Abstract
Pulmonary arterial hypertension (PAH) is a severe progressive disease characterized by elevated mean pulmonary arterial pressure, right ventricular hypertrophy, and eventual progression to right heart failure and death. This study aimed to examine the effect of the natural product vanillic acid (VA) on monocrotaline (MCT)-induced PAH in rats. The arginase inhibitory activity and enzyme kinetic reaction of VA were also investigated. The results showed that VA could improve pulmonary arterial pressure, pulmonary artery vascular remodeling, and right ventricular remodeling induced by MCT in rats and reduce the degree of pulmonary tissue fibrosis. Moreover, VA downregulated the gene and protein expression levels of Hif-2α, Hif-1β, and Arg2 and increased the P-eNOS/eNOS levels, thus increasing nitric oxide (NO) levels in PAH rats. Furthermore, VA was determined to be a mixed competitive arginase inhibitor with an IC50 of 26.1 μM. In conclusion, the arginase inhibitor VA exerted protective effects on MCT-induced PAH and pulmonary vascular remodeling by enhancing NO signaling pathways.
Keywords
Introduction
Arginase is a metalloenzyme that catalyzes the hydrolysis of L-arginine to L-ornithine and urea. 1 Arginase exists as two isoforms: arginase I, a cellular enzyme that is part of the liver urea cycle, and arginase II, which is located in the mitochondria. They are structurally similar, with more than 50% homology of amino acid residues and 100% homology in the key region of the catalytic L-arginine metabolic function. 2 Arginase isoforms are expressed in the aorta, carotid artery, pulmonary artery, and many other vessels.3-6
Nitric oxide (NO), a vasodilatory factor, is generated from the conversion of L-arginine by eNOS. The inhibition of L-arginine transport in endothelial cells by arginase may further decrease the substrate utilization of eNOS.7-9 Excess arginase then causes a decrease in endothelial NO bioavailability, causing endothelial cell dysfunction and further triggering the development of various diseases, such as pulmonary arterial hypertension (PAH). 7
PAH is a severe progressive disease characterized by elevated mean pulmonary arterial pressure, right ventricular hypertrophy, and eventual progression to right heart failure and death. 10 Studies have shown that the NO pathway is one of the key pathways in the pathophysiology of PAH and provides multiple molecular targets, including arginase, eNOS, and NO, that play a central role in the regulation of vascular tone in the pulmonary circulatory system. 11 The beneficial effect of arginase inhibition has been demonstrated in pulmonary hypertension.12-14
Vanillic acid (VA) is a phenolic compound that is widely distributed in edible plants and fruits.15,16 It has been associated with a variety of pharmacological activities, including antioxidant, antihypotensive, cardioprotective, hepatoprotective, and antiapoptotic.17-20
In our previous study, the arginase inhibitory properties of phenolic compounds were reported. Hence, VA was chosen for our study since there are no investigations on its arginase inhibitory properties and protective effects against PAH to date. In this study, we concentrated on the effect and mechanism of VA against monocrotaline (MCT)-induced PAH in rats, and we also sought to reveal the arginase inhibitory properties of VA.
Results
Effect of VA on Hemodynamic and Hematological Indices in PAH Rats
On day 28, the right ventricular systolic pressure (RVSP) in the model group was 46.20 ± 6.22 mm Hg, which was significantly higher than that in the control group (19.62 ± 1.33 mm Hg, P < .05). After treatment with VA (50 and 100 mg/kg/d), the RVSP levels were significantly lower than those in the MCT model group (P < .05, Figure 1A) at 33.43 ± 5.62 and 34.49 ± 2.69 mm Hg, respectively.

Effect of vanillic acid on hematological indices in PAH rats. (A) Five groups of RVSP waveform combination diagrams. (B) RVSP, indicates RV systolic pressure. (C) RV/BW, indicates the ratio of RV to body weight (BW). (D) RV/LV + S, indicates the ratio of RV weight to the left ventricle (LV) with septum (S) weight (*P < .05 vs MCT, n = 10.)
Body weight was recorded every 4 days during the experiment, and the body mass of rats in the control group increased from 171.2 ± 13.82 g to 327.22 ± 21.29 g within 28 days. Compared with the control group, right ventricle/body weight (RV/BW) was significantly increased in the MCT model group (P < .05, Figure 1C). The RV/BW in the VA intervention group (50 and 100 mg/kg/d) was decreased to 1.04 and 1.31, respectively, which were significantly lower than that in the MCT model group (P < .05, Figure 1C). Compared with the control group, RV/(LV + S) was significantly increased in the MCT model group (P < .05, Figure 1D). RV/(LV + S) decreased to 0.52 and 0.59 in the VA intervention group (50 and 100 mg/kg/d), respectively, which were significantly lower than that in the MCT model (P < .05, Figure 1D). Additionally, except for the ratio of liver weight to body weight in the VA intervention group (25, 50, and 100 mg/kg/d), which was significantly different from that in the model group (P < .05, Table 1), there were no significant changes in the coefficients of all organs in the VA groups compared with those in the MCT group.
Effects of Vanillic Acid on Dirty Body Ratio of PAH Rats
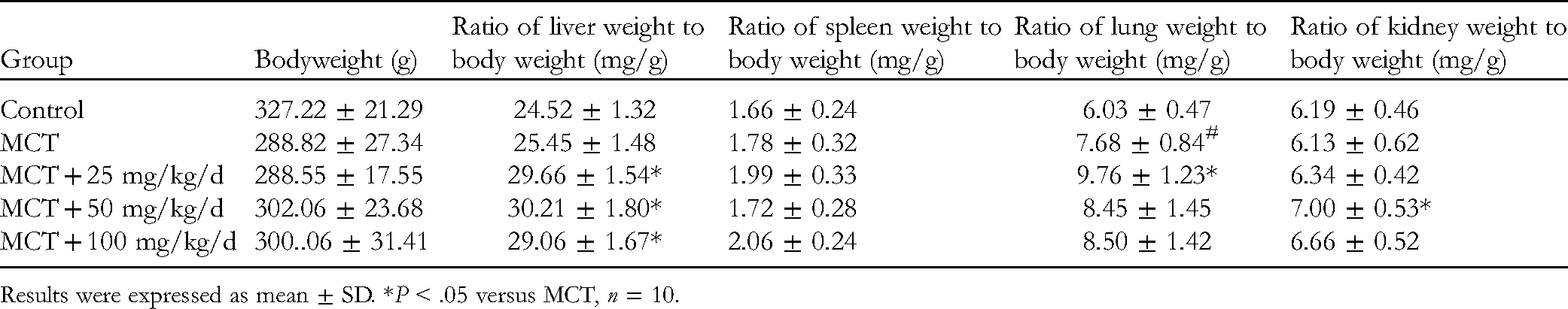
Results were expressed as mean ± SD. *P < .05 versus MCT, n = 10.
The values of HGB, HCT, RBC, and PLT were increased and WBC was decreased in the MCT model group compared with the control group. After the administration of VA, HGB, HCT, and RBC values decreased and WBC values increased compared with the model group (P < .05, Table 2). However, VA intervention did not affect the PLT values of PAH rats.
Effects of Vanillic Acid on Hematology-Related Indicators in PAH Rats

Abbreviations: HGB, hemoglobin; HCT, hematocrit; RBC, red blood cell; WBC, white blood cell; PLT, platelet.
Results were expressed as Mean ± SD *P < .05 versus MCT.
Effect of VA on Pulmonary Artery Morphology in PAH Rats
Based on hematoxylin-eosin (HE) staining of rat lungs, the WA%, WT%, and LA% for vascular diameters less than 100 μm were calculated. The results showed that the pulmonary artery vessel wall was thickened and the lumen diameter was reduced in the MCT group (Figure 2), and its WT% and WA% were significantly higher than those of the control group (P < .05), with values of 37.27 ± 0.03% and 60.06 ± 0.04%, respectively. The LA% in the MCT group was 39.94 ± 0.04%, which was significantly lower than that in the control group (P < .05). After VA intervention (25, 50, and 100 mg/kg/d), WA%, WT%, and LA% were remarkably improved compared to those of the MCT group (P < .05). VA significantly inhibited pulmonary artery vascular remodeling in MCT-induced PAH rats.

Effect of vanillic acid on pulmonary vascular remodeling in PAH rats. The vascular morphology of pulmonary arteries (30-100 µm) in HE-stained samples of lung tissue from five groups of rats (400 × ) (*P < .05 vs MCT, n = 5).
The degree of fibrosis in the lung tissues was assessed using Masson staining. As shown in Figure 3, there was significant fibrosis in the lung tissue of the MCT group, and the level of fibrosis decreased after VA intervention. After quantification by ImageJ 6.0 software (Figure 3), the results showed that the fibrosis level was elevated in the MCT group compared to the control group (P < .05). VA (25, 50, and 100 mg/kg/d) improved the fibrosis level in the lung tissue compared to the MCT group (P < .05).

Effect of vanillic acid on fibrotic lung tissue in PAH rats (Masson Staining, 200 × ) (*P < .05 vs MCT, n = 5).
Effect of VA on NO Levels in the Serum of PAH Rats
The NO levels in rat serum were measured by the nitric oxide (NO) assay kit, and the standard curve equation (Y = 0.007X + 0.0421 [R2 = 0.9998]) was obtained using the concentration of sodium nitrate as the horizontal coordinate and the absorbance at 540 nm after color development as the vertical coordinate. As the results show (Figure 4), the serum NO level was significantly decreased in the MCT group compared to the control group (P < .05). VA (25, 50, and 100 mg/kg/d) increased the serum NO levels compared to the MCT group (P < .05).
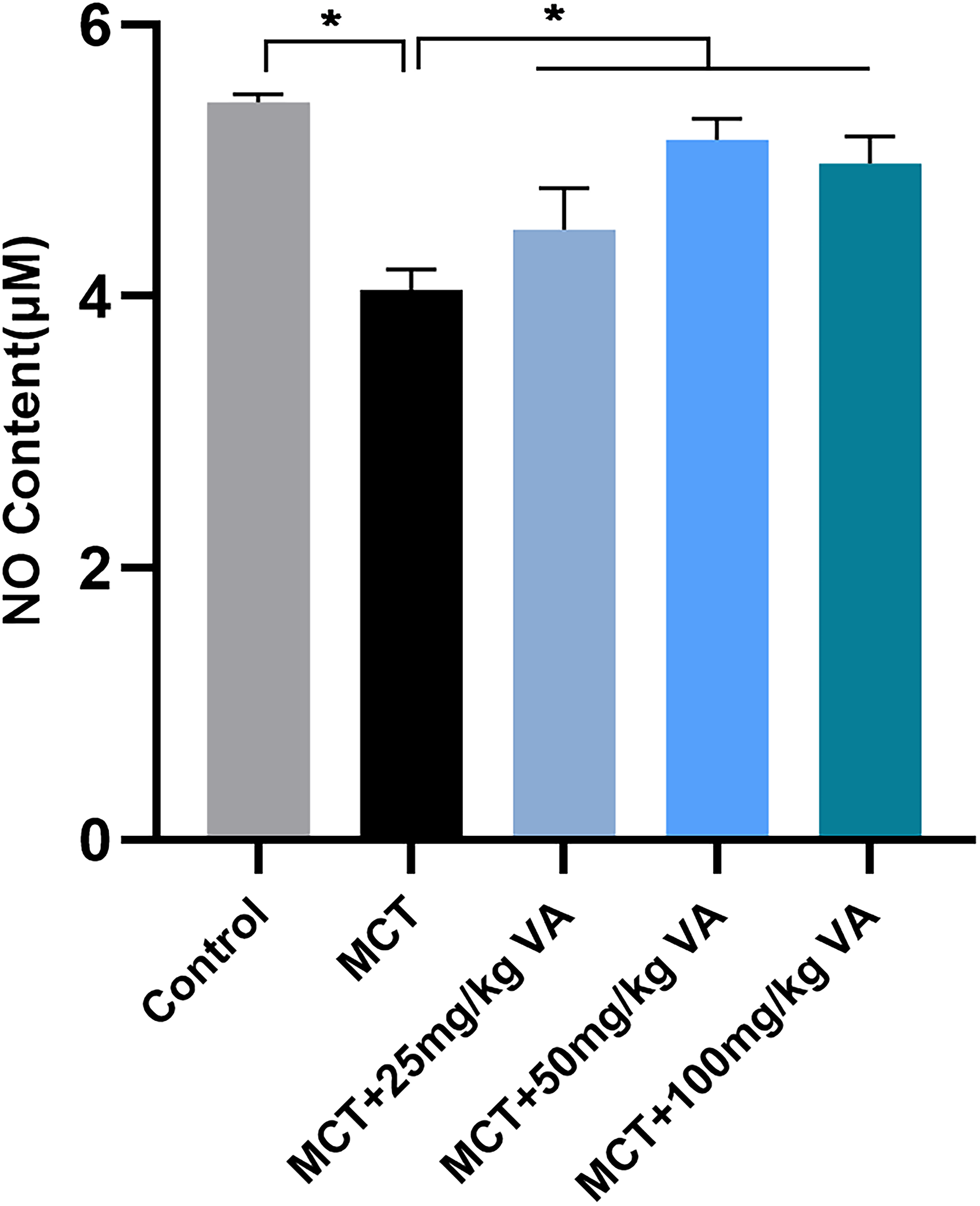
Effect of vanillic acid on NO in the serum of PAH rats (*P < .05 vs MCT, n = 5).
Effect of VA on the NO Pathway of Pulmonary Vascular Remodeling in PAH Rats
Compared with the control group, MCT upregulated the mRNA expression of Arg2, Hif-2α, and Hif-1β (P < .05, Figure 5). After VA intervention, the mRNA expression of Arg2, Hif-2α, and Hif-1β was downregulated significantly (P < .05, Figure 5). Additionally, the mRNA expression of eNOS was decreased by MCT (P < .05 vs control group). VA (50 and 100 mg/kg/d) upregulated the mRNA expression of eNOS compared with that in the MCT group (P < .05, Figure 5).

Effects of vanillic acid on the mRNA levels of Arg2, eNOS, Hif-2α, and Hif-1β in the lung tissue of PAH rats (*P < .05 vs MCT, n = 5).
The results of the Western blot experiment showed that the protein expression of Arg2, Hif-2α, and Hif-1β was significantly increased in the MCT group (P < .05 vs control group). After VA intervention, the protein expression of Arg2, Hif-2α, and Hif-1β was effectively inhibited compared with that in the MCT group (P < .05, Figure 6).

Effects of vanillic acid on the protein levels of Arg2, Hif-2α, and Hif-1β in the lung tissue of PAH rats (*P < .05 vs MCT group, n = 5).
In addition, the protein levels of P-eNOS and eNOS and the ratio of P-eNOS/eNOS were significantly reduced in the MCT group compared with the control group (P < .05). After VA intervention, P-eNOS/eNOS levels were significantly higher than those in the MCT model group (P < .05, Figure 7), which demonstrates that VA could promote the phosphorylation of eNOS.

Effects of vanillic acid on the protein levels of eNOS and P-eNOS in the lung tissue of PAH rats (*P < .05 vs MCT group, n = 5).
Changes in the Level of Arginase Activity in the Lung Tissue of PAH Rats
The arginase activity of the lung tissue was determined by the urea chromogenic method. The results showed (Figure 8) that arginase activity in lung tissue was significantly higher in the MCT group than in the control group (P < .05). In the VA intervention group (50 and 100 mg/kg/d), arginase activity in lung tissue was significantly inhibited (vs MCT group, P < .05).

Levels of arginase activity in lung tissue (*P < .05 vs MCT, n = 5).
The Constants Ki and Ki' and the Inhibition Mechanism of VA
We adapted a colorimetric method developed by Corraliza et al. to measure arginase activity. 21 The urea standard curve equation (Y = 0.718X + 0.0371 [R2 = 0.997]) was obtained with the concentration of urea solution as the horizontal coordinate and the absorbance at 550 nm as the vertical coordinate.
Based on the arginase inhibitory sigmoidal curves (Figure 9A), the IC50 of VA on arginase was determined to be 26.1 µM. To further determine the types of arginase inhibition by VA, enzyme kinetics studies were carried out. 22 The Lineweaver‒Burk curve (Figure 9B) was obtained using the inverse of the substrate focus as the horizontal coordinate and the reciprocal of the reaction rate as the vertical coordinate. The results showed that the maximum reaction rate Vm value decreased, and the Michaelis constant Km increased with the concentration of VA. Linear regression was performed with the concentration of VA as the horizontal coordinate and the slope of the different lines in the Lineweaver‒Burk plot as the vertical coordinate to obtain the inhibition constant Ki with 260.2 µM (Figure 9C). Linear regression was performed with the intercept of the Lineweaver‒Burk plot with the vertical axis as the vertical coordinate to obtain the inhibition constant Ki′ with 130.5 µM (Figure 9D). In conclusion, the inhibition constant Ki′/Ki = 0.5 indicates that VA has a mixed competitive inhibition effect on arginase.

Concentrations-response curves of vanillic acid on arginase. (A) Percentage of arginase inhibition at different concentrations of vanillic acid (IC50: 26.1 µM). Dixon (C) and Lineweaver‒Burk (B, D) plots allow the determination of Ki, Ki′, and the inhibition type (mixed competitive). Values are means ± SDs from 3 separate experiments.
Discussion
In the present study, we reported that VA could attenuate MCT-induced PAH and pulmonary vascular remodeling. In MCT-induced PAH rats, the RVSP, RV/LV + S, RVHI, and pulmonary artery wall thickness were increased. The increase was inhibited by treatment with VA. Additionally, we demonstrated that VA could attenuate MCT-induced PAH by increasing NO synthesis by regulating the balance of Arg2 and eNOS.
PAH is a progressive cardiovascular disease with increased pulmonary vascular resistance and pulmonary arterial pressure due to lesions of the pulmonary arteries (mainly the small pulmonary arteries). The etiology of PAH is multifactorial, and the pathophysiological features include pulmonary vasoconstriction and remodeling of pulmonary arterial circulation. 23 Although the origin of PAH is complex, impairments in vasodilator (NO) pathways underlie the evolution of the disease. 24 Clinical studies have found that the levels of intrapulmonary nitrate and NO-related reaction products in the bronchoalveolar fluid are significantly lower in patients with pulmonary hypertension. Endogenous NO was negatively correlated with pulmonary arterial pressure in pulmonary hypertension, and successful treatment of pulmonary hypertension was associated with an increase in NO levels. 25 The NO pathway plays an important role in regulating vascular tension in the pulmonary circulatory system by interacting with other signaling pathways. NO is produced from L-arginine and oxygen in a reaction catalyzed by NO synthase (NOS). L-arginine, the substrate of eNOS, can also be hydrolyzed by arginase to ornithine and urea. 1 Therefore, upregulation or activation of arginase can impair NO generation by eNOS. Thus, regulating Arg expression and activity is a major way to regulate endogenous NO and improve PAH. 26 Xu et al. found that arginase II expression and activity are strongly increased in patients with PAH. 27 Kao et al. found that arginase II activity was higher in PAH patients than in healthy controls. 28 In interleukin-13-overexpressing transgenic mice, arginase II deficiency significantly reduced the medial wall thickness of the pulmonary vasculature. 29 Thus, arginase is a potential target for the treatment of PAH.
eNOS is an important enzyme isoform for maintaining intravascular homeostasis. Therefore, any alteration in the NO-mediated system may result in a variety of cardiovascular diseases. Cowburn et al. proposed a mechanism by which chronic hypoxia could enhance the stability of Hif-2α, which leads to increased arginase expression and dysregulation of normal vascular NO homeostasis. These data suggested that hypoxia-inducible factor plays an important role in the regulation of arginase expression. 30
VA is a well-known phenolic compound that is abundant in various nuts, fruits, and herbs.31,32 It is frequently used as a food additive with cardioprotective, 33 kidney-protective, 34 and antioxidant effects. 35 In this study, we investigated the effect of VA on improving vascular endothelial function in MCT-induced PAH rats. The results showed that VA significantly reduced the RVSP and right ventricular hypertrophy index. HE results showed that the MCT group had thickened pulmonary artery vessel walls, reduced lumen diameters, and disturbed alveolar structures. The VA group exhibited a reduction in the thickening of the pulmonary artery vessel wall with a decrease in WT% and WA% and an increase in LA% compared with the MCT group (P < .05). Masson staining results showed that there were more collagen fibers in the MCT group. The degree of fibrosis in the intrapulmonary vascular wall and alveolar wall in the VA intervention group was significantly alleviated compared with that in the MCT group. It is suggested that VA could reduce pulmonary artery vascular remodeling and fibrosis in PAH rats and further reduce pulmonary vascular resistance and right ventricular pressure, thus alleviating right ventricular remodeling. Regarding the mechanism, the results showed that VA could significantly inhibit the gene and protein expression levels of Arg2 by downregulating the gene and protein expression levels of Hif-2α and Hif-1β. VA was also found to upregulate the gene and protein expression levels of eNOS and increase the phosphorylation level of eNOS. Therefore, VA could improve NO synthesis by regulating the balance of Arg2 and eNOS and further protect pulmonary vascular endothelial function, thus inhibiting the remodeling and fibrotic effects of the pulmonary vasculature. In addition, arginase activity in the lung tissue was also inhibited by VA. The arginase inhibition activity of VA was evaluated in vitro with an IC50 of 26.1 μM. VA was determined to be a mixed competitive inhibitor of arginase. Therefore, VA inhibits not only the expression of arginase but also the activity of arginase. To the best of our knowledge, there are no studies on arginase inhibition by VA. It may be a potential new arginase inhibitor for the treatment of PAH.
In conclusion, the arginase inhibitor VA exerted a protective effect on MCT-induced PAH and pulmonary vascular remodeling by enhancing NO signaling pathways. The mechanism might be that VA could inhibit the expression of Arg2 by downregulating the expression of Hif-2α and Hif-1β and increasing the phosphorylation of eNOS. This finding provides scientific evidence for the new arginase inhibitor VA in protecting against PAH.
Materials and Methods
Drug and Reagents
Arginase (Lot# M20A11Y120133, Specification: BR, 100 u/mg), MCT (Lot# K20N11K131409) and VA were purchased from Shanghai Yuanye Biotechnology Co. All other chemicals used were of analytical grade.
Animals
This experimental protocol was approved by the Institutional Animal Care and Use Committee of Qinghai University. Fifty healthy specific pathogen-free male Sprague‒Dawley (SD) rats (170 ± 20 g) were supplied by the Animal Center of Xi'an Jiao Tong University (permit number SCXK [Shan] 2018-001, Xi'an) in China. The animals were kept at a survival temperature of 22 ± 2 °C and relative humidity of 45–55% throughout the experiment and were allowed to feed ad libitum with standard pelleted feed and water. The experimental rats were randomly divided into five groups: control, model (a single subcutaneous injection of 40 mg/kg MCT), and model + VA (25, 50, and 100 mg/kg/d, continuous gavage for 4 weeks). No rat deaths were recorded during the entire experiment.
Hemodynamic and Hematological Index Measurement
The MCT model was established for 28 days. Then, the rats were weighed and anesthetized using 20% urethane (8 mL/kg) by intraperitoneal injection. The RVSP of each group of rats was measured using a Power Lab physiological recorder and biological acquisition system by the intravenous catheter method. The right ventricle (RV), left ventricle (LV), and septum (S) of the rats were separated according to the septal margins and weighed to calculate the right heart index RV/(LV + S).
After recording the hemodynamic parameters, blood was taken from the abdominal aorta and stored in normal serum tubes and EDTA anticoagulation tubes in a refrigerator at 4 °C. The blood in the normal serum tubes was separated on the same day: centrifuged at 2500 r/min for 20 min, and the supernatant was collected in 1.5 mL EP tubes and stored at −80 °C. The blood samples in the EDTA anticoagulation tubes were measured using the Mindray BC-5000 Vet Hematology analyzer to obtain the blood cell parameters: hemoglobin (HGB), red blood cell pressure (HCT), red blood cell count (RBC), platelet count (PLT), and white blood cell count (WBC).
Morphometric Evaluation of Lung Tissues
After the determination of RVSP, the rats were sacrificed, and lung tissues were fixed in 4% paraformaldehyde solution for 48 hours. Tissues were dehydrated, and paraffin-embedded sections were obtained (RM2135 type paraffin microtome; Leica, Solms, Germany). Histomorphological changes were observed by HE staining. The lung apical tissues were stained with Masson trichrome to evaluate the degree of fibrosis (collagen fibers stained blue). The vessel wall cross-sectional area (WA%), vessel wall thickness (WT%), and lumen area percentage (LA%) were measured in pulmonary artery vessels with diameters less than 100 µm using Image-Pro Plus 6.0 software (Media Cybernetics, Rockville, Maryland, USA). Quantification of the percentage of pulmonary fibrosis was measured using ImageJ software.
NO Levels in Serum (NO Release)
The NO concentration was detected using a NO Assay Kit (Beyotime, China) based on Griess Reagent. A series of concentrations of standards (0, 1, 2, 5, 10, 20, 40, 60, and 100 µM) were obtained using IP cell lysate. The absorbance at 540 nm was measured using a microplate reader (Infinite M200, TECAN) after mixing Griess standard solution (or serum) and Griess Reagent I and II. The concentration of NO in the samples could be calculated from the NaNO2 standard curve.
Quantitative Real-Time PCR
Total RNA was extracted from frozen lung tissue using TRIzol reagent (269204, Ambion, USA). cDNA was synthesized from 2 µg of RNA in a 20 µL reaction system using the Tiangen First Strand cDNA Synthesis Kit (Tiangen Biotechnology Co., Ltd, Beijing, China) according to the manufacturer's instructions. Quantitative real-time PCR was performed within an ABI 7500 Real-Time PCR System (Bio-Rad, California, USA) using Tiangen SYBR Green Supermix (Tiangen Biotechnology Co., Ltd, Beijing, China), and β-actin was used as a loading control. The primer sequences used for real-time PCR are shown in Table 3. The relative expression of genes was calculated using the 2−ΔΔct method and normalized to β-actin.
Primers Used for Real-Time PCR Analysis

Abbreviations: eNOS, nitric oxide synthase 3; ARNT, aryl hydrocarbon receptor nuclear translocator.
Western Blotting
The protein expression levels of eNOS, Arg2, Hif-1β, Hif-2α, and P-eNOS in lung tissues were determined by Western blotting. Lung tissues were homogenized rapidly in a lysis solution containing protease inhibitors. The homogenate was centrifuged at 12 000 r/min for 15 min at 4 °C. The protein concentration in the supernatant was determined using a BCA protein assay kit provided by Beyotime Institute of Biotechnology, Shanghai, China. The proteins (30 µg/lane) were separated by sodium dodecyl sulfate‒polyacrylamide gel electrophoresis (SDS‒PAGE) and transferred to polyvinylidene difluoride membranes. Membranes were blocked and immunoblotted with primary antibodies specific for β-actin [66009-1-Ig, Proteintech], eNOS (A15075, ABclonal), P-eNOS (9570S, Cell Signaling Technology), Arg2 (A6355, ABclonal), Hif-1β (SC-55526, Santa Cruz Biotechnology, INC.), and Hif-2α (NB100-122, Novusbio) overnight at 4 °C. The membranes were then incubated with goat anti-mouse IgG (AS003, ABclonal) and goat anti-rabbit IgG (AS014, ABclonal) secondary antibodies at a dilution of 1:10000 for 60 min at room temperature, followed by observation with an enhanced chemiluminescence kit (16H23C97, Boster Biological Technology Co., Ltd). β-Actin was used as an internal reference. The immunocomplexes were visualized using an Amersham Imager 600 (Cytiva, USA).
Levels of Arginase Activity in Lung Tissue
We adapted the colorimetric method to measure arginase activity developed by Corraliza et al. 21 Urea solutions of 0.1, 0.2, 0.4, 0.8, and 1.2 mg/mL series of concentrations were prepared with 50 mM Tris·HCl; 50 µL of different concentrations of urea solutions were taken and added to 43 µL of 50 mM Tris·HCl, 75 µL of 10 mM MnCl2 (prepared with 50 mM Tris·HCl), 50 µL of arginase substrate (0.5 M L-arginine, pH 9.7), 200 µL of acid solution (H2SO4: H3PO4: H2O, 1:3:7), and 25 µL of 9% α-isonitrosopropiophenone (prepared with anhydrous ethanol). The samples were heated at 100 °C for 45 min and then left at room temperature for 10 min, protected from light. The absorbance at 550 nm was measured by a fully automated Microplate Reader (Infinite M200, TECAN). The urea standard curve was obtained using the concentration of urea solution as the horizontal coordinate and the absorbance value as the vertical coordinate.
Lung tissues were homogenized rapidly in a lysis solution containing protease inhibitors. The supernatant of the homogenate obtained by centrifugation at 12 000 × g for 15 min at 4 °C was added to 75 µL Tris·HCl (50 mmol/L, pH 7.5) containing 10 mmol/L MnCl2. After heating the mix to 55 °C for 10 min, 50 µL of arginase substrate (L-arginine 0.5 mol/L, pH 9.7) was added and incubated at 37 °C for 1 hour. The reaction was terminated by adding 200 µL of acidic solution (H2SO4:H3PO4:H2O, 1:3:7). To determine the content of urea, α-isonitrosopropiophenone (25 µL, 9% concentration in ethanol) was added to the mixture and heated to 100 °C for 45 min. Finally, the samples were left at room temperature in the dark for 10 min, and the absorbance values at 550 nm were measured by a microplate reader (Infinite M200, TECAN).
Determination of the Constants Ki and Ki' and the Mechanism of Inhibition
The arginase solution was obtained by taking arginase and preparing a solution of 2 mg/mL with 50 mM Tris·HCl and adjusting the pH to 9.5. The inhibition rate of arginase by different concentrations of VA was calculated according to the above urea standard curve (1 unit of enzyme activity was defined as the conversion of 1 µmol of the substrate in each minute as the amount of urea), and the quantitative efficacy curve was plotted, and the IC50 was obtained.
For the evaluation of enzyme kinetics, three L-arginine substrate concentrations (25, 50, and 100 mM) and four VA concentrations (25, 50, 100, and 200 µM) were set up for the assay of arginase inhibitory activity, as described above in the levels of arginase activity in lung tissue. The equilibrium-state model (Michaelis‒Menten model) was fitted with SPASS 27.0 software, and Lineweaver‒Burk plots were plotted. The slopes of the different lines in the Lineweaver‒Burk plot were linearly regressed against the concentration of VA to obtain its inhibition constant Ki with arginase, and the intercepts of the different lines in the Lineweaver‒Burk plot against the vertical axis were linearly regressed against the concentration of VA to obtain the inhibition constants Ki′ with the “arginase-arginine.”
Statistical Analysis
Data, analyzed using SPSS 27.0 software (SPSS, Inc., Chicago, IL, USA), are presented as the mean ± standard deviation. Comparisons of means between groups were determined by one-way analysis of variance. Significance was defined as P < .05.
Footnotes
Acknowledgments
This work was supported by the National Natural Science Foundation of China (81860768 and 32060088), Applied Basic Research Project of Qinghai province, China (2020-ZJ-784), West Light Foundation of The Chinese Academy of Sciences, and Qinghai Province “Kunlun Talents, High-end Innovative and Entrepreneurial Talents” Project.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
