Abstract
The free sterol fraction from the deep-sea holothurian Orphnurgus cf glaber Walsh, 1891, has been isolated and studied by GC–MS. Sterols containing Δ7, Δ0, Δ5, polyunsaturated and 5,8-epidioxy modified cores were found. A high percentage of compounds with polyunsaturated cores distinguish this composition from those of previously studied holothurians. Almost all identified compounds had saturated side chains, which is also a specific feature of this fraction. Peculiarities of biosynthesis and metabolism of the found sterols are discussed.
Introduction
Seamounts, including the Hawaiian-Emperor Chain, are areas with high biological productivity of benthic and pelagic communities, including important biological resources that support a commercial fishery. The Emperor Chain, located in the North-Western Pacific area, could be affected by uncontrolled commercial fishing which seriously threatens the natural balance in the area. The North Pacific Fishery Commission (NPFC https://www.npfc.int) recognized at the meeting held in Yokohama (in 2018) the lack of information on the distribution and population density of bottom organisms in the region and the urgent need to study the bottom communities, classified as vulnerable marine ecosystems due to their slow recovery after overfishing.1,2 Data on holothurian diversity, as well as natural compounds from any echinoderms from the Chain, are absent in the literature (Table 1).
Free Sterols From Orphnurgus cf glaber Analyzed as Acetate Derivatives.

* The retention times reported for the corresponding sterol acetates under the conditions specified in 2.1. Analytical methods.
** Abundances are given as % of total sterol fraction.
Sea cucumbers, marine invertebrates belonging to the class Holothurioidea (the phylum Echinodermata), are one of the numerous and very ancient forms of marine life playing important biological and biogeochemical roles in the transformation of marine detritus. In deep-sea ecosystems, their role is especially great. However, deep-sea holothurians are poorly studied chemically. Sterols are key biochemical components, being functionally involved in the construction of membranes, as well as in hormonal regulation. There are only a few studies (see Table S1, Supplemental Materials) on sterols from holothurians that are not available for collection by diving. Ponomarenko et al 3 and Stonik et al 4 investigated epipelagic holothurians (from 50 to 200 m); Stonik et al, 4 Dmitrenok et al, 5 and Ballantine et al 6 investigated mesopelagic holothurians (from 200 to 1000 m); Ballantine et al 6 investigated bathyal holothurians (depths of 1000 to 4000 m); Amaro et al, 7 Drazen et al, 8 Neto et al, 9 and Ginger et al10,11 investigated abyssal holothurians (depth of 4000 to 6000 m). Holothurians of the Porcupine Abyssal Plain were investigated most thoroughly.9–11 Studies in other locations are sporadic and deep-sea species of holothurians inhabiting under-water mountains in the western part of the Pacific Ocean remained unstudied.
The sea cucumber Orphnurgus cf glaber Walsh, 1891, is a rare and scattered deep-sea holothurian. In the present research, free sterols from this holothurian have been investigated and compounds

Structures of the sterols from Orphnurgus cf glaber.
Results and Discussion
Only a single specimen of the deep-water holothurian Orphnurgus cf glaber Walsh, 1891, was collected from a depth of 2231 m. The sterol fraction was isolated from an ethanol extract by chromatography on silica gel and was separated into three subfractions. The 1H NMR spectrum of subfraction 1 showed signals of angular methyl groups, СH3-18 at 0.68 and CH3-19 at 1.01 ppm, which are characteristic of 5(6)-unsaturated sterols.
12
Stanols (СH3-18 at δH 0.65 and CH3-19 at δH 0.80) and Δ7-sterols (СH3-18 at δH 0.54 and CH3-19 at δH 0.81) dominated in the subfractions 2 and 3, respectively. The subfractions were acetylated and analyzed by GC-MS. The structures of individual components were established based on the analysis of the mass spectra of their acetate derivatives, the elution order under GC conditions and comparison of these with the literature data.3,4,13,14 The identification of cholest-5-en-3β-ol (
5(6)-Unsaturated C29 sterol
A number of polyunsaturated sterols were present along with common Δ7-sterols in subfraction 3. Thus, compound
Compound
Compound
Similarly to the above, compound
Compounds
A similar pattern is observed in the mass spectrum of C29-oxysterol
The unidentified substances C273F and C293F were also a pair of homologs with a high degree of unsaturation. Compound C273F has an RT equal to that of cholesterol (25.8 min) and a mass spectrum characterizing a tri-unsaturated C27 sterol (m/z 424 M+, 364 (M – AcOH)+, 349 (M – AcOH – CH3)+). Compound C293F has an RT slightly less than that of 24-ethylcholest-5-en-3β-ol (RT 30.1 min) and a mass spectrum characterizing a tri-unsaturated C29 sterol (m/z 452 M+, 392 (M – AcOH)+, and 377 (M – AcOH – CH3)+). The ratio of the abovementioned peaks was almost the same for these two compounds. The ratio of the fragment peaks at m/z 311, 257, 251, 235, 223, 209, 197, and 195 was also very similar. Unfortunately, it was not possible for us to determine the exact structures of these C273F and C293F sterols from the available data.
Thus, the free sterol composition of the deep-sea holothurian Orphnurgus cf glaber includes mainly cholesterol (
On the contrary, the ability to transform the sterol nucleus, converting dietary Δ5-sterols into their Δ7-analogs, is inherent for most sea cucumbers.18 Holothurians usually produce triterpene glycosides as chemical protection agents against predators, but their own cells resist the action of their cytotoxins due to the presence of Δ7-sterols in the membrane.19 For example, frondoside A caused significantly less permeabilization in liposomes containing Δ7 holothuroid sterols than those containing cholesterol.
20
Although no triterpene glycosides were found in the Orphnurgus cf glaber specimen studied by us using the standard methods,
21
its sterol composition demonstrates the intensive transformation of dietary Δ5-sterols. This mixture contains 14.4% Δ5-sterols, 12.6% stanols, 51.6% Δ7-sterols, and 20.6% polyunsaturated sterols and oxygen adducts. An intriguing feature of the sterol composition was the presence of compounds with a polyunsaturated sterol nucleus. Probably, processes of dehydrogenation take place in the body of the animal itself because, to the best of our knowledge, the corresponding sterols have not been found in algae and/or detritus until now. Ginger et al,
10
investigating deep-sea holothurians from the Porcupine Abyssal Plain, found a number of Δ5,7-sterols and an unidentified C27-diene. They assumed that “relatively higher amounts of unsaturated compounds in deep-sea animals are ascribed to an adaptation of the latter to maintain membrane fluidity at high pressure and low temperature.”
10
It seems to us that our finding of numerous di- and tri-unsaturated sterols confirms this suggestion. Orphnurgus cf glaber did not contain Δ5,7-sterols, which are intermediates in Δ5- to Δ7-double bond migration in shallow-water holothurians,
22
but it contained oxygen adducts
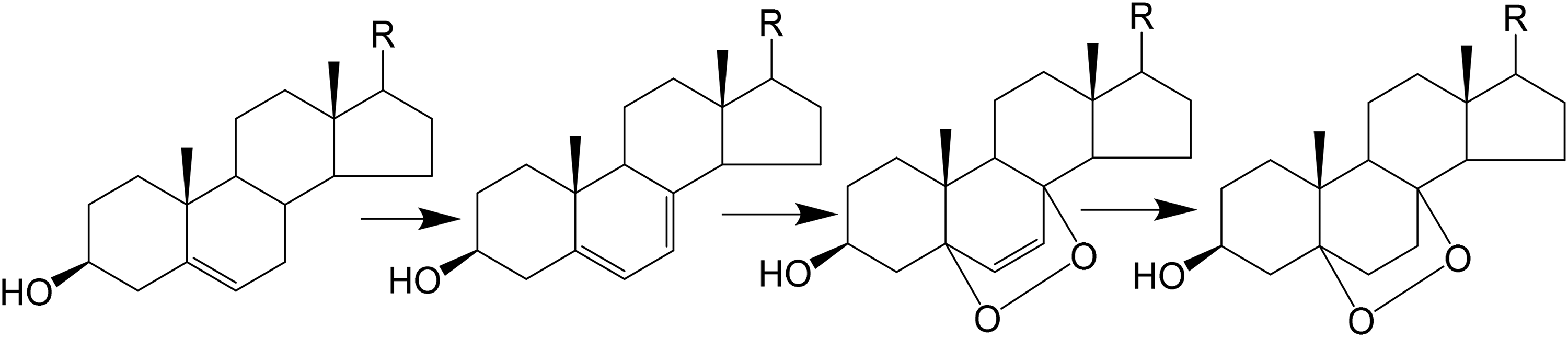
Hypothetical pathway for the formation of 5,8-epidioxysterols.
Conclusion
A specimen of the deep-sea holothurian Orphnurgus cf glaber collected at the Hawaiian-Emperor Chain was investigated for its free sterol composition. Twenty-one components containing Δ7, Δ0, Δ5, polyunsaturated or 5,8-epidioxy modified cores and predominantly saturated side chains were found. Like other holothurians of the family Deimatidae (Oneirophanta mutabilis8–10 and Deima validum7,10 see Supplemental Materials, Table S1), this sea cucumber contains dietary detritus sterols and products of their modification. The high percentage of sterols with saturated side chains and those with polyunsaturated nuclei distinguishes O. cf glaber from previously studied holothurians, both deep-sea and shelf ones. We assume that the detritus is redesigned by fungi or bacteria in either surface sediments or holothurian gut. According to preliminary research from mycological cultures of the intestinal contents of O. cf glaber, 3 species of micellar fungi were isolated (Dr Borzykh O.G., unpublished data). However, we do not exclude that some polyunsaturated sterols are intermediates in the conversion of Δ5- to Δ7-sterols, like Δ5,7-sterols found by Ginger et al 10 in other holothurians in small amounts.
Experimental
Analytical Methods
1H NMR spectra were recorded using a Bruker Avance III 700 spectrometer, at 700 MHz, δ in ppm referenced to CDCl3 at δH 7.26. GC-MS analyses were carried out on a Hewlett Packard HP 5973 MSD: HP 6890 chromatographic system, with a capillary column HP-5MS with a temperature regime of 100 °C followed by an increase at a rate of 10 °C/min and then in isothermal mode at 280 °C. Helium was used as the carrier gas (1 mL/min) and the ionizing voltage was 70 eV.
Animals
A single specimen (the length was about 40 cm) of the holothurian Orphnurgus cf glaber Walsh, 1891 (the Phylum Echinodermata, Class Holothurioidea, Subclass Actinopoda, Order Synallactida, Family Deimatidae) was collected during the expedition of the A.V. Zhirmunsky National Scientific Center of Marine Biology FEB RAS aboard the RV Akademik M. Lavrentyev (cruise no. 94) in July to September 2021. The collection was performed using a remote-operated vehicle Comanche 18 at the Koko guyot, Emperor Chain (35.789° N, 171.049° E) from a depth of 2231 m. The sample was frozen and stored (−20 °C) until moving to the laboratory.
Extraction and Isolation of Free Sterols
The specimen (148.0 g) was dissected and thrice extracted with ethanol. The obtained extract was concentrated in vacuo and was separated for the study of glycosides (0.9 g) and sterols (1.9 g). The search for glycosides was carried out according to the standard method. 21 The second part of the extract was purified by silica gel column chromatography in the system hexane/ethylacetate (12:1, vol/vol) to obtain the fraction of free sterols (2.2 mg). This fraction was separated by HPLC using an Altex UltrashereTM-Si column (hexane/ethylacetate 5:1, vol/vol) into three subfractions (0.4 mg, 0.3 mg, and 1.4 mg), which were analyzed by NMR spectroscopy; then they were acetylated by acetic anhydride in pyridine at room temperature (24 h) and analyzed by GC-MS.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221142791 - Supplemental material for Free Sterol Composition of Deep-Sea Holothurian Orphnurgus cf. glaber
Supplemental material, sj-docx-1-npx-10.1177_1934578X221142791 for Free Sterol Composition of Deep-Sea Holothurian Orphnurgus cf. glaber by Ludmila P Ponomarenko, Irina I Kapustina, Salim S Dautov and Tatiana N Dautova, Valentin A Stonik in Natural Product Communications
Footnotes
Acknowledgments
The study was carried out using the equipment of the Collective Facilities Center “The Far Eastern Center for Structural Molecular Research (NMR/MS) of PIBOC FEB RAS.”
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Science and Higher Education of the Russian Federation (Grant No. 13.1902.21.0012 (contract No. 075-15-2020-796)).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
