET showed a significantly higher antispasmodic efficacy compared to tannin albuminate in patients with abdominal cramps. Within 24 h, abdominal cramps resolved in 90.5% of patients on TA/ET and 50% of patients on tannin albuminate.
A second RCT investigated the efficacy of TA/ET compared to the motility inhibitor loperamide in the treatment of acute, unspecific diarrhea. 26 Forty hospitalized, adult patients received either TA/ET (500 mg/50 mg, 2 tablets tid) or loperamide (2 mg, 1 tablet tid) for 4 days. Primary outcome measures were stool frequency, weight, and consistency. Both medications showed a marked antidiarrheal effect, with stool frequency and weight continuously decreasing in both groups. Stool frequency was reduced from 6.2 to 1.9 stools/day with TA/ET and from 7.0 to 2.2 stools/day with loperamide. No statistically significant differences were found between the treatment groups. The percentage of patients with liquid stools on day 5 was 0% with TA/ET and 15% with loperamide.
The efficacy of TA/ET in treating traveler's diarrhea was investigated against Carbo medicinalis in an RCT including 186 adult tourists visiting Turkey (Ziegenhagen et al, 1992 27 ). Patients with traveler's diarrhea received daily either TA/ET (500 mg/50 mg, 2 tablets tid) or Carbo medicinalis (250 mg, 2 tablets qid) until normalization of stool frequency. Patients treated with TA/ET showed a significantly quicker reduction of stool frequency within 3 days compared to patients treated with Carbo medicinalis (−9.7 vs −4.7; see Figure 3). The proportion of patients who reported moderate to severe abdominal pain was also reduced with TA/ET compared to Carbo medicinalis: About 80% of patients taking Carbo medicinalis recorded moderate to severe abdominal pain, whereas about 50% of patients treated with TA/ET stated either no or only mild abdominal pain.

Effects of tannin albuminate/ethacridine lactate monohydrate (TA/ET) and Carbo medicinalis on stool frequencies on days 1, 2 and 3 of diarrhea (modified from Ziegenhagen et al, 1992 27 ).
A further RCT compared the efficacy of TA/ET in treating traveler's diarrhea against the probiotic yeast Saccharomyces boulardii in tourists visiting Tunisia (Bruns et al, 1995 28 ). Sixty adult tourists with traveler's diarrhea received either TA/ET (500 mg/50 mg, 2 tablets tid) or S. boulardii (50 mg, 3 tablets qid) for 5 days. Both medications showed marked antidiarrheal effects. The mean duration of diarrhea was significantly lower with TA/ET compared to S. boulardii (33.8 h vs 51.4 h; p = .0394). In addition, TA/ET showed greater spasmolytic effects compared to S. boulardii: from day 1 to day 2, the proportion of patients with severe abdominal pain decreased from 28% to 0% in the TA/ET group and from 18% to 6% in the S. boulardii group.
In the above-described RCTs, TA/ET was compared to well-established antidiarrheal substances. TA/ET was either equivalent or superior to S. boulardii, Carbo medicinalis, loperamide, and tannin albuminate in the reduction of diarrhea.
Children were not included in any of the RCTs analyzing the effects of TA/ET in acute diarrhea. However, three NIS including more than 1 400 patients of all age groups (thereof >400 children aged 1-18 years) demonstrated the effectiveness and tolerability of TA/ET in real-life settings in children and adults.29,36,37 In each study, more than 90% of physicians rated the effectiveness and tolerability of TA/ET as very good to good. On average, diarrhea resolved within 2.5 days of TA/ET intake,36,37 and the mean number of stools per day was markedly reduced from initially 6.8 to 1.5 at the end of the treatment 29 (see Table 1).
Treatment of Chronic Diarrhea with TA/ET
Two clinical studies evaluated the efficacy of TA/ET in the treatment of chronic diarrhea caused by inflammatory bowel diseases (see Table 2). In a pilot study, patients with chronic diarrhea due to Crohn's disease received TA/ET (500 mg/50 mg, 2 tablets tid) for 5 days in addition to their basic Crohn's disease treatment, followed by a washout phase and a subsequent, second 5-day TA/ET treatment phase (Plein et al, 1993 30 ). One RCT compared the effects of TA/ET (500 mg/50 mg, 2 tablets tid) and loperamide (2 mg; 1 tablet tid) in treating chronic diarrhea due to ulcerative colitis or radiation colitis (Raedsch, 1995 31 ). In both studies, TA/ET treatment resulted in a clinically relevant reduction in stool frequency and improvement in stool consistency. However, well-designed, placebo-controlled studies with larger patient numbers are needed to further evaluate the efficacy of TA/ET in the treatment of chronic diarrhea.
Prevention of Traveler's Diarrhea with TA/ET
A randomized, double-blind, placebo-controlled trial investigated the efficacy of TA/ET in the prevention of traveler's diarrhea (Raedsch et al, 1991 32 ). Seventy-seven adult European tourists administered either TA/ET (500 mg/50 mg, 1 tablet bid) or placebo during a 16-day ship cruise in Egypt. The primary outcome was the occurrence of diarrhea, defined as three or more loose or watery stools within 24 h, accompanied by another clinical symptom of enteric disease. The prophylactic intake of TA/ET significantly reduced the incidence of traveler's diarrhea compared to placebo (52.9% vs 81.4%, p < .0125, see Table 3).
Furthermore, the effectiveness of TA/ET in the prevention of traveler's diarrhea was investigated in a large NIS including more than 1.300 tourists in Kenya. 33 The study compared tourists taking TA/ET (500 mg/50 mg, 1 tablet bid) and those taking no prophylactic medication. Only 12% of tourists taking TA/ET developed traveler's diarrhea compared to 50.5% of tourists without prophylactic medication (see Figure 4).

Incidence of traveler's diarrhea in tourists visiting Kenya taking no prophylactic medication or prophylactic tannin albuminate/ethacridine lactate monohydrate (TA/ET) (modified from Plentz, 1989 33 ).
Meta-analyses Assessing Tannin in Other Complexations for the Treatment of Diarrhea
Gelatin tannate, a complex of tannic acid and gelatin collagen, has been extensively studied in recent years. Two meta-analyses, published in 2019 and 2020, respectively, evaluated the evidence of gelatin tannate in the treatment of acute diarrhea and/or gastroenteritis in children (see Table 4). The meta-analysis of Florez et al, 2020, identified three RCTs that investigated the efficacy of gelatin tannate compared to either placebo or no treatment in treating diarrhea in children under 18 years of age. 35 The meta-analysis of Aloi and Mennini, 2019, identified three RCTs that investigated the effects of gelatin tannate on diarrhea-related outcomes compared to oral rehydration solution and/or placebo in children aged 0-12 years. 34 Whereas Florez et al, 2020, did not find a significant effect of gelatin tannate on stool frequency or diarrhea duration (at day 2), Aloi and Mennini, 2019, found that gelatin tannate improved stool frequency and consistency after 12 h.34,35 However, a clear effect on the duration of diarrhea was not shown. Further clinical trials are needed to validate the antidiarrheal properties of gelatin tannate.
Safety and Tolerability
TA/ET showed a very good safety profile in all RCTs. Only one adverse event (strong meteorism) was reported for TA/ET among all RCTs, which could not be attributed to the intake of TA/ET with certainty. However, the RCTs were not primarily designed for safety evaluation. In all NIS, including more than 1.400 patients, tolerability was also rated as very good to good by physicians and patients. Only five adverse events were reported, all of which were related to a lack of acceptance by children due to the bitter taste of TA/ET tablets. Four of the RCTs 31 25,26,30 analyzed clinical laboratory parameters, including parameters of the hematopoietic system, liver and kidney function, or urinary status. No pathological changes or abnormalities were reported in any of these studies. Both active ingredients of TA/ET - tannin albuminate and ethacridine lactate monohydrate - act locally in the gut lumen and on the gut wall and are not absorbed to a relevant extent. Thus, systemic effects are not expected.
Conclusions
The efficacy/effectiveness, safety, and tolerability of TA/ET have been evaluated in several RCTs and NIS including a total of more than 3 000 children and adults. The results demonstrated that TA/ET reduces the duration of diarrhea and concomitant abdominal pain and, when taken prophylactically, reduces the risk of traveler's diarrhea. Moreover, TA/ET showed a very good safety/tolerability profile. However, larger, well-designed, placebo-controlled trials are needed to confirm these findings, especially in pediatric patient populations and chronic diarrhea.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X231170998 - Supplemental material for Antidiarrheal Properties of the Combination of Tannin Albuminate and Ethacridine Lactate - A Narrative Review
Supplemental material, sj-docx-1-npx-10.1177_1934578X231170998 for Antidiarrheal Properties of the Combination of Tannin Albuminate and Ethacridine Lactate - A Narrative Review by Andera Salama-Müller and Nadine Roese in Natural Product Communications
Footnotes
Acknowledgements
The authors thank Michaela Ruhmann, PhD, an employee of MEDICE Arzneimittel Pütter GmbH & Co. KG., for editorial support.
Author Contributions (Roles)
N.R. conducted the primary literature search, which was critically reviewed by A.S-M. Both authors discussed the results and contributed to the final manuscript. Both authors reviewed and approved the final manuscript.
Declaration of Conflicting Interests
The present paper was supported by MEDICE Arzneimittel Pütter GmbH & Co. KG, Germany, who provided writing and editorial support for this publication. N.R. reports employment at MEDICE Arzneimittel Pütter GmbH & Co. KG. A.S-M. reports personal consulting fees from MEDICE Arzneimittel Pütter GmbH & Co. KG for the submitted work.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
