Abstract
Keywords
Introduction
Macrophages are immune cells dispersed throughout the body tissues, where they ingest and process foreign materials, dead cells, and debris and recruit additional macrophages in response to inflammatory signals. 1 The proinflammatory cytokine tumor necrosis factor-α (TNF-α) and nitric oxide (NO) synthesized by inducible NO synthase (iNOS) are the major macrophage-derived inflammatory mediators. 2 TNF-α is primarily produced by monocytes, macrophages, and T cells and has various proinflammatory effects on many cell types. 3 The small amounts of NO produced by constitutive NOS TNF-α are important for regulating physical homeostasis, whereas the larger amounts of NO produced by iNOS have been correlated with the pathophysiology in inflammation. 4
Although it can help combat infection, in a similar way to TNF-α, when NO is at high concentrations, it can have certain disadvantageous effects. Potent inflammatory cytokines may lead to edema, cellular metabolic stress, and tissue necrosis. 5 Thus, the inhibition of the excessive production of TNF-α and/or NO can be employed as a criterion to evaluate the anti-inflammatory effects of drugs. 6 Studies have described that many chronic diseases such as asthma, atherosclerosis, obesity, metabolic disease, 7 and rheumatoid arthritis are related to inflammation. 8 So a compound capable of modulating macrophage activation and/or function holds great promise for use in the treatment of diseases. Furthermore, both in vitro9, 10 and in vivo11,12 studies have demonstrated that some saponins isolated from Panax notoginseng have anti-inflammatory properties and these could be potential sources of therapeutic agents against inflammation.
Saponins are the most abundant organic compounds in P notoginseng and are the main active ingredients in isolated extracts. Over 70 saponins have been isolated from this plant and all of these were found to be dammarane-type triterpenoids saponins (Figure 1). Even though they have similar skeletons, the observed biological activities with respect to inhibition of NO and TNF-α level are varied and this is most likely due to the side chains attached to the core structure.13,14 Surprisingly, there are very few studies that dealt with the inhibition of NO and/or TNF-α induced by macrophages RAW264.7 by different saponins.

The basic structural formula of dammarane triterpenoids-type saponins. For the configurations of R1, R2, R3, and R4, please refer to Table 1.
Inhibitory Effects of the Tested Compounds on NO and TNF-α Production in LPS-Induced RAW264.7 Cells (n = 3). For the Structural Positions of R1, R2, R3, and R4, Please Refer to Figure 1.
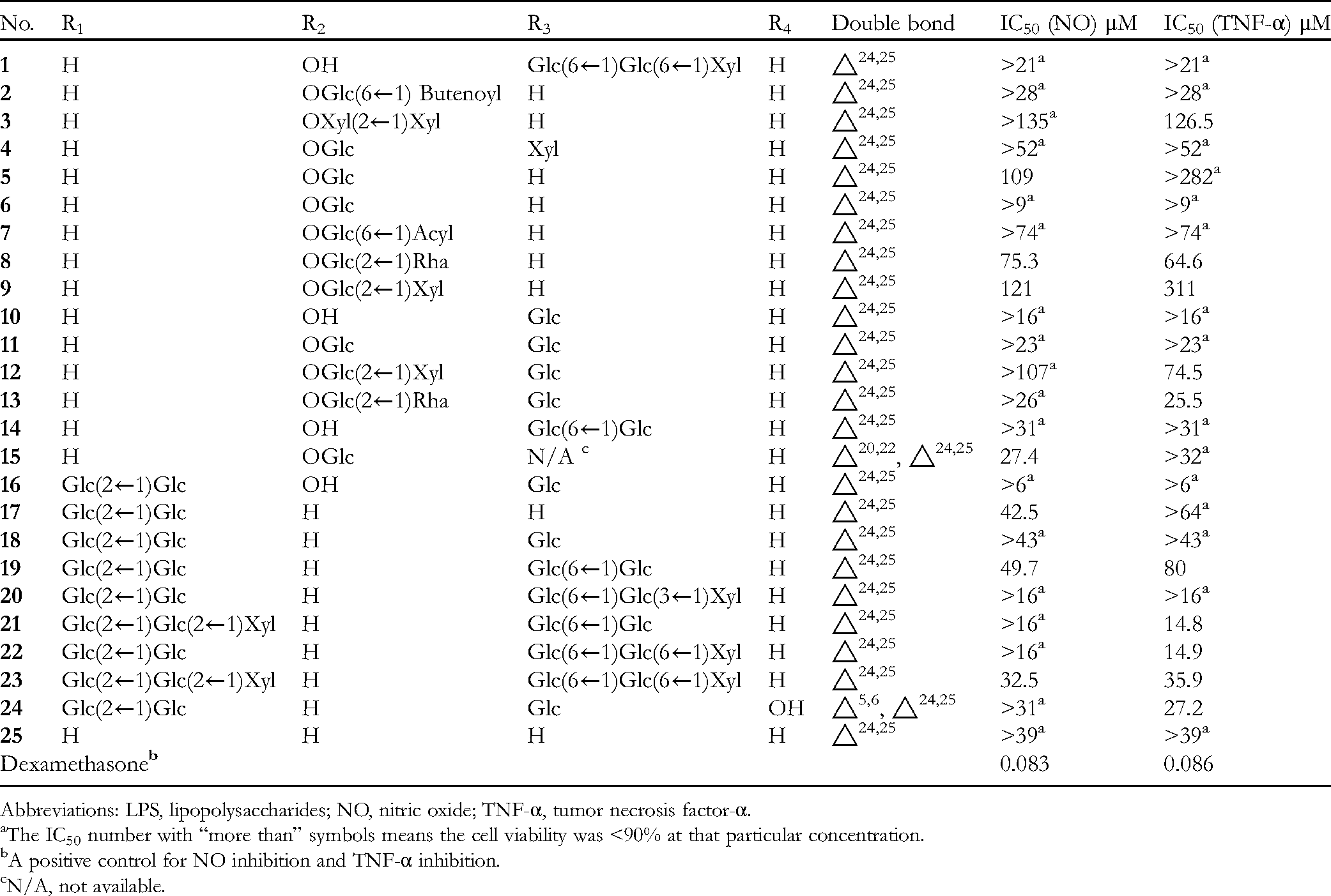
Abbreviations: LPS, lipopolysaccharides; NO, nitric oxide; TNF-α, tumor necrosis factor-α.
The IC50 number with “more than” symbols means the cell viability was <90% at that particular concentration.
A positive control for NO inhibition and TNF-α inhibition.
N/A, not available.
Therefore, in this study, the effects of different saponins on inhibition of NO and TNF-α were examined to understand the structure–activity relationships of their anti-inflammatory properties. The purpose of this study was also to evaluate the anti-inflammatory activities of 25 different saponins and provide an insight into their mechanism of action.
Materials and Methods
Test Compounds
Phytochemical studies of the 80% ethanol extract from the air-dried roots of P notoginseng were used to isolate 24 dammarane-type triterpenoids as described in our previous reports,
15
including 16 protopanaxatriol-type saponins (compounds
Reagents
High glucose Dulbecco's Modified Eagle Medium (DMEM) and fetal bovine serum (FBS) were purchased from Thermo Fisher Scientific (China) Co. Ltd. The mixture of penicillin and streptomycin (100×), lipopolysaccharides (LPS), dimethyl sulfoxide (DMSO), and 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyltetrazolium bromide (MTT) were purchased from Solarbio Technology (China) Co. Ltd. Phosphate buffer saline was purchased from Maixin-Biotechnology Co. Ltd. NO detection kits were purchased from Beyotime Institute of Biotechnology and ELISA for TNF-α assays was purchased from R&D Systems.
Cell Culture
RAW264.7, a mouse monocyte-macrophage cell line, was purchased from the Cell Bank of WuHan Boeter Bio-engineering Co. Ltd and suspended in high glucose DMEM supplemented with 10% heat-inactivated FBS, penicillin (100 U/mL), and streptomycin (100 μg/mL). Cultures were maintained at 37°C in a humidified incubator containing 5% CO2. Cells were stimulated by LPS (0.1 μg/mL) in the presence or absence of test compounds for the measurement of secreted TNF-α and proinflammatory cytokines as well as NO production.
Cytotoxicity Assay
Cytotoxicity was measured by using the MTT assay. A specific concentration of 1 mitogen was selected for subsequent assays. RAW 264.7 cells (1 × 104 cells/well) were seeded in 96-well plates. After 24 h, cells were treated with various concentrations of the test compounds (1-325 μM) and LPS (0.1 μg/mL) for a further 24 h. An aliquot of 5 mg/mL MTT solution was added to each cell supernatant and incubated for 4 h at 37°C in the dark. The culture medium was discarded and then 100 μL of DMSO was added into the wells followed by 5 min of horizontal shaking to solubilize the formazan. The optical density was read at 490 nm with a microplate reader.
NO Assay
Accumulated nitrite (NO2−) in culture media is a quantitative indicator of NO production, and this metabolite was determined using the Griess method. RAW264.7 cells were plated at a density of 1 × 105/well in 96-well plates and incubated for 24 h. A 0.1 μg/mL of LPS was then added and incubated for a further 2 h and then the cells were cotreated with the test compounds at a series of concentrations (based on viability ≥90% in unstimulated cells) for another 24 h. Dexamethasone was used as the positive control. The culture supernatants were mixed with an equivalent volume of Griess reagent (1% sulfanilamide and 0.1% N-[1-naphthyl]-ethylenediamine dihydrochloride in 5% phosphoric acid) and the absorbance was read at 490 nm. The concentration of NO2− was calculated by using standards. The inhibition rate of NO production induced by LPS was calculated using the NO2− levels.
TNF-α Assay
Cells were plated, cultured, and treated as described above. After all the treatments, the TNF-α levels in the medium were determined with a commercially available ELISA kit according to the manufacturer's instructions.
Results and Discussion
The inhibitory effects of compounds
Table 1 shows the IC50 values for the inhibitory effects of the test compounds on the NO production from LPS-induced RAW264.7 macrophages. As shown in Table 1, dexamethasone was used as a positive control. Compounds
A sugar moiety in C-3 was found to be important for inhibitory activity in LPS-induced macrophages (eg,
The effects of the isolated compounds on the inhibition of TNF-α production by LPS-induced macrophages were also examined. Table 1 shows that compounds
The protopanaxadiol-type glycosides
These results suggest that further investigations of the anti-inflammatory properties of ginsenoside saponins are warranted. These results also highlight the importance of how small structural differences between 2 compounds can lead to significant differences in biological activities, and that this principle should be taken into account when potential new drugs are being designed for clinical use.
Footnotes
Acknowledgments
The authors would also like to thank Dr Dev Sooranna, Imperial College London, for editing the manuscript.
Ethical Approval
Ethical approval is not applicable for this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by The National Natural Science Foundation of China (No. 21662005) and NMPA Key Laboratory for Quality Monitoring and Evaluation of Traditional Chinese Medicine, Chinese Materia Medica (KFKT2022-6).
