Abstract
This study examined the molecular processes behind the effects of vanillic acid (VA) on right ventricular (RV) hypertrophy and function in rats with monocrotaline (MCT)-induced pulmonary arterial hypertension (PAH). There were 40 male Sprague‒Dawley (SD) rats that were separated into 4 groups: Control, PAH, MCT + VA (50 mg/kg/d), and MCT + VA (100 mg/kg/d). Male SD rats were injected with MCT once under the skin to create the PAH model (40 mg/kg). RV morphological properties were evaluated using Masson and hematoxylin and eosin (H&E) staining. Echocardiography was used to evaluate RV functioning and right ventricle–pulmonary artery (RV-PA) coupling. In addition, Rho-associated protein kinase (ROCK) pathway-related factors were evaluated using Western blotting. Enzyme-linked immunosorbent assay (ELISA) was used to detect inflammatory markers as well as atrial natriuretic peptide (ANP) and brain-type natriuretic peptide (BNP) in the blood of PAH rats. As a result, VA effectively reduced the development of RV cardiomyocyte hypertrophy and fibrosis in PAH rats; levels of ANP, BNP, and inflammatory markers in the blood of PAH rats were also significantly decreased by VA intervention. Additionally, VA enhanced RV functioning and RV-PA coupling in PAH rats. In response to VA, the expression of proteins related to the ROCK pathway (ROCK1, ROCK2, NFATc3, P-STAT3, and Bax) was downregulated, whereas Bcl-2 expression was elevated. This study found that VA could attenuate RV remodeling and improve RV-PA coupling in PAH rats. RV remodeling and dysfunction may be linked to the dysregulation of the ROCK pathway, and the protective action of VA on RV function may be due to a block in the ROCK signaling pathway or its downstream signaling molecules.
Introduction
Right ventricular (RV) hypertrophy and remodeling are symptoms of pulmonary arterial hypertension (PAH), which is characterized by the remodeling of the pulmonary vascular system and a gradual rise in pulmonary vascular load. 1 Given its sensitivity to afterload changes, the RV is a thin-walled construction. The left conical heart is encircled by a depression with a crescent form. 2 The RV first increases contractile activity and then undergoes hypertrophy to counteract the rise in pulmonary arterial pressure caused by the afterload. 3 As the PH worsens, the RV will go from compensating to decompensating, which will cause tricuspid regurgitation, volume overload, and pressure overload. 2 RV dilation becomes more pronounced resulting in decreased RV contractility 4 and eventually leading to RV failure and death.5,6 RV function is an important prognostic predictor in different types of PH. Currently, approved drugs primarily targeting pulmonary artery vasoconstriction for PAH treatment have failed to significantly improve long-term survival, 7 and even if the pulmonary hemodynamic condition has improved, 6 the prognosis of PAH patients experiencing persistent impaired RV function remains poor.8,9 However, there are no RV-focused treatments. RV-directed therapy would enhance the RV's capacity to compensate for the rise in pulmonary artery pressure, prevent decompensated responses and likely enhance RV function as a prognosis for PH patients.
Vanillic acid (VA) is a natural phenolic aromatic acid. 10 It has been documented that VA has antiapoptotic, 11 antibacterial, anti-inflammatory, antitumor, and hepatoprotective effects. 10 Our earlier research on monocrotaline (MCT)-induced PAH rats demonstrated that VA exerted protective effects on pulmonary vascular remodeling by enhancing nitriic oxide (NO) signaling pathways. 12 However, its impact on the RV in rats with MCT-induced PAH is still unknown. In this study, we examined VA’s positive effects on RV hypertrophy and function.
Materials and Methods
Materials
VA (LOT: J13A11T120848) and MCT (LOT: K20N11K131409) were purchased from Shanghai Yuanye Bio-Technology Co., Ltd, enzyme-linked immunosorbent assay (ELISA) kits from Elabscience Biotechnology Co., Ltd (Hubei, Wuhan, China), anti-STAT3 (ab76315), anti-Bcl-2 (ab196495), anti-Bax (ab32503), anti-ROCK1 (ab45171), anti-ROCK2 (ab71598), and anti-beta actin (ab8226) from Abcam (Cambridge, Massachusetts, USA), and anti-NFATc3 (18222-1-AP) from Proteintech (Chicago, USA).
Animals
The Institutional Animal Care and Use Committee at Qinghai University approved the experimental protocol in accordance with the regulations for the management of animals issued by the Ministry of Health of China. Sprague‒Dawley (SD) rats were acquired from Xi’an Jiaotong University's Experimental Animal Center (permission number SCXK (Shan) 2018-001). Forty male SD rats (weight = 170 ± 20 g, 7 weeks old) were separated into 4 groups at random (n = 10): control, MCT (40 mg/kg MCT subcutaneous injection singly), MCT + VA (50 mg/kg/d) and MCT + VA (100 mg/kg/d). VA was dissolved in 0.5% sodium carboxymethylcellulose solution. VA (50 mg/kg/d, 100 mg/kg/d) and sildenafil (30 mg/kg/d) were given orally to the rats throughout a 4-week period. A comparable amount of 0.5% sodium carboxymethylcellulose solution was given to the rats in the control and MCT groups.
Echocardiography
Using Vevo 3100LT imaging equipment, echocardiography was performed (probe M250, Visual Sonics). Using a Small Animal Anesthesia Machine – Basic Model (SN: H1649701-023 L) with the specification Model R500IP at room temperature, the rats’ hair was briefly removed from their chests before they were anesthetized with a 2% isoflurane/oxygen mixture. The two-dimensional model from the echocardiography was used to estimate the RV diameter during diastole (RVID-Dia). From the apical 4-chamber perspective, tricuspid annular plane systolic excursion (TAPSE) was quantified using two-dimensional and M-modes. From the parasternal short-axis perspective, the pulmonary valve annulus during pulse wave Doppler inquiry was used to calculate the pulmonary artery acceleration time (PA-AT), mean pulmonary artery pressure (MPAP), and preejection time (PET). RV end-systolic area (ESA), RV end-diastolic area (EDA), and E/A were measured in the RV-focused apical 4-chamber. The formula for fractional area change (FAC%) was (EDA−ESA)/ESA˟100%. The echocardiographic examination was followed by the calculation of hemodynamic parameters.
Morphometric Evaluation of RV Tissues
The RV tissues were then embedded in paraffin and sectioned (5 m, RM2135 paraffin microtome; Leica) after being fixed for 48 h in 4% paraformaldehyde. Hematoxylin and eosin (H&E) were used to examine the morphological changes. The H&E-stained cardiomyocyte area was quantified using Image-Pro Plus 6.0.
To assess the degree of fibrosis in the RV tissues, Masson's trichrome dye was used (fibers of collagen dye blue). Images were captured using a digital microscope BA400 with a digital interface from Motic China Group Co., Ltd in Xiamen, China. ImageJ was used to quantify the myocardial fibrosis Masson stains.
RV Inflammatory Markers and Remodeling Factors
A serum index analysis was performed in SD rats using ELISA. SD rats were anesthetized by intraperitoneal injection using 1.2 mg/kg urathane. Blood was obtained from the abdominal aorta, and serum was obtained after centrifugation. The ELISA kit was used according to the experimental procedure, and the OD value was measured at 450 nm using an enzyme standardization instrument. The concentrations of IL-6, TNF-α, IL-1β, atrial natriuretic peptide (ANP), and brain-type natriuretic peptide (BNP) in the samples were calculated by plotting standard curves.
Western Blotting
Western blotting was used to examine the protein expression levels of ROCK1, ROCK2, NFATC3, P-STAT3, Bax, and Bcl-2 in RV tissues. To homogenize snap-frozen, RV tissues, RIPA buffer containing protease inhibitors was used. Following centrifugation (13,000 rpm, 20 min), the protein content in the supernatant was assessed using a BCA protein assay kit (Beyotime Institute of Biotechnology). The proteins were separated using SDS-PAGE (30 g•lane) and then transferred to polyvinyl difluoride membranes. Anti-ROCK1 (1:4000), anti-ROCK2 (1:1000), anti-NFATc3 (1:4000), anti-P-STAT3 (1:2000), anti-Bax (1:2000), anti-Bcl-2 (1:1000), and anti-actin (1:5000) antibodies were incubated on the membranes overnight at 4 °C after blocking with TBST containing 5% nonfat dry milk. An enhanced chemiluminescence (ECL) kit was used to view the membranes after they had been treated with goat, anti-mouse, or rabbit secondary IgG antibodies at a dilution of 1:5000 (Beyotime Institute of Biotechnology). Actin was used as a loading control. Blots were quantified using ImageJ.
Statistical Analysis
SPSS 27.0 software was used to examine the data (SPSS, Inc.). Statistical data were presented as the means ± SDs. Multiple comparisons were performed using either Dunnett's test or Student–Newman‒Keuls test after one-way analysis of variance (ANOVA). A P < .05 was used to indicate statistical significance.
Results
Effect of VA on RV Function in PAH Rats
The impact of VA on RV function was studied using noninvasive echocardiography. In the MCT group, PA-AT was dramatically reduced after 28 days. Intervention with VA increased PA-AT significantly compared to the MCT group, indicating that VA could improve right heart output pressure and reduce pulmonary artery pressure (P < .05, Figure 1A and C). Meanwhile, mPAP data from the echocardiographic (color mode) calculations increased among the MCT group (P < .05 vs control group, Figure 1D). VA attenuated the increase in mPAP induced by MCT (P < .05 vs MCT group, Figure 1D). VA increased PA-AT and improved mPAP (P < .05 vs MCT group, Figure 1A, C, and D). Additionally, RVID-Dia values increased in the MCT group, but decreased after VA intervention (P < .05 vs MCT group, Figure 1B and E), which indicates that VA could attenuate right ventricular dilatation in rats with MCT induced PAH.

Vanillic acid (VA) restored right ventricular (RV) function in pulmonary arterial hypertension (PAH) rats. The following groups of rats were established: Control group, PAH group (PAH, to replicate PAH, male Sprague‒Dawley [SD] rats were given a single subcutaneous injection of monocrotaline [40 mg/kg]), and momnocrotaline (MCT) + VA groups (monocrotaline was administered subcutaneously to rats once, and VA was also administered [50 mg/kg/d, 100 mg/kg/d] for 4 weeks; n = 5). Analysis of the pulmonary artery acceleration time (PA-AT) in rats with PAH in (A and C). (B) The echocardiogram E/A ratio is an indicator of the heart’s diastolic function. Peak E is the first blood flow peak into the right ventricle from the right atrium while peak A is the second blood flow peak into the right ventricle from the atrium. Using echocardiography, the mean pulmonary artery pressure (MPAP) was assessed after 4 weeks (D and F).
Compared to controls, an increase in the E/A value, which indicated reduced right ventricular function, was noticed in the MCT group (P < .05 vs MCT group, Figure 2A). After VA intervention, the E/A value was reduced in contrast to the MCT group (P < .05 vs MCT group, Figure 2A) (Table 1), indicating that VA could improve right ventricular function. An insignificant decrease in TAPSE was observed in the MCT group. After intervention with VA, TAPSE increased compared to the MCT group. (P < .05 vs MCT group, Figure 2B and D), indicating that VA could improve right ventricular ejection and systolic function.
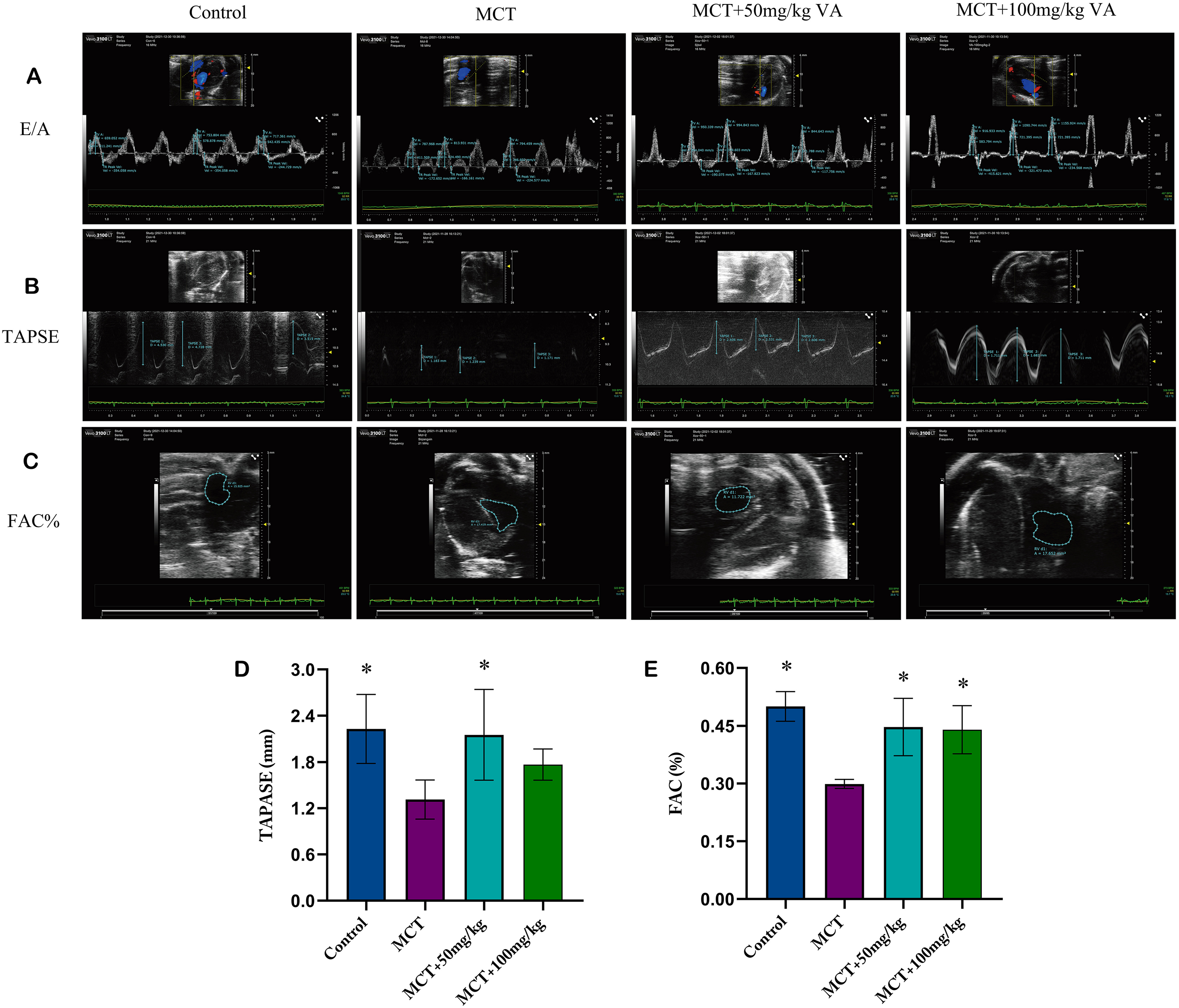
The effect of vanillic acid (VA) on right ventricular (RV) function in pulmonary arterial hypertension (PAH) rats (n = 5). Analyses of RVID-Dia in PAH rats are shown in (A and D). (B and E) Cardiac parameters of PAH rats are evaluated with TAPSE. ESA/ESA*100% is a formula for calculating the fractional area change (FAC) in (C and F). After 4 weeks, RVID-Dia, TAPSE, and FAC% were evaluated by echocardiography. The detection distance is highlighted by the blue dotted lines from A to C.
Effect of Vanillic Acid (VA) Intervention on Right Ventricular (RV) Function in PAH Rats (n = 5).

PAH versus control group, P < .05.
Drug group versus PAH group, P < .05.
Results were expressed as mean ± SD.
Abbreviations: A represents late diastolic atrial contraction; E stands for passive filling of the ventricles in early diastole; E/A: evaluation of right ventricular function; FAC%: fractional area change, calculated as (end-diastolic area−ESA)/ESA*100% (ESA represents end-systolic area); MCT, monocrotaline; mPAP: mean pulmonary artery pressure (assessed by echocardiography); PAH, pulmonary arterial hypertension; PAT/PET: ratio of right ventricular preejection time to acceleration time evaluation of right heart systolic function; TAPSE: tricuspid annular plane systolic excusion, obtained from echocardiography and estimation of right ventricular function.
For PAH to occur, there must be a right ventricle–pulmonary artery (RV-PA) link between RV contractility and pulmonary arterial load. In this study, the efficiency was assessed of the RV-PA coupling assessment indices FAC (%), TAPSE/PASP, FAC/mPAP, and PASP/ESA ratios. In the MCT group, as shown in Table 1, the FAC (%), TAPSE/PA-AT, TAPSE/PASP, and FAC/mPAP ratios all showed a substantial decline. These indices significantly increased after intervention with VA (P < 0.05 vs MCT group, Table 1), indicating that VA could improve RV-PA coupling in MCT-induced PAH rats.
Effect of VA on Protecting Against Structural Remodeling of the RV
When compared to the control group, the MCT group's Fulton index RV/(LV + S) ratio was significantly higher (P < .05, Figure 3E). After VA intervention, the RV/(LV + S) ratios were considerably lower than those in the MCT group (P < .05, Figure 3E), indicating that VA showed a significant improvement in right heart hypertrophy in PAH rats (Figure 3E). Additionally, H&E staining revealed that the RV myocyte cross-sectional area in PAH rats was significantly enlarged. The hypertrophy of RV myocytes was significantly inhibited in the rats after VA intervention compared to the MCT group (P < .05, Figure 3A and C). Meanwhile, Masson staining showed that myocardial fibrosis (blue colloid region) was intensified in MCT-induced PAH rats, and lysis was also observed in some regions (P < .05, Figure 3B and D). However, the degree of fibrosis was significantly inhibited after VA intervention (P < .05, Figure 3B and D).

Effect of vanillic acid (VA) on right ventricular (RV) structure in pulmonary arterial hypertension (PAH) rats (n = 5). (A) Sample micrographs of the morphological changes of the RV tissue in PAH-exposed rats taken at 400× magnification; scale bar, 40 m. (B) Illustrations of RV tissue under a microscope with Masson’s trichrome staining (200), scale bar = 100 m. (C) Statistical evaluation of the cross-sectional RV cardiomyocyte area. (D) Statistical evaluation of RV tissue hypertrophy; RV/(LV + S) is shown by the symbol (E).
VA Inhibits Inflammation and Cardiac Remodeling Factors
The experimental results showed that brain natriuretic peptide (ANP and BNP) was significantly elevated in the MCT model group compared to the control group, and the ANP and BNP in the blood SD fluid of PAH rats after VA intervention were significantly reduced (Figure 4D and E). Similarly, the inflammatory markers IL-6, TNF-α, and IL-1β were significantly elevated in the rat's blood in the MCT model group and then significantly decreased after VA intervention (Figure 4A, B, and C).

Effect of vanillic acid (VA) on inflammatory markers and cardiac remodeling factors in pulmonary arterial hypertension (PAH) rats (n = 5). (A, B, and C) indicate the analysis of changes in the inflammatory markers IL-6, TNF-α, and IL-1β in rat serum. (D and E) Analysis of changes in brain natriuretic peptide (ANP and BNP) in rat serum, respectively.
Molecular Mechanism by Which VA Protects RV Function
Many disorders, including hypertension and pulmonary hypertension, are caused by an increase in ROCK activity.13,14 ROCK1 and ROCK2 are involved in the etiology of cardiac hypertrophy and fibrosis. Therefore, this study was conducted based on ROCK pathway-related protein expression. The findings showed that ROCK1 and ROCK2 protein expression was much lower in the RV of VA-treated rats than in the RV of PAH-treated rats. (P < .05 vs MCT group, Figures 5A, B, and C). In addition, the levels of the 2 downstream transcription factors P-STAT3 and NFATc3, which are regulated by the ROCK signaling pathway, 15 were similarly reduced compared to those in the MCT group treated with VA (P < .05 vs PAH group, Figure 5A, D, and E).

Effect of vanillic acid (VA) on P-STAT3, NFATc3, ROCK1, and ROCK2 protein expression in right ventricular (RV) tissue in pulmonary arterial hypertension (PAH) rats (n = 4). P-STAT3, NFATc3, ROCK1, and ROCK2 protein bands are shown in (A). (B) A statistical examination of the amount of P-SATA3 expression in RV tissue. The amount of NFATc3 expression in RV tissue was statistically analyzed in (C). (D) Statistical evaluation of the ROCK1 expression level in RV tissue. The amount of ROCK2 expression in RV tissue was statistically analyzed in (E).
Cardiomyocyte apoptosis is a major cause of abnormal cardiac function, and it causes functional remodeling of the heart16–18 The experiment was conducted based on the common apoptotic protein Bax and the apoptosis-inhibiting Bcl-2 protein. The experimental data demonstrated that the Bax protein expression level was downregulated in RV tissues of PAH rats after VA intervention, and conversely, the Bcl-2 protein expression level increased, while the Bax/Bcl-2 ratio decreased (P < .05 vs PAH group, Figure 6), indicating that VA could inhibit cardiomyocyte apoptosis in MCT-induced PAH rats.

Effect of vanillic acid (VA) on Bcl-2 and Bax protein expression in righr ventricular (RV) tissue in pulmonary arterial hypertension (PAH) rats (n = 4). Proteins for Bcl-2 and Bax are shown in (A). The amount of Bax expression in RV tissue was statistically analyzed in (B). The Bax/Bcl-2 ratio is shown in (C).
Discussion
RV ventricular remodeling due to pulmonary hypertension is characterized by centripetal hypertrophy in the early stage to compensate for the PAH-induced increase during afterload.6,19 As PAH continues to worsen, the process of RV remodeling is accompanied by the onset of cardiac fibrosis and myocardial hypertrophy. A lesion that causes systolic and diastolic dysfunction would be severe enough to lead to decompensated and eventual death from heart failure19–21 Meanwhile, the transfer of tension during contraction depends on collagen, but excessive collagen fibrosis may lead to coupling disorders and impaired myocardial contractions. 22 Considering our previous finding on the protective effect of VA (50 mg/kg, 100 mg/kg) in ameliorating MCT-induced PAH and pulmonary arterial remodeling, 12 in this study, we investigated whether VA would improve right ventricular function. Thus, we investigated the changes in RV morphological structure and function in MCT-induced PAH rats by VA and further investigated the underlying molecular mechanisms.
Our findings demonstrate that VA prevented the MCT-induced decrease in PA-AT, indicating that VA intervention could reduce pulmonary arterial pressure. Additionally, we found that VA could prevent the increase in RVID-Dia in MCT-induced PAH rats, which indicates that VA could attenuate RV dilatation in PAH rats. Combining the reliable findings of RV morphological structure and Fulton's constant, we discovered that VA administration effectively prevented RV cardiomyocyte enlargement and improved fibrosis (P < .05 vs PAH group, Figure 3A and C).
ANP and BNP infusion results in improvement of pulmonary hemodynamics in humans.23,24 Elevated natriuretic peptides in the blood of patients with PAH cause derangements in their cardiac function. 25 Clinical experimental validation of PAH has shown that inflammatory factors such as IL-1β and IL-6, as they are elevated in the blood of PAH patients, are positively correlated with PAH mortality,26,27 while at the same time, these classical inflammatory factors are closely associated with RV dysfunction caused by PAH. 28 ANP and BNP are increased when right ventricular pressure is high in patients with PAH, and BNP levels are also strongly associated with mPAP and pulmonary vascular resistance (PVR).24,25 As shown in this experiment, VA significantly reduced the levels of ANP and BNP in the blood of PAH rats (P < .05 vs PAH group, Figure 4D and E). Similarly, the data suggest that VA also has an inhibitory effect on inflammatory markers (P < .05 vs PAH group, Figure 4A, B, and C). The effect may be to promote pulmonary vasodilation, reduce the inhibition of RV structural remodeling by the production of inflammatory factors, reduce RV afterload, or to attenuate myocardial hypertrophy. However, our data on cardiac function showed a significant improvement; so we conclude, in conjunction with cardiac function, that VA may have a direct effect on RV. However, the specific evidence needs to be verified with the pulmonary artery ligature technique. 29
RV function is commonly assessed using the EES/EA ratio in invasive pressure‒volume loops, which is crucial for PAH. 30 FAC (%), TAPSE/PASP, FAC/mPAP, and PASP/ESA ratios based on echocardiographic data were also found to be useful indices for evaluating RV function.18,31 FAC and TAPSE are critical markers of RV systolic activity that are measured by echocardiography and are reliable indicators for prognosis of PAH patients. 32 As shown in Figure 2 (B) and (D), VA intervention could elevate the TAPSE index to improve the contraction and ejection of blood from the RV. 31 The FAC% value [converted by the equation (end-diastolic area−ESA)/ESA×100%] was decreased, indicating that VA could reduce the RV load and improve the compensatory situation.6,33 E/A, calculated as the ratio of peak early filling velocity (E) to late peak atrial filling velocity (A), was used as an indicator of diastolic function.34,35 The ratio was noticeably lower in the rats from the VA group (Figure 2A, P < .05 vs MCT group, Table 1), indicating a decrease in diastolic function, which can be matched with RVID-Dia.
In most cases, RV-PA coupling refers to the dynamic adjustment of the RV's systolic function to the afterload. 30 RV function is ensured by proper RV-PA coupling, which enhances RV efficiency and guarantees optimum cardiac output. 30 As PAH continues to progress, the RV enhances contractility to adapt to the increasing afterload. 31 However, as PAH continues to develop, RV-PA coupling gradually declines. The TAPSE/PAT from noninvasive echocardiographic data showed that VA had a positive effect on RV-PA coupling in PAH rats. Other coupling data TAPSE/mPAP, FAC/mPAP, and FAC% all showed that VA improved RV-PA coupling (P < .05 vs PAH group, Table 1). Based on the above assessment, we concluded that RV function in PAH rats is significantly protected by VA.
VA has been shown to protect against RV remodeling and functional compensation in PAH rats, and we further explored its mechanism of action. According to studies, individuals with PAH, cardiac failure, cardiac remodeling, and stable angina pectoris have increased ROCK protein expression levels. The Rho/ROCK pathway has reportedly been connected to cardiac fibrosis and cardiomyocyte hypertrophy.36–38 In this study, we discovered that MCT-induced PAH rats had considerably greater levels of ROCK1 and ROCK2 protein expression than control rats (P < .05, vs MCT group, Figures 5A, B, and C), and their expression was inhibited by VA. Additionally, we discovered that VA might inhibit the transcription factors STAT3 and NFATc3, which are involved in cardiac remodeling (P < .05, vs MCT group, Figures 5A, D, and E) and are regulated by the ROCK signaling pathway. 15 VA reduced RV hypertrophy and improved RV function, which could be related to the regulation of the ROCK pathway. In the latest research, we found a considerable rise in the Bax/Bcl-2 ratio in the MCT group. The Bax/Bcl-2 ratio and Bax expression both significantly decreased after VA treatment, in contrast to a large rise in Bcl-2 expression (P < .05 vs MCT group, Figure 6), indicating that VA could inhibit apoptosis of myocardial cells. Additionally, it has been proposed that the death of cardiomyocytes is mediated through the ROCK signaling pathway. 38 Bax/Bcl-2-mediated apoptosis can activate the ROCK signaling pathway.36,37 VA may thus have an inhibitory impact on the ROCK signaling pathway due to its anti-apoptosis function.
This study revealed that VA significantly reduced RV hypertrophy and enhanced RV function in PAH rats. In the experimental model of PAH, right ventricular failure is secondary to pulmonary vascular system disease. Therefore, the improvement in right ventricular function with the current treatment may be secondary to the effect of afterload reduction on the pulmonary vasculature and is a valid consideration in PAH prognosis. Although animal models of pulmonary hypertension and pressure loading are important for studying the pathophysiology of RV, PAH pathology models cannot be used to study the right ventricular response independent from pulmonary vascular effects.29,39 Protecting the structure and function of the right ventricle by reducing afterload or by acting directly on the myocardium is a key target for the treatment of pulmonary hypertension. The real answer to whether or not it acts directly on the myocardium alone needs to be demonstrated with pulmonary artery ligation experiments.1,29 Studies have shown that VA has a protective impact on RV function by inhibiting the ROCK signaling pathway and the signaling molecules that it produces downstream. VA’s suppression of the ROCK signaling pathway may prevent cardiac fibrosis, promote cardiomyocyte enlargement, and prevent cardiomyocyte apoptosis.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research and/or authorship of this article: This work was supported by the National Natural Science Foundation of China (grant numbers 81860768, 32060088, and 82060786), Applied Basic Research Project of Qinghai Province of China (2020-ZJ-784), West Light Foundation of the Chinese Academy of Sciences, and Qinghai Province “Kunlun Talents, High-end Innovative and Entrepreneurial Talents” Project.
Ethical Approval
The Institutional Animal Care and Use Committee at Qinghai University, China, authorized this study.
Statement of Human and Animal Rights
The Institutional Animal Care and Use Committee at Qinghai University, China, provided oversight for all animal experimentation procedures.
Informed Consent
Informed permission is not needed, and there are no human subjects in this article.
