Abstract
Keywords
Introduction
The human catastrophe brought about by Coronavirus disease 2019 (COVID-19) has lasted for two full years. The emergence of Alpha, Delta and Omicron mutant strains has brought three peaks of the epidemic. Fortunately, vaccination can induce a long-lasting specific T-cell response and build an immune barrier, significantly reducing the severity and fatality rate of covid-19. 1 Therefore, preventing mild to severe disease has become our biggest challenge. A large number of studies have shown that the main reason for the change in covid-19 patients from mild to severe or even death is the outbreak of cytokine storm (CS).2,3 CS refers to an uncontrolled excessive inflammatory response, which starts locally and spreads further in the body through the systemic circulation, resulting in acute lung injury (ALI), and eventually progressing to acute respiratory distress syndrome (ARDS), multiple organs functional failure and even death, causing medical staff to lose the opportunity to rescue.4,5 Therefore, anti-inflammatory drugs such as dexamethasone(Dex) and Chinese medicine are new strategies to suppress excessive inflammation caused by inflammatory immune cells, especially suppressing conversion from mild to critical disease.6‐8 However, due to its unclear clinical efficacy and potential adverse reactions, it limits the wide range of clinical treatments. Tibetan medicine is the treasure of traditional Chinese medicine that may have a therapeutic effect on CS.
The traditional Tibetan medicine Rhodiola crenulata (RC; Tibetan name: Solomabao) has a good reputation as “plateau ginseng.” It boosts metabolism, enhances cell vitality, improves immune capacity, and is often used to treat infectious diseases such as tuberculosis, pneumonia, and others.9,10 RC inhibits the transcription of pro-inflammatory genes and the production of IL-6 and other pro-inflammatory factors and protects against damage caused by inflammatory reactions. 11 In vivo and in vitro studies of this research group have found that salidroside, an important component of RC, can reduce the secretion of inflammatory factors such as IL-6, ACE2, GM-CSF, and increase the content of ACE2 in various tissues, which proves that salidroside is effective Lung injury caused by CS has a significant protective effect and effectively fights inflammatory storm. 12 Nevertheless, the underlying mechanism of this activity remains unknown.
Network pharmacology permits a holistic and comprehensive understanding of a drug's potential mechanisms of action. Molecular docking predicts the geometric and energy-matching degree between receptors and ligands through the calculation to screen for active ingredients. 13 This procedure provides the possibility of using RC to prevent CS. In the present study, we analyzed the active components, targets, and signaling pathways involved in preventing and treating CS using network pharmacology and molecular docking.
Materials and Methods
Network Pharmacology Analysis
Collection of Active Ingredients and Targets of RC
The chemical composition of RC was drawn from the Organchem database (http://www.organchem.csdb.cn/), and the chemical composition was determined using PubChem database (https://pubchem.ncbi.nlm.nih.gov/). Swiss target prediction and Swiss ADME (http://www. swisstargetprediction.ch/) (Probability* > .8) online databases were used to screen compounds and obtain predictive targets. Finally, the UniProt (https://www.uniprot.org/) database 14 was used to standardize the target acquisition.
Drug-Disease Target Prediction
We searched for disease-related targets in Genecards (https://www. genecards.org/) and OMIM (https://www.omim.org/) databases using the keywords “cytokine storm” and “hypercytokinemia.” We used Venny 2.1 to determine intersections between diseases and RC ingredients and displayed them using Wayne diagrams.
Construction of a Component-Target Network
The selected active components of RC and disease targets were obtained using Cytoscape software (V3.7.2) (http://www.cytoscape.org/). The active components of RC that prevent CS were further analyzed.
Construction and Screening of PPI Networks
The drug-disease target in item “2.2” was analyzed using the STRING (http://string-db.org) database. 15 The species was set as “homosapiens,” the minimum interaction score value was set as ≥ 0.7, and the nodes of no connection in the network were hidden. The network diagram and the output drug-disease target protein interaction results were obtained. The core targets are screened using Cytoscape software (V3.7.2), and the bar chart of the top 10 core targets was drawn.
GO Functional Enrichment Analysis and KEGG Pathway Enrichment Analysis
The selected core targets were used in DAVID (https://david.ncifcrf.gov/) online database and Rstudio for GO function enrichment and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment analyses. The results were sorted from small to large, and the results were analyzed (P < .05 represented a significant difference). 16
Molecular Docking
We downloaded the two-dimensional structure of the core small molecular compounds in 1.3 from the PubChem (https://pubchem.ncbi.nlm.nih.gov/) database and used Chemoffice software to convert them into three-dimensional structures, then saved in *mol format. We opened them with AutoDock Tools1.5.6, deleted water molecules, added atomic charges, assigned atomic types, defaulted all flexible keys rotatable, minimized the small-molecule ligand energy, and saved them as * PDBQT files. We downloaded the crystal structure of IL-6, SARS-CoV-2 3CL, STAT3, and ACE2 (PDB ID:1ALU, 6Lu7, 1BG1, and 1R42) from the PDB database (http://www.rcsb.org). PyMol and AutoDdock software were used to delete the irrelevant small molecules, dehydrating and deactivating ligands, and hydrogenation in the protein molecules of the target protein, and then saved these as *PDBQT files. Finally, Auto Ddock Vina was used to conduct molecular docking between small molecule compounds and IL-6, SARS- CoV-2 3CL, STAT3, and ACE2, and the results were visualized using PyMOL and the Discovery Studio Visualizer.
Animal Experimental Validation
Based on the results of network pharmacology, molecular docking, and pre-experiments, we used an animal model to induce CS to determine whether kaempferol would inhibit inflammatory factors and mitigate tissue damage.
Chemicals and Reagents
Lipopolysaccharide (LPS, E. coli055: B5) was provided by Sigma Chemical Co. Kaempferol (Kae, K8220) was purchased from Solarbio. Dexamethason (Dex) was purchased from CISEN. Rat TNFα, IL-6, and IL-10ELISA kits were purchased from Thermo Fisher, USA.
Animals and Treatments
Specific pathogen-free male Sprague Dawley rats weighing 180 g to 200 g, aged 6 to 7 weeks, were purchased from Chengdu Dashuo Experimental Animal Co., Ltd, Sichuan Province. All animal experiments were complied with the ARRIVE guidelines and were carried out according to the National Institutes of Health guide for the care and use of laboratory animals (NIH Publications No. 8023, revised 1978).
Rats were randomly divided into six groups with six rats in each group: (1) control with PBS; (2) 5 mg/kg LPS; (3) 5 mg/kg dexamethasone (Dex) + 7 mg/kg LPS; (4) 25 mg/kg Kae + 7 mg/kg LPS; (5) 50 mg/kg Kae + 7 mg/kg LPS; and (6) 100 mg/kg Kae + 7 mg/kg LPS. Rats were fasted for 12 h. Kae was given by intraperitoneal (IP) injection, whereas IP gave dex as a positive control, control and LPS group were given an equal volume of distilled PBS. 0.5 h later, the rats in groups 2, 3, 4, 5, and 6 were IP challenged with 7 mg/kg LPS. Six hours after LPS treatment, rats were sacrificed for analysis.
Morphologic Analysis
Heart, liver, spleen, lung, and kidney tissues were removed from rats immediately after sacrifice, and morphologic analysis was performed by camera imaging.
Histological Analysis
Tissues were rapidly dissected and fixed in 4% paraformaldehyde (v/v) for 24 h before being embedded in paraffin. They were then sectioned and stained using standard histological methods. In addition, sections were stained with hematoxylin and eosin (H&E), and pathological changes were examined using light microscopy (Nikon). The histological characteristics of the organ's injury (including alveolar edema and hemorrhage, the number of infiltrating leukocytes, and the thickness of the alveolar wall and epithelium) were evaluated. Each histological characteristic was evaluated on a scale of 0–3 (0, normal; 1, mild; 2, moderate; and 3, severe).
Wet/Dry Ratio of the Lung
The inferior lobe of the right lung was separated, and the wet weight was determined. Subsequently, the lungs were incubated at 80 ◦C for 48 h to remove all moisture, then the dry lungs were weighed, and the lung wet-to-dry weight ratios (W/D) were calculated.
Cytokine Measurement
According to the manufacturers’ instructions, homogenized lung tissue and serum were collected, and concentrations of TNFα, IL-6, and IL-10 were measured using ELISA kits.
Statistical Methods
Data were expressed as means ± SD. One-way ANOVA was used for multiple comparisons. P < .05 was considered statistically significant. *, #P < .05, **, ## P < .01, ***, ### P < .001.
Results
Screening of Active Components and Targets of RC
Based on the research hypothesis, a technical roadmap was obtained (Figure 1). Using the Swiss ADME database, the collected chemical components were screened according to the gastrointestinal absorption = HIGH, drug-likeness ≥ 2, and the chemical components and corresponding targets meeting the screening conditions. A total of 16 chemical constituents (Table 1) and 414 drug targets were selected.
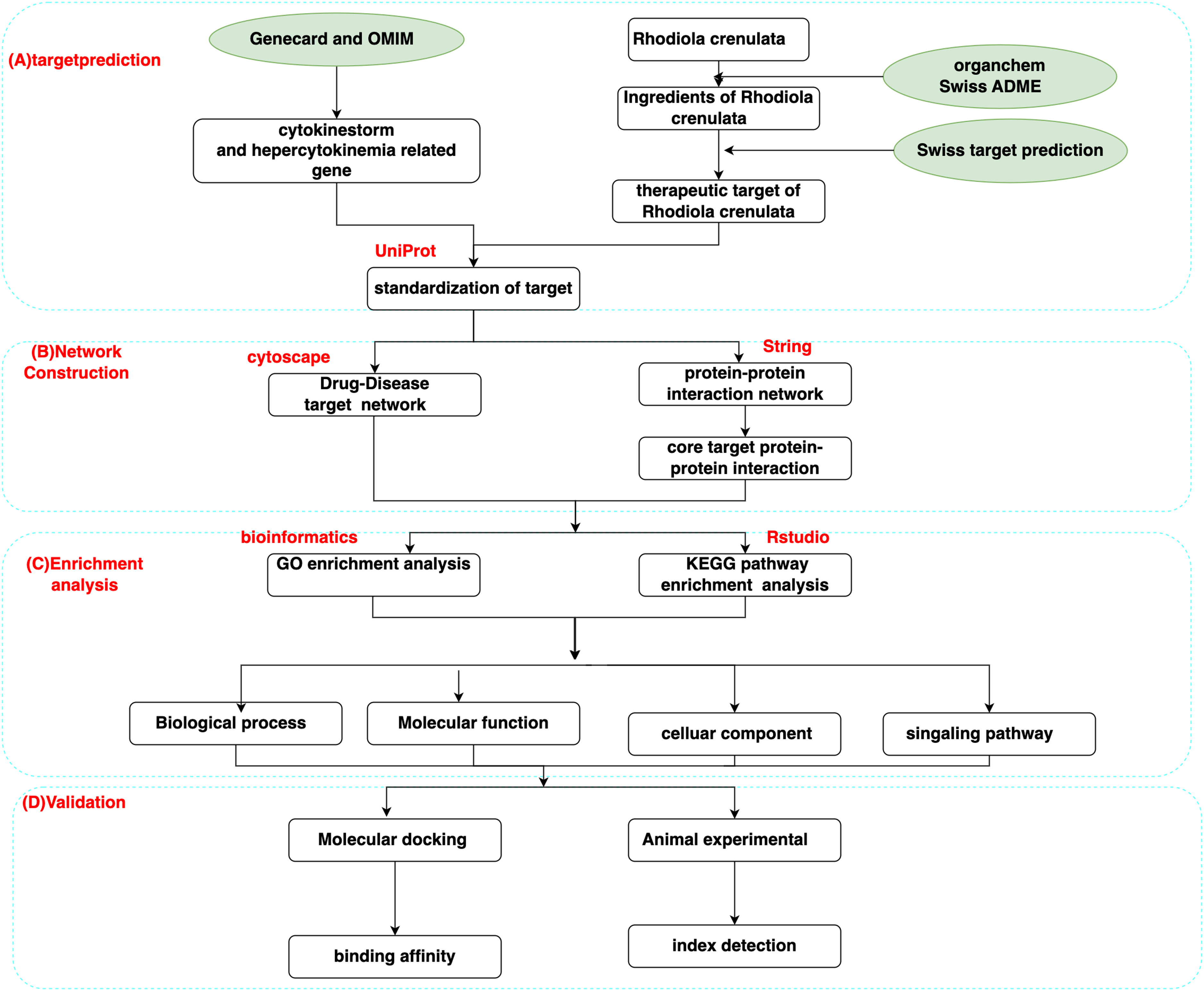
Workflow for this study.
Basis Information of Active Components in Rhodiola crenulata.

Intersection of Disease and Drug-Disease Targets
Genecards and OMIM databases were used to search for targets related to CS, and a total of 484 targets were obtained after excluding duplicates. Using Venny 2.1.0 (https://bioinfogp.cnb.csic.es/tools/venny/), the component targets and the disease targets are intersected to identify common targets (49 in total) that were targets of RC to prevent CS (Figure 2 and Table 2).
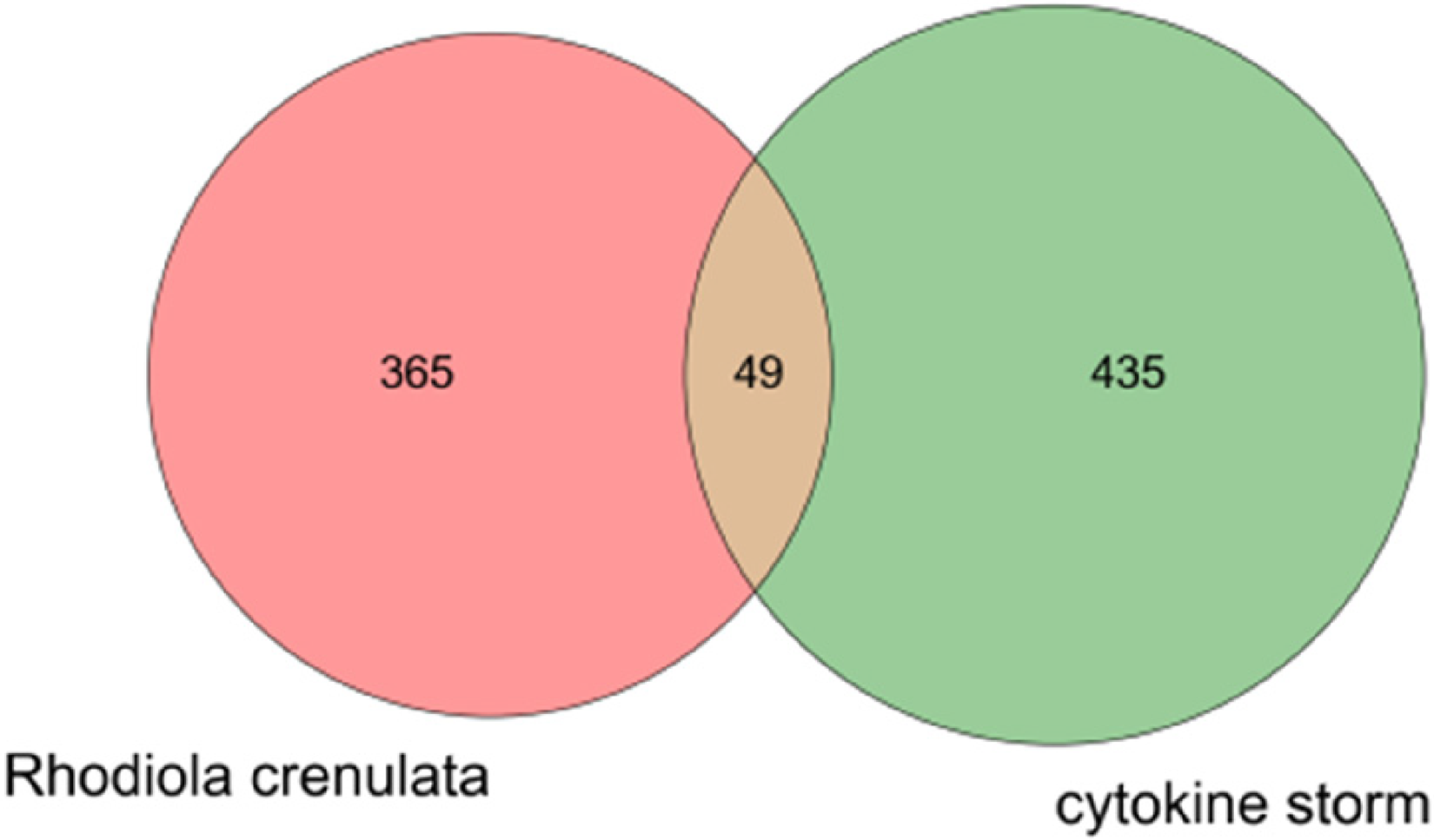
Venny diagram of Rhodiola crenulata therapeutic targets for cytokine storms.
Key Targets of Rhodiola crenulata for Controlling Cytokine Storms.
Component-Target Regulation Network map
We imported the active ingredients and targets of RC into Cytoscape 3.7.2 to construct a visual analysis network diagram. The octagons in the figure represent genes, and the circles represent chemical components. Figure size and color depth positively correlated with the degree, and the value was related to the size of the component in the disease. The greater the value, the greater the possibility of the component in the treatment of the disease. These findings suggest that kaempferol (DH16), linoleic acid (DH12), dibutyl phthalate (DH14), ethyl octanoate (DH1), and oleic acid (DH4) are the primary active components that control CS in RC (Figure 3).

Rhodiola crenulata ingredient-target network diagram. Sixteen RC ingredients (octagon, pink), four hundred and fourteen targets (circle, croci).
PPI Network Maps and Core Targets
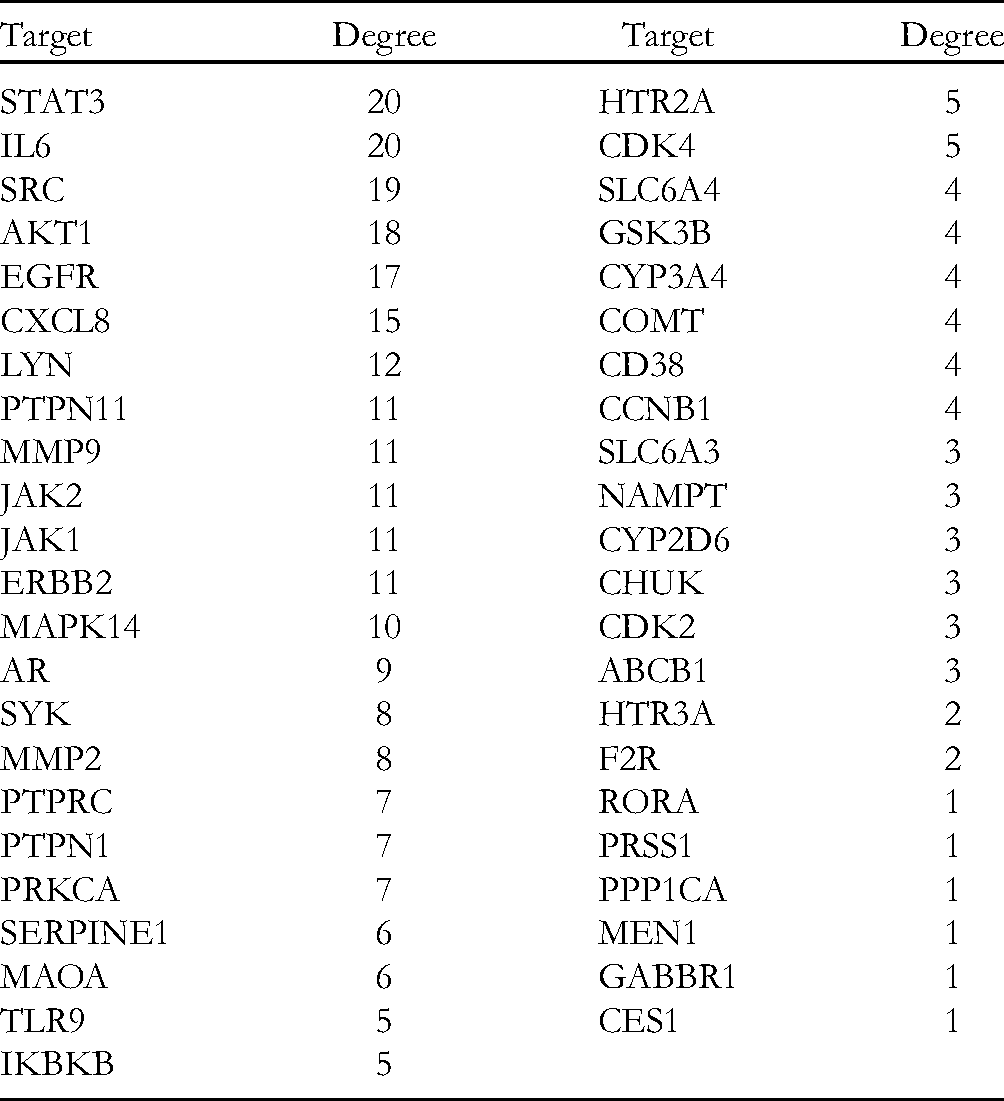
According to the conditions in section 2.1.4, the targets from the mapping of RC and CS were introduced into the STRING database, and the network diagram of the interaction between RC and CS was obtained. The higher the edge number, the larger the figure and the darker the color, the more likely the node becomes the core target (Figure 4). Cytoscape 3.7.2 software was used to identify the interaction network of CS targets of RC and draw a bar chart according to the number of adjacent nodes (Figure 5). The number of adjacent nodes is related to the probability of becoming the core gene; the higher the number, the greater the probability of becoming the core target. IL6, STAT3, SRC, AKT1, EGFR, and CXCL8 were closely related to CS.

PPI network diagram.

Core targets bar chart.
GO and KEGG Enrichment Analyses
The core targets in the PPI network maps and core targets section were used in the DAVID database and R software for GO functional and KEGG pathway enrichment analyses. GO functional enrichment analysis revealed that the biological process of CS was related to drug response, protein phosphorylation, and inflammatory reaction. In terms of cell composition, the results show that the efficacy of RC involves cell solute, cytoplasm, and nucleus. RC may affect protein kinase activity, ATP binding, and protein binding (Figure 6). KEGG pathway enrichment analysis revealed that CS-related factors were associated with IL6-JAK2-ST AT3 signaling pathway and PI3K-Akt signaling pathway (Figures 7,8, and 9).
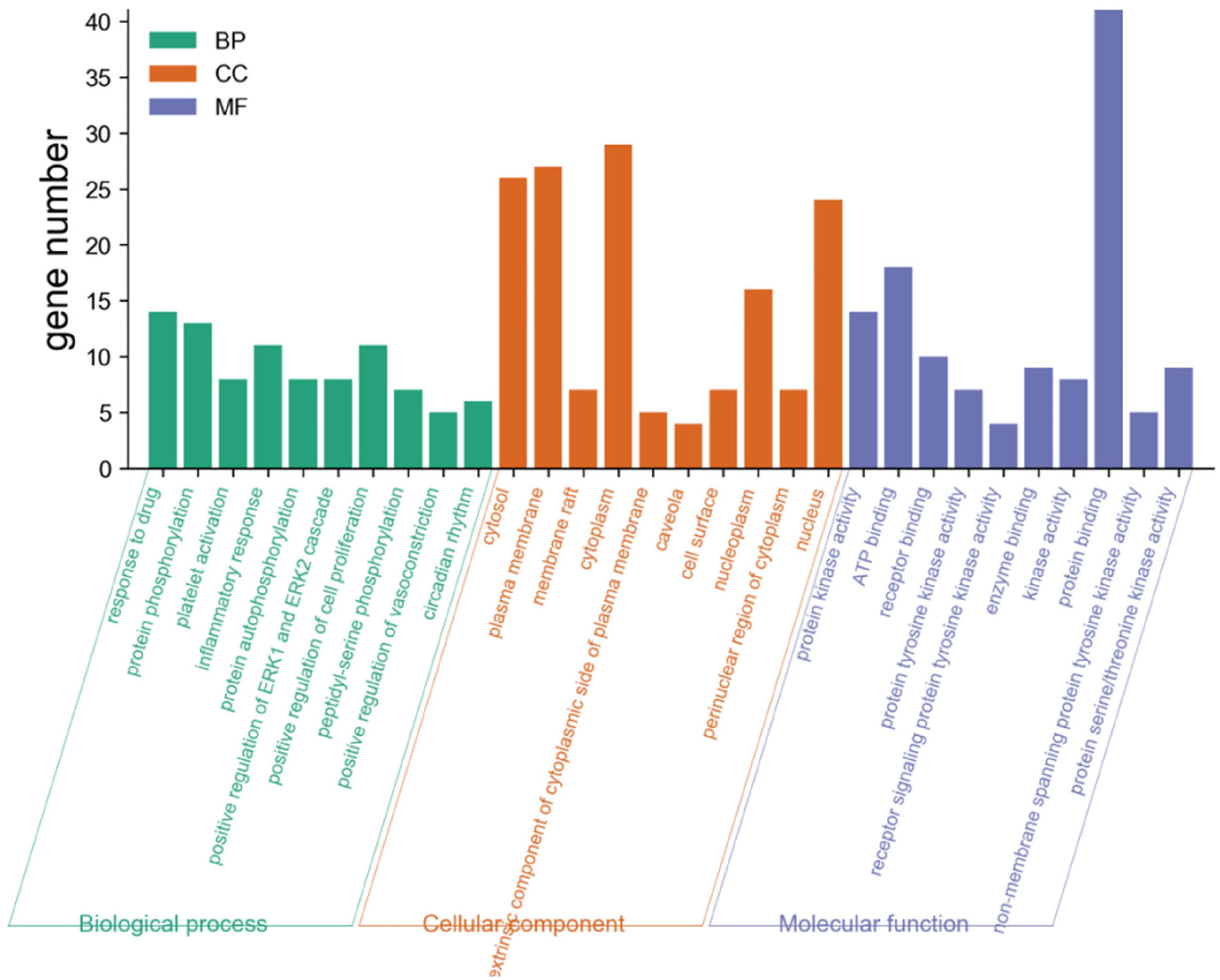
Bar chart of GO functional enrichment analysis.

Bubble diagram of KEGG enrichment analysis.

Results of KEGG analysis. The top 20 signaling pathways from KEGG analysis.

Distribution of the target proteins of RC on the predicted pathways (The red nodes are potential target proteins of RC).
Molecular Docking Results of the Core Components with IL-6, SARS-CoV-2 3CL, STAT3, and ACE2
The lower the binding energy, the greater the affinity between the receptor and ligand, and the greater the possibility of binding. Select key targets (IL-6 and STAT3) in the PPI network diagram for molecular docking verification. Because SARS-CoV-2 receptor binding and high expression levels of ACE2 are critical for COVID-19 invasion and have been used in the discovery of targeted drugs, 17 molecular docking was also carried out on SARS-CoV-2 3CL and ACE2 verify. Molecular docking results showed that the core chemical components of RC had good binding with receptor proteins (Table 3). The molecular docking energies of kaempferol with IL-6, SARS-CoV-2 3CL, STAT3, and ACE2 were −6.5 kJ/mol, −7.8 kJ/mol, −7.8 kJ/mol, and −7.6 kJ/mol, respectively (the molecular docking model diagrams are shown in Figures 10-13). The active sites of kaemferol and protein showed a compact binding pattern in the active pocket and formed hydrogen bond interaction with Asp187 and Ser 144 in SARS-CoV-23Cl hydrolase. Hydrogen bond interactions with GLU106 and THR43 were formed in IL-6. Hydrogen bond interactions with Ala396, Trp566, Tyr196, and Gln98 were formed in ACE2. Hydrogen bond interactions with CYS418, ASN420, and ARG379 were formed in STAT3. In the AutoDock environment, the absolute value of binding energy > 0.5 suggests that the result is meaningful, and the absolute value of binding energy > 5 suggests that the result is good. Based on these results, the conformation energy of kaempferol and the receptor proteins IL-6, SARS- CoV-2 3CL, STAT3, and ACE2 were low, and the conformation and conformation were stable.

kaempferol-SARS-CoV-2 3CL molecular docking mode.

kaempferol-IL-6 molecular docking mod.

kaempferol-ACE2 molecular docking pattern.

kaempferol-STAT3 molecular docking mode.
Binding Energy of the Core Active Components of Rhodiola crenulata with SARS-CoV-2 3Cl Hydrolase and ACE2.
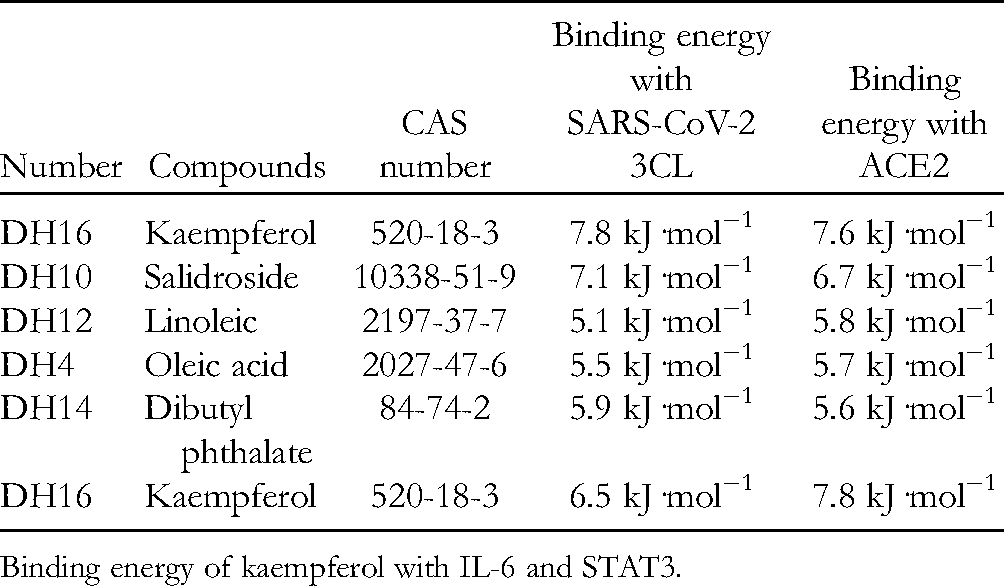
Binding energy of kaempferol with IL-6 and STAT3.
Kaempferol Effectively Attenuate Multiple Organ Injury in CS-Induced Rat
As shown in Figure 14A, the surface of lung tissues was dry and without hyperemia in the control group; lung tissues in the LPS group appeared hardened, congested, and oozed red liquid, a typical phenotype ALI. The morphological organization was better, and there was significantly less bleeding in the Kae and Dex groups.

(A) morphologic analysis (B) histochemistry analysis, magnification: 200×.
H&E staining was used to reveal the pathological changes in various organizations. As shown in Figure 14B, LPS administration was associated with significantly more inflammatory cell infiltration than the control group. The Kae and Dex group results suggested varying degrees of protection against LPS-induced MODS. Lung injury scores were calculated (Figure 14D) to show significant protective effects of Kae on the lung tissues compared with that of control group.
The Wet/Dry ratio reflects the degree of increased permeability of the pulmonary capillary membrane. We found that the Wet/Dry ratio in the lung was significantly higher than control in the LPS-treated group. In contrast, Kae appeared to reduce the Wet/Dry ratio (Figure 14C).
These results suggest that Kae attenuates the increased pulmonary capillary membrane permeability, relieves pulmonary edema, and reduces lung tissue injury and inflammatory infiltration in CS rats.
Kaempferol Inhibit LPS-Induced CS in Rat
We used an LPS-induced CS rat model to evaluate the inhibitory activity of kaempferol on systemic inflammation. The cytokine in serum and lung tissue homogenate of each group changed significantly. Compared with the normal control group, IL-10 decreased, and TNF-α and IL-6 increased significantly in the model group compared with the normal control group. Compared with the Kae treatment group, IL-10 increased, TNF-α, IL-6 decreased (Figure 15A). Compared with the normal control group, IL-10, TNF-α, and IL-6 increased significantly in the model group compared with the normal control group. Compared with the Kae treatment group, IL-10, TNF-α, IL-6 decreased (Figure 15B). These findings suggest that Kae potentially inhibits inflammatory cytokines secretion in the rat CS model.

(A) Detection of TNFα, IL-6, and IL-10 concentration changes in lung tissue homogenate by ELISA. (B) Detection of TNFα, IL-6, and IL-10 concentration changes in serum by ELISA. (C) The ratio of wet-to-dry weight. (D) Lung injury score.
Discussion
The COVID-19 pandemic exacted substantial social and economic costs worldwide. Previous studies showed that SARS-CoV-2 infects lung epithelial cells and elicits a robust local inflammatory response. When pro-inflammatory factors reach the circulation, they further stimulate immature granulocytes in the bone marrow to release large amounts of IL −6, TNF-α, and IFN-γ, culminating in CS and inducing ARDS. Levels of TNF-α, IFN-γ, IL-2, IL-6, and IL-1β in patients with severe disease were higher than those in the control group, and IL-6 levels positively correlated with COVID-19 severity.18,19 These findings suggest that direct inhibiting of massive cytokines released by hyperactivated immune cells, curbing the development of CS is critical to the treatment of severe COVID-19.
The Tibetan medicine RC has strong anti-inflammatory properties that reduce the secretion of inflammatory factors (TNF-α, IL-6, and IL-1β), 11 significantly reduce the infiltration of pulmonary neutrophils and inflammatory exudation, reduce pulmonary edema, and protect against ALI.
In the network of ingredient-target-disease, ingredients with higher degrees included kaempferol, linoleic acid, dibutyl phthalate, salidroside, and oleic acid. Kaempferol is a flavonoid component. Studies showed that kaempferol reduces LPS-induced lung injury by regulating TRAF6 polyubiquitination and exerts antibacterial and anti-inflammatory effects. 20 Kaempferol reduces the production of macrophage-derived chemokines, various pro-inflammatory factors (IL-6 and TNF-α), significantly reduces HMGB1 at the transcription and translation levels, and inhibits the TLR4/MyD88 inflammatory pathway, thereby inhibiting the development of inflammation.21,22 Studies showed that salidroside has substantial anti-inflammatory properties and participates in reducing various viscera inflammatory responses, primarily through NF-κB, MAPK, and other pathways to inhibit the secretion of inflammatory mediators. Salidroside reduced the accumulation of inflammatory cells in lung tissues of mice by inhibiting the phosphorylation of NF-κB, reducing pulmonary edema, and decreasing levels of TNF-α, IL-6, and IL-1β, thereby increasing survival in mice with sepsis. 23 Previous studies on the anti-inflammatory effects of RC, we found that salidroside inhibits inflammations, apoptosis, and autophagy in serum and liver tissues. 24 Other studies showed shown that salidroside significantly inhibited NF-κB phosphorylation in a dose-dependent manner via the Notch/HES signaling pathway, and also blocked extracellular signal-regulated kinase/mitogen-activated protein kinase, thereby reducing the secretion of TNF-α, IL-6, and IL-1β, significantly reducing the infiltration and inflammatory exudation of lung neutrophils, reducing pulmonary edema and increasing the survival of septic mice; it also exerted anti-infection and protective effects against ALI caused by sepsis in mice.25,26 Studies showed that salidroside could also ameliorate alcohol-induced hepatitis damage by regulating the inflammatory response of macrophages. 27 In a study of ALI caused by LPS, Sal significantly reduced the activity of pro-inflammatory factors TNF-α, IL-6, and IL-1β in lung tissue homogenates by inhibiting the TLR /NF-κB pathway. Sal increased serum levels of anti-inflammatory cytokines to a relatively high level, reducing excessive activation of inflammatory cells, the massive release of inflammatory mediators, and the loss of balance in the regulatory network. 28 Dibutyl phthalate (DBP) is often used as a medicine and nutritional supplement in medical treatment. Li et al showed that levels of IL-1β, IL-6, and TNF-α in macrophages of peritoneal exudates after 24-h treatment with DBP decreased significantly, suggesting that DBP inhibits the production of inflammatory cytokinins. 29 Linoleic and oleic acids are unsaturated fatty acids that regulate cytokine levels. Oleic acid inhibits the nuclear translocation of GATA binding protein 3 (GATA-3) and prevents the activation of GATA-3 through upregulation of Foxp3, thereby inhibiting the infiltration of inflammatory cells in bronchoalveolar lavage fluid and inhibiting the gene expression of IL-6, IL-13, and COX-2 in lung tissue. 30 In the treatment of experimental sepsis, oleic acid was shown to balance cytokine production, control inflammation, and increase bacterial clearance by activating the expression of peroxisome proliferator-activated receptor γ (PPARγ), upregulating IL-10 and other anti-inflammatory factors, and downregulating TNF-α, IL-1β, and other pro-inflammatory cytokines. 31 Linoleic acid inhibits the expression of TNF-α cell surface receptor TNFR2 through a G protein-coupled receptor (GPR40), thereby reducing the expression of TNF-α and its mRNA levels in the colon and alleviating inflammation. 32 Linoleic acid also promotes wound healing by reducing inflammatory cells’ number of inflammatory factors (IL-6, IL-1, and TNF-α) and macrophage inflammatory proteins. Linoleic acid also inhibits the activation of NF-κB by long-chain fatty acid receptor PPARγ to inhibit NF-κB activity and mediate anti-inflammation. 33 In summary, the active ingredients in RC, including dibutyl phthalate, kaempferol, oleic acid, salidroside and linoleic acid, preventing CS, consistent with our hypothesis.
We predicted that the core targets of CS prevention in RC were IL6, STAT3, SRC, AKT1, EGFR, and CXCL8. COVID-19-related CS is triggered by rapid activation of pathogenic Th1 cells and secretion of pro-inflammatory cytokines and granulocyte-macrophage colony-stimulating factor (GM-CSF) and IL-6. GM-CSF activates CD14+ CD16+ inflammatory monocytes, producing IL-6, TNF-α, and other cytokines. 34 IL-6 is an essential inflammatory factor that enhances the adhesion and aggregation of neutrophils and delays apoptosis in neutrophils, 35 allowing inflammatory cytokines to accumulate in large quantities. A study showed that IL-6 and other inflammatory factors increase expression with the severity of the disease, and higher CS levels are associated with more severe disease. 36 Therefore, IL-6 can be used to perform rapid diagnosis in patients at high risk of disease progression. IL-6 blockers can balance immune activation and inflammation suppression and provide treatment strategies for patients with severe and critical COVID-19.
Akt1 is an essential downstream factor in the PI3K signaling pathway. PI3K and Akt1 protein expression positively correlated with the content of pro-inflammatory factors (such as IL-6) that mediate inflammatory damage to the alveoli; moreover, PI3K inhibitors and Akt1 gene deletion enhanced neutrophil apoptosis, thereby reducing the influx of neutrophils into the lung and reducing the expression of pro-inflammatory factors. 37 STAT3 mediates signal transduction of cytokines in cells, and there is a positive feedback regulation between STAT3 and IL-6 that promotes the production and secretion of IL-6 and other cytokines; in short, a series of inflammatory responses are activated; therefore, blocking IL-6-STAT3 may be a simple way to prevent COVID-19-induced CS. 38 These results suggest that RC may help resist CS through the synergistic activity of several components and targets, providing a basis for developing Tibetan antiviral drugs.
GO enrichment analysis results showed that biological processes were related to drug response, protein phosphorylation, and inflammatory response. Cell composition is related to cell solute, cytoplasm, and nucleus. Molecular functions influence protein kinase activity, ATP binding, and protein binding.
Studies showed that avian influenza virus infection causes severe tissue damage by excessively inducing airway epithelial cells to produce interferon and pro-inflammatory cytokines; the infection also promotes functional transformation through S473 phosphorylation, leading to increased levels of IFN-β, IL-6, and IL-8, triggering inflammatory responses to mediate respiratory damage, consistent with our GO analysis. 39 Combined with KEGG pathway enrichment analysis, the pathways involved in the treatment process included the TNF signaling pathway, the PI3K-Akt signaling pathway, and the IL6-JAK2-STAT3 signaling pathway. These signal pathways are related to immunity and inflammation, suggesting that RC may be used to prevent CS. This signal pathway inhibits the activation of inflammatory factors (IL-6), reduces excessive immune responses, and mediates anti-inflammation. These pathways are closely related to CS, which may be RC's mechanism of prevention and treatment.
IL6-JAK2-STAT3 signaling pathway participates through the following mechanisms in inflammation, IL-6 binds to GP130 receptors on the surface of T-lymphocytes and macrophages, upregulates the STAT3 phosphorylation pathway to activate NK-κB, and induces the polarized expression of intercellular adhesion molecules, triggering CS and ALI/ARDS. 40 TNF participates in inflammation, cell proliferation, and cell death. It is the principal inflammatory cytokine that activates the NF-κB signaling pathway. When the NF-κB signaling pathway is activated, various inflammatory cytokines and related enzymes are expressed, including COX-2, TNF-α, and IL-6, which stimulate macrophages to form inflammatory cascade reactions, exacerbating inflammation. 41 Zhao et al demonstrated that the PI3K/Akt1 signaling pathway participates in re-inducing pulmonary inflammation. The PI3K/Akt1 pathway is responsible for delayed lung neutrophil apoptosis. 37 These studies suggest that the activation of signaling pathways such as IL6-JAK2-STAT3, PI3K/Akt, TNF-α/NF-κB, mediate organ damage caused by CS and can serve as Kae potential therapeutic target.
Combined with previous studies on the anti-inflammatory effects of RC, we found that Kae resists inflammation and is a potential therapeutic agent for treating lung injury. These results are consistent with our KEGG results analysis. IL-6 levels in the blood are highly correlated with COVID-19 mortality and can be used as biomarkers to determine disease severity. Taken together, our findings suggest RC inhibits CS via multi-component, multi-target, and multi-signaling pathways.
Molecular docking is a computer-aided drug design method. The binding energy of docking molecules and targets is divided into three categories by Vina: −4.0 kJ/mol < 0 kJ/mol, indicating binding activity; −7.0 kJ/mol < −4.0 kJ/mol, indicating good binding activity; binding energy < −7.0 kJ/mol indicates strong binding activity. According to the docking results, the core components of RC, including kaempferol (DH16), linoleic acid (DH12), dibutyl phthalate (DH14), salidroside (DH10), and oleic acid (DH4), had high absolute conformational binding energy to SARS-CoV-2 and ACE2 had good binding ability. These results suggest that these five core compounds combine with SARS-CoV-2 3Cl hydrolase and ACE2 to prevent and treat COVID-19-induced inflammatory CS. Of these, kaempferol was the best active component. This method was used for reverse verification of drug target matching simulation to interpret the scientific nature of RC's activity against anti-COVID-19-induced inflammatory CS and to provide a basis for future research on targets and pathways for Chinese and Tibetan medicines. This method can also explore the synergies involved in pharmacological mechanisms of various Chinese and Tibetan medicines.
After COVID-19 patients infected with the virus, cytokines such as TNF-α, IL-10, and IL-6 increase rapidly, and with the imbalance of pro-inflammatory and anti-inflammatory balance, aggravating the inflammatory response, the immune function of the body is disordered, abnormal immune responses appear, the occurrence of CS is triggered. 42 We found that Kae significantly reduced IL-6 and TNF-α levels in serum and balanced inflammatory response protect the body from inflammatory damage. Kae has a wide range of pharmacological effects, the most significant of which is inhibition of inflammatory factors and reducing their expression levels. In the present study, we demonstrate that Kae inhibits LPS-induced ALI in rats. Furthermore, Kae significantly attenuates the release of proinflammatory cytokines TNF-α and IL-6 and increases the release of anti-inflammatory cytokines IL-10 in lung tissue homogenates. Kae effectively protects various tissues and organs from CS damage while reducing systemic inflammatory response. This may provide evidence for the treatment of using RC.
In summary, the main active components, targets, key signal pathways, and biological functions of RC anti-CS predicted by network data mining suggest that RC involves several components and targets. Our findings are consistent with the reported literature, which provides a basis for future research. Network pharmacology is based on the data in the literature and uses many computer data calculation methods to provide references for the research; however, it may cause the results to become inaccurate. This is the limitation of the prediction. Therefore, further research is needed in the future.
Conclusion
In summary, RC was identified to effectively prevent the development of CS in LPS-induced rats. They can also effectively alleviate the lung injury of LPS-induced rats. Emerging evidence have supported that the CS triggered by immune disorders leads to the disorder of proteins such as CDC42, RAC1/RAC2 and RHOA, lead to the depletion of ATP required for the dynamic regulation of renal podocyte-actin, which in turn causes podocyte dyskinesia, glomerular sclerosis, and kidney damage. We may pay close attention to the clinical signs of kidney injury in patients. 43 It is suggested that retrospective studies may be performed to further determine Kae anti-CS effects in COVID-19 patients combined with kidney injury.
Footnotes
Authors note
All authors are currently affiliated with Engineering Research Center of Tibetan Medicine Detection Technology”, Engineering Research Center of Tibetan Medicine Detection Technology, Ministry of Education, School of Medicine, Xizang Minzu University, Xianyang 712082, China.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This study was supported by the National Natural Science Foundation of China (Grant No. 81760332), the Tibet Natural Science Foundation (Grant No. ZRKX2021000055), Youth project of the Tibet Natural Science Foundation (Grant No. XZ202001ZR0053G), the Research Found for Advanced Talents of Xizang Minzu University (Grant No. RCYJ602111), National Natural Science Foundation of China, Medical School of Xizang University for Nationalities (Grant No. XZMZ-M2022N02), and 2020 national university students innovation research project (Grant No. 202010695029).
