Abstract
Ganoderma lucidum (G. lucidum) is one of the well-known mushrooms in China, which has G. lucidum polysaccharides (GLP) that have been widely studied for various biological activities, such as antioxidant, antitumor, antiinflammatory, antiviral, antidiabetes, and immunomodulatory activities. A signal transducer and activator of transcription (STAT) signaling pathway is related to cell proliferation and apoptosis. The relationship between STAT and intestinal protection of GLP is still unknown. We studied the inhibitors AG490 in the STAT pathway and its downstream molecules to analyze the unique effects in the protection of GLP against palmitic acid (PA)-induced porcine intestinal epithelial cells (IPEC-J2) injury. Compared to PA treatment, GLP + PA obviously decreased Ca2+ concentration, H2O2 production, NF-E2-related factor 2 (Nrf2) nuclear translocation, STAT1 and STAT2 protein levels, and increased nuclear factor kappa-B (NF-κB) nuclear translocation and p-STAT3/STAT3 ratio in IPEC-J2 cells. After inhibition of STAT3 signaling, p-STAT3/STAT3 ratio, NF-κB nuclear translocation obviously decreased and Nrf2 nuclear translocation significantly increased in the GLP + PA group. The protection of GLP on proliferation and apoptosis of PA-induced IPEC-J2 cells was suppressed by inhibiting STAT3. The STAT3 pathway regulated the enterocyte-protective effects of GLP by modulating the nuclear translocation of Nrf2 and NF-κB. We provide new insights into the mechanism of STAT signaling for the protection of GLP on PA-induced intestinal epithelial cell injury.
Introduction
Palmitic acid (PA) is the main saturated fatty acid naturally occurring in animal fats and vegetable oils. In recent decades, palm oil containing a high concentration of PA has been widely used in edible oils. A high-fat diet led to barrier dysfunction, exacerbated dextran sodium sulfate-induced colitis, and increased intestinal tumorigenesis in mice. 1 A new study showed that dietary PA, but not oleic acid or linoleic acid, promotes metastasis of oral carcinomas and melanoma in mice. Researchers provided evidence that a dietary metabolite induces stable transcriptional and chromatin changes that lead to the long-term stimulation of metastasis, and that this is related to a proregenerative state of tumor-activated Schwann cells. 2 However, most previous research has ignored the effect of a high-fat diet on the intestine before the occurrence of the disease.
As an important Chinese food and product used to promote health, Ganoderma lucidum (G. lucidum) has been used in China for more than 2000 years. 3 The current research found that G. lucidum has a high nutritional value, which has received great attention from businesses and academia in Asia, North America, and some European countries. 4 It is included in the “Pharmacopoeia of the People's Republic of China” as Chinese medicine and in the “American Herbal Pharmacopoeia and Therapeutic Compendium” as herbal medicine. Compared with herbicides and other products, the safety of human food and health products is essential. 5 The levels of CD3, CD4, CD8, CD19, and markers of inflammation including plasma interleukin (IL)-18, interferon-γ (IFN-γ)-inducible protein 10, and monocyte chemoattractant protein 1, were unchanged after oral administration of G. lucidum in a double-blind randomized placebo-controlled trial, suggesting that G. lucidum is safe and well tolerated. 6 Polysaccharides are one of the main active ingredients of G. lucidum. G. lucidum polysaccharides (GLP) improved the expression of occludin, nuclear factor kappa-B (NF-κB) p65, and secretory immunoglobulin A in the ileum enhanced the levels of IFN-γ, IL-2, and IL-4, and increased the microbiota richness in cecum compared with the control group of rats. 7 It was reported that GLP extracted from spores alleviated paclitaxel-induced small intestinal barrier dysfunction in breast cancer mice and IEC-6 cells. 8 Furthermore, GLP also displayed recovery activities on the intestinal barrier by restraining macrophage-responsible inflammation. 9 Spores of G. lucidum can reverse paclitaxel-induced body weight loss and remodel the histology of the small intestine, accompanied by increased proliferation but suppressed apoptosis in intestinal cells. 8 Some evidence suggests that Ganoderma atrum polysaccharides could ameliorate Cy-induced impairment of intestinal immunity and mucosal integrity, which may be associated with autophagy in mice. 10
The signal transducer and activator of transcription (STAT) pathway is a rapid signaling pathway from the extracellular milieu to the nucleus, the stimulation of which is initiated by binding of the transmembrane cytokine receptor monomers to extracellular ligands, resulting in receptor dimerization. 11 The STAT protein family consists of seven proteins (STAT1, 2, 3, 4, 5A, 5B, and 6) that are cytoplasmic transcription factors that play a key role in cell fate, which has made them the focus of many investigations. 12 Among them, STAT3 is the most active member of the STAT family. Whether in neural stem cells or embryonic stem cells, STAT3 plays a fundamental role in regulating cell fate. 13 After STAT3 is phosphorylated, it dimerizes and is transferred into the cell nucleus, where it binds to the DNA promoter of its target genes to activate their transcription. 14 STAT1 and STAT2 are mainly activated by IFN, which regulates the expression of many genes involved in cellular physiological processes. In the classic process of IFN signaling, tyrosine at position 701 of the STAT1 protein and tyrosine at position 690 of the STAT2 protein are phosphorylated, thereby forming hetero or homodimers in the nucleus and inducing downstream gene expression to prevent harmful material damage. 15 The rapid inactivation of STAT by its phosphorylation makes its regulation of downstream genes a time-controlled process.16,17 Zhang et al 18 noted that intestinal vitamin D receptor protects mice against dysbiosis by regulating the STAT pathway in tumorigenesis. Other studies have shown that bleomycin as a DNA damage-inducing reagent, stimulates intestinal stem cells to proliferate noncell-autonomously through the Janus kinase (JAK)/STAT pathway. 19 Lactobacillus rhamnosus GG upregulates leptin levels in the gut epithelium, and leptin then acts as a chemokine promoting cell proliferation through increased JAK-STAT pathway signaling. 20 GLP can reduce melanogenesis in melanocytes by inhibiting the paracrine effects of keratinocytes and fibroblasts via the IL-6/STAT3/fibroblast growth factor 2 (FGF2) pathway. 21 Although this signaling pathway has not been thoroughly studied, it has been clearly identified as an important intracellular signal transduction pathway that participates in cell proliferation, differentiation, and apoptosis. 22
Our previous study found that GLP effectively alleviated intestinal oxidative stress caused by high-fat diets and reduced apoptosis and autophagy caused by a high lipid composition in intestinal epithelial cells via suppression of the AMP-activated protein kinase/Akt/mammalian target of rapamycin (mTOR) signaling pathway. 23 To our knowledge, there is limited information available thus far about the relationship between the effects of GLP on high-fat diet-induced intestinal cell damage and the STAT pathway. As the intestine is one of the key immune and absorptive organs consisting of a monolayer of epithelial cells, and there is a good protective effect of GLP on the intestine, we hypothesized that GLP might protect against saturated fatty acid-mediated intestinal epithelial cell proliferation and apoptosis via the STAT pathway.
Therefore, the current study investigated whether GLP would have beneficial effects on the prevention of endothelial cell injury under high-fat conditions via regulation of the STAT pathway. Our data suggested that STAT3 acted as an important molecular target in the protective actions of GLP against PA-mediated IPEC-J2 cell injury, including apoptosis, cell cycle arrest, and cytotoxicity, wherein NF-E2-related factor 2 (Nrf2) and NF-κB were downstream pathways.
Materials and Methods
Polysaccharides Preparation
A fruit body slice of G. lucidum (Leyss. ex Fr.) was obtained by Changsha Diwei Agricultural Technology Co., Ltd. GLP was extracted and purified according to our previous method. 24 The polysaccharide structure was determined by examining infrared spectra, ultraviolet spectrum, and high-performance anion exchange chromatography, and the primary structure of GLP was mainly consisting of glucose, arabinose, cellulose, and galactose in the molar ratio of 16.67:61.11:5.56:16.67. The molecular weight composition of the GLP is as follows: >50 kDa, 46.1%; 30-50 kDa, 21.8%; 10-30 kDa, 32.1%. GLP has an α-glycosidic bond and is an acidic polysaccharide containing 11% of uronic acid and 89% of total carbohydrate.
Cell Culture and Drug Treatment
Porcine enterocytes IPEC-J2 were donated by Hunan Agricultural University, which were cultured in 1640 medium (Sigma), supplemented with 10% fetal bovine serum (Sigma), 1% penicillin-streptomycin (Beyotime) in a humidified 5% CO2 atmosphere at 37°C. GLP was dissolved in 1640 medium to provide a stock solution of 10 mg/mL.
Cell Growth
MTT Cell Cytotoxicity Assay Kit (Beyotime) at 490 nm on a microplate reader (Thermo Multiskan MK3) according to the manufacturer's instructions.
Intracellular Calcium Concentration
Cells were treated with 600 μM PA or/and GLP (0.4 and 0.8 mg/mL) for 24 h, and then loaded with 1 μM Fluo-4 AM working solution in phosphatic buffer solution (PBS, pH 7.4) to cover the cells and incubated at 37°C for 30 min. Subsequently, cells were washed with phenol red-free 1640 medium 3 times. Pictures were captured under an inverted fluorescence microscope (Olympus, CKX41SF) and a microplate reader (TECAN, Infinite 200 PRO) by excitation/emission at 488 nm/520 nm wavelength.
Intracellular H2O2 Production
After incubation with PA or/and GLP for 24 h, cells were washed by PBS and lysed in RIPA lysis buffer (Beyotime) at 4°C. Lysis buffer was centrifuged for 5 min at 4°C at 10 000 × g. Then, cells were added to the reaction system and the accumulation of intracellular H2O2 was analyzed using a hydrogen peroxide assay kit (Jiancheng) as the description of the manufacturer's instructions. The optical density at the wavelength of 405 nm was detected using a microplate reader (Thermo).
Cell Apoptosis Assay
Cells were pretreated with 50 µM AG490 for 1 h and then incubated with 600 µM PA or 0.6 mg/mL GLP for 12 h. After washing with PBS twice, cells were collected by centrifugation for 5 min at 2000 × g. The collected cells were added to 500 μL binding buffer and the mixture of an equal volume of 5 μL Annexin V-FITC and 5 μL propidium iodide (PI) followed by incubation for 15 min in the dark at room temperature. Cell apoptosis was analyzed on a Beckman flow cytometer (Beckman).
Cell Cycle
Cells were pretreated with 50 µM AG490 for 1 h and then incubated with 600 µM PA or 0.6 mg/mL GLP for 12 h. They were washed three times in 10 mL of prechilled PBS and centrifuged at 800 × g for 5 min. The cell pellet was gently resuspended in 400 μL PBS to separate them into individual cells, followed by adding 1.2 mL of prechilled 100% ethanol (final concentration 75%) and held at 4°C overnight. After ethanol was separated by centrifugation at 800 × g for 5 min, a sample was added 150 μL PI and incubated in the dark at 4°C for 30 min. The cell cycle was acquired on a flow cytometer (Beckman).
Western Blot Analysis
Treated IPEC-J2 cells were collected in protein buffer with phosphatase and protease inhibitors (1% Triton X-100, 150 mM NaCl, 50 mM Tris–HCl). After cells were lysed by RIPA Lysis Buffer (Beyotime), total protein was extracted using a Total Protein Extraction Kit (Jiancheng), and cytosolic and nuclear proteins were isolated using a Cytosol Protein Extraction Kit (Jiancheng), and nuclear protein extraction kit (Jiancheng), respectively. Protein concentration was determined by the BCA Protein Assay Kit (Jiancheng). Proteins were separated by 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis and transferred onto a polyvinylidene fluoride membrane. Finally, the protein bands were quantified using a chemiluminescence-detecting kit (GE Healthcare). The primary antibodies employed for western blot analysis were as follows: NF-κB (10745-1-AP, Proteintech, 1:1000), Nrf2 (16396-1-AP, Proteintech, 1:700), STAT1 (10144-2-AP, Proteintech, 1:500), STAT2 (16674-1-AP, Proteintech, 1:500), STAT3 (60199-1-Ig, Proteintech, 1:3000), p-STAT3 (ab76315, Abcam, 1:3000), proliferating cell nuclear antigen (PCNA; 16396-1-AP, Proteintech, 1:2000), and β-actin (60008-1-Ig, Proteintech, 1:5000).
Statistical Analysis
All values were expressed as mean ± SD and all experiments were performed three times. The statistical comparisons were performed using a one-way analysis of variance with a subsequent least significant difference test using the SPSS (v.19.0). P < .05 and P < .01 were considered to be significant and extremely significant, respectively.
Results
The Effect of PA and GLP on IPEC-J2 Cell Growth
IPEC-J2 cells were treated with GLP or PA for 24 h, and IPEC-J2 cell growth was tested by MTT assay. The data shown in Table 1 indicate that the treatment of IPEC-J2 cells with different concentrations of PA caused a concentration-dependent reduction in cell growth. GLP alone did not prevent cell growth, and 0.6 and 1.2 mg/mL GLP even promoted growth, suggesting that GLP in the concentration ranges tested did not lead to IPEC-J2 cell death. Moreover, treatment of IPEC-J2 cells with GLP significantly inhibited the PA-induced reduction in cell growth (Table 2).
Effect of PA and GLP Alone on IPEC-J2 Cell Growth.

Abbreviations: GLP, Ganoderma lucidum polysaccharides; PA, palmitic acid.
Data are presented as the mean ± SD. a <0.01 versus control.
Effect of GLP on the Growth of IPEC-J2 Cell Exposed to H2O2.

Abbreviations: GLP, Ganoderma lucidum polysaccharides; PA, palmitic acid.
Data are presented as the mean ± SD. a <0.05 and <0.01 versus control; b <0.01 versus PA treatment.
GLP Prevented the PA-Mediated Increase in the Intracellular Ca2+ Concentration in IPEC-J2 Cells
As a critical second messenger, Ca2+ is essential for the modulation of cell growth and apoptosis. Therefore, the intracellular Ca2+ concentration was examined in IPEC-J2 cells using Fluo-4 AM dye. As shown in Figure 1, compared to control cells, IPEC-J2 cells treated with PA exhibited an ∼2-fold increase in green fluorescence, showing a clear reduction in intracellular Ca2+ concentration in PA-treated cells. Compared to PA treatment, the 0.4 and 0.8 mg/mL doses of GLP obviously attenuated the PA-induced increase in the concentration of intracellular Ca2+ (P < .01), evidencing a significant reduction in the intensity of the signal in PA + GLP-treated cells.
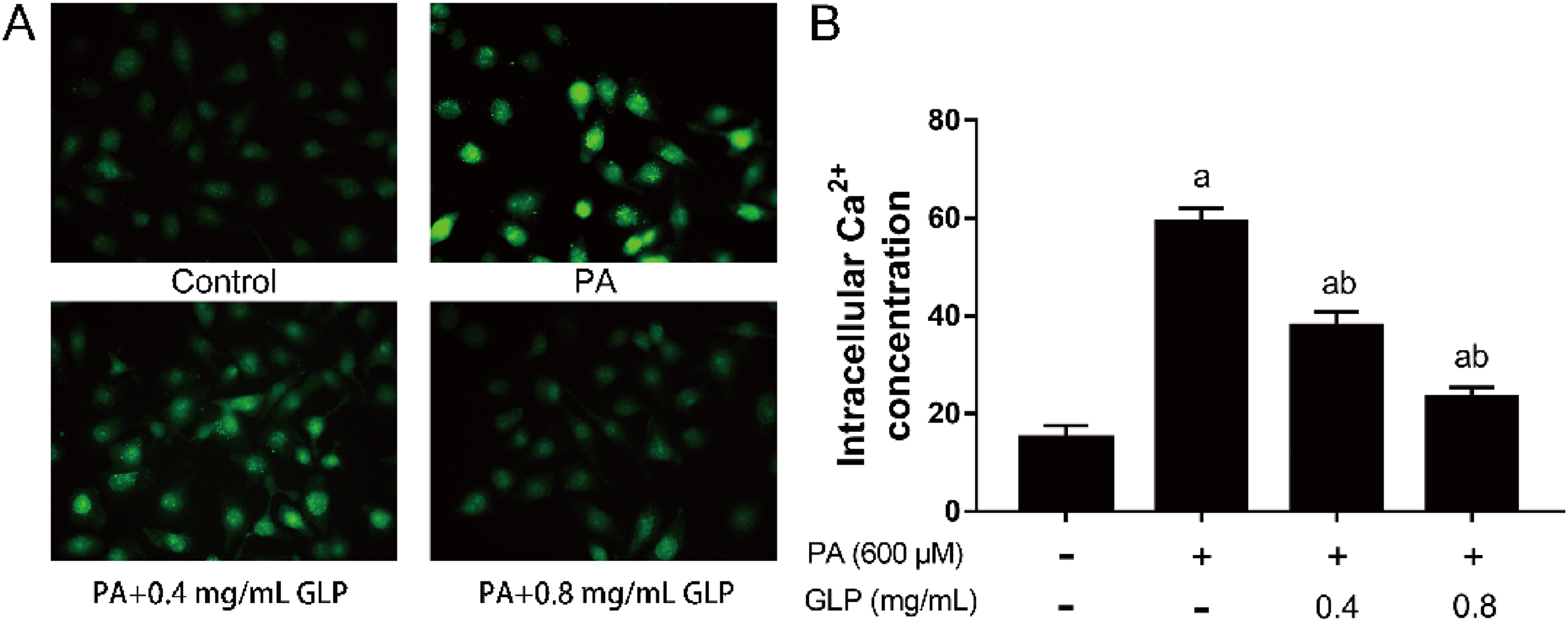
GLP prevented Ca2+ concentration from increasing in PA-treated IPEC-J2 cells. After cells were treated with 600 μM PA or/and GLP (0.4 and 0.8 mg/mL) for 24 h, the intracellular Ca2+ concentration was evaluated under an inverted fluorescence microscope (A) and using a microplate reader (B). Magnifications: 200×; scale bars: 100 μm. Data are presented as the mean ± SD. aP < .01 versus control; bP < .01 versus PA treatment.
GLP Inhibited PA-Mediated Intracellular H2O2 Production in IPEC-J2 Cells
We next examined whether GLP affected the accumulation of intracellular H2O2 in PA-treated cells. The results shown in Figure 2 indicated that increased production of intracellular H2O2 was observed in the PA-treated cells compared with the control cells. Intracellular H2O2 production was obviously reduced by 0.8 mg/mL GLP compared to PA-treated IPEC-J2 cells (P < .01), indicating that GLP effectivity prevents the PA-induced production of intracellular H2O2. However, a dose of 0.4 mg/mL GLP did not obviously decrease the intracellular H2O2 production in IPEC-J2 cells induced by PA.

GLP inhibited PA-treated intracellular H2O2 production in IPEC-J2 cells. After cells were treated with 600 μM PA or/and GLP (0.4 and 0.8 mg/mL) for 24 h, intracellular H2O2 production was detected using a microplate reader at the wavelength of 405 nm. Data are presented as the mean ± SD. AP < .05 and aP < .01 versus control; bP < .01 versus PA treatment.
GLP Activated the STAT Signaling Pathway in PA-Treated IPEC-J2 Cells
As the STAT signaling pathway plays a critical role in cell apoptosis and growth, we next determined the effect of GLP on PA-mediated STAT1, STAT2, and STAT3 in IPEC-J2 cells. As illustrated in Figure 3A, compared with untreated cells, PA-treated cells showed a decrease in p-STAT3 protein expression and increases in the expression levels of STAT1 and STAT2 proteins. Upregulation of STAT1 and STAT2 induced by PA was reduced, and downregulation of p-STAT3 caused by PA was increased in the presence of PA and GLP, indicating that GLP activated the STAT pathway at the protein level. Additionally, compared with control cells, the STAT3 protein level did not show a marked change in either PA-treated cells or PA + GLP-treated cells. Notably, GLP elevated the decreased p-STAT3/STAT3 ratio induced by PA at 12 h but not at 24 h.

GLP regulated the expression levels of STATs, Nrf2, and NF-κB proteins in PA-treated IPEC-J2 cells. After cells were treated with 600 μM PA or/and 0.6 mg/mL GLP for 12 and 24 h, relative protein levels of (A) STAT1, STAT2, STAT3, and p-STAT3, as well as (B) the expression levels of Nrf2 and NF-κB in the cytoplasm and nucleus, were detected by western blotting. β-actin and PCNA were used as controls. Data are presented as the mean ± SD. AP < .05 and aP < .01 versus control; bP < .01 versus PA treatment.
GLP Disrupted PA-Mediated Nrf2 Nuclear Translocation and NF-κB Protein Expression
As shown in Figure 3B, we found that PA incubation promoted the expression of NF-κB protein in IPEC-J2 cells, while cotreatment with GLP and PA obviously attenuated the increase in the expression of NF-κB protein. In addition, PA-induced IPEC-J2 cells showed an upregulation of Nrf2 protein expression and a downregulation of Nrf2 expression in the nucleus as well as a downregulation of Nrf2 protein expression and an upregulation of NF-κB expression in the cytoplasm GLP also disrupted the nuclear translocation of Nrf2 and promoted the nuclear translocation of NF-κB at a dose of 0.6 mg/mL for 12 and 24 h. Furthermore, the effects of GLP on NF-κB protein and Nrf2 nuclear translocation were significantly greater at 12 h than at 24 h.
AG490 Reversed the Cytoprotective Effect of GLP in PA-Treated IPEC-J2 Cells
Because activation of the STAT pathway was positively associated with the protection of GLP, we next examined IPEC-J2 cell growth in the presence of different concentrations of the specific JAK inhibitor AG490 using an MTT assay. After incubating cells with 10, 30, or 50 µM AG490 for 24 h, we observed that the IPEC-J2 cell numbers were significantly reduced compared to the vehicle-treated cells (Figure 4A). Additionally, IPEC-J2 cells exposed to PA and GLP for 12 and 24 h exhibited significantly higher cell growth than those cotreated with PA, GLP, and AG490 for 12 h and 24 h, respectively (Figure 4B). This revealed that AG490 treatment effectively prevented the cytoprotective effect of GLP against IPEC-J2 cell damage via inhibition of PA-induced activation of the STAT3 pathway. 25

AG490 reduced the cytoprotective effect of GLP in PA-induced IPEC-J2 cells. (A) Cells were exposed to 10, 30, and 50 µM AG490 for 24 h, *P < .05 and **P < .01 versus control; (B) cells were pretreated with 50 µM AG490 for 1 h, and then incubated with 600 µM PA or/and 0.6 mg/mL GLP for 12 or 24 h, **P < .01. Cell growth was determined by MTT assay. Data are presented as the mean ± SD.
AG490 Blocked the Protective Effect of GLP on PA-Induced Cell Cycle Arrest and Apoptosis in IPEC-J2 Cells
To further explore the mechanism of cell growth inhibition by AG490, we examined the effects of AG490 on apoptosis and cell cycle progression in PA + GLP-treated IPEC-J2 cells. As shown in Figure 5A, the populations in the G2 and S phases were markedly decreased, and that in the G1 phase was increased after the cells were treated with PA for 12 h, compared with control cells. GLP exposure partly reversed the G1 phase arrest caused by PA. In addition, suppression of the STAT signaling pathway decreased the G1 and G2 phase populations and increased the S phase population at 12 h after cells were pretreated with 50 μM AG490 for 1 h. These results indicated that cell growth inhibition by AG490 is mediated by a combination of apoptosis induction and inhibition of cell cycle progression. As shown in Figure 5B, 600 μM PA significantly promoted apoptotic IPEC-J2 cells, while 0.6 mg/mL GLP inhibited PA-induced cell apoptosis. IPEC-J2 cell pretreatment with AG490 for 1 h markedly increased the early apoptotic rate and the later apoptotic rate in the presence of PA and GLP.

AG490 reversed the cytoprotective effect of GLP in PA-induced IPEC-J2 cells. (A) Cells were exposed to 10, 30, and 50 µM AG490 for 24 h, *P < .05 and **P < .01 versus control; (B) cells were pretreated with 50 µM AG490 for 1 h, and then incubated with 600 µM PA or/and 0.6 mg/mL GLP for 12 or 24 h, **P < .01. Cell growth was determined by MTT assay. Data are presented as the mean ± SD.
AG490 Reversed the Effect of GLP on NF-κB and Nrf2 Proteins in PA-Treated IPEC-J2 Cells
To systematically elaborate on the intricate regulatory mechanism, we further explored the possible crosstalk between the Nrf2/NF-κB and JAK2/STAT3 signaling pathways. The results in Figure 6 show that AG490 reduced not only NF-κB nuclear translocation but also p-STAT3 protein expression. Nonetheless, AG490 efficiently increased Nrf2 nuclear translocation and STAT3 protein expression. It is suggested that GLP and PA treatment inactivated the STAT3 pathway, inhibited the NF-κB pathway, and activated the Nrf2 pathway. Taken together, these results demonstrated that STAT3 connected the crosstalk between Nrf2 and NF-κB signals in the protective effect of GLP.
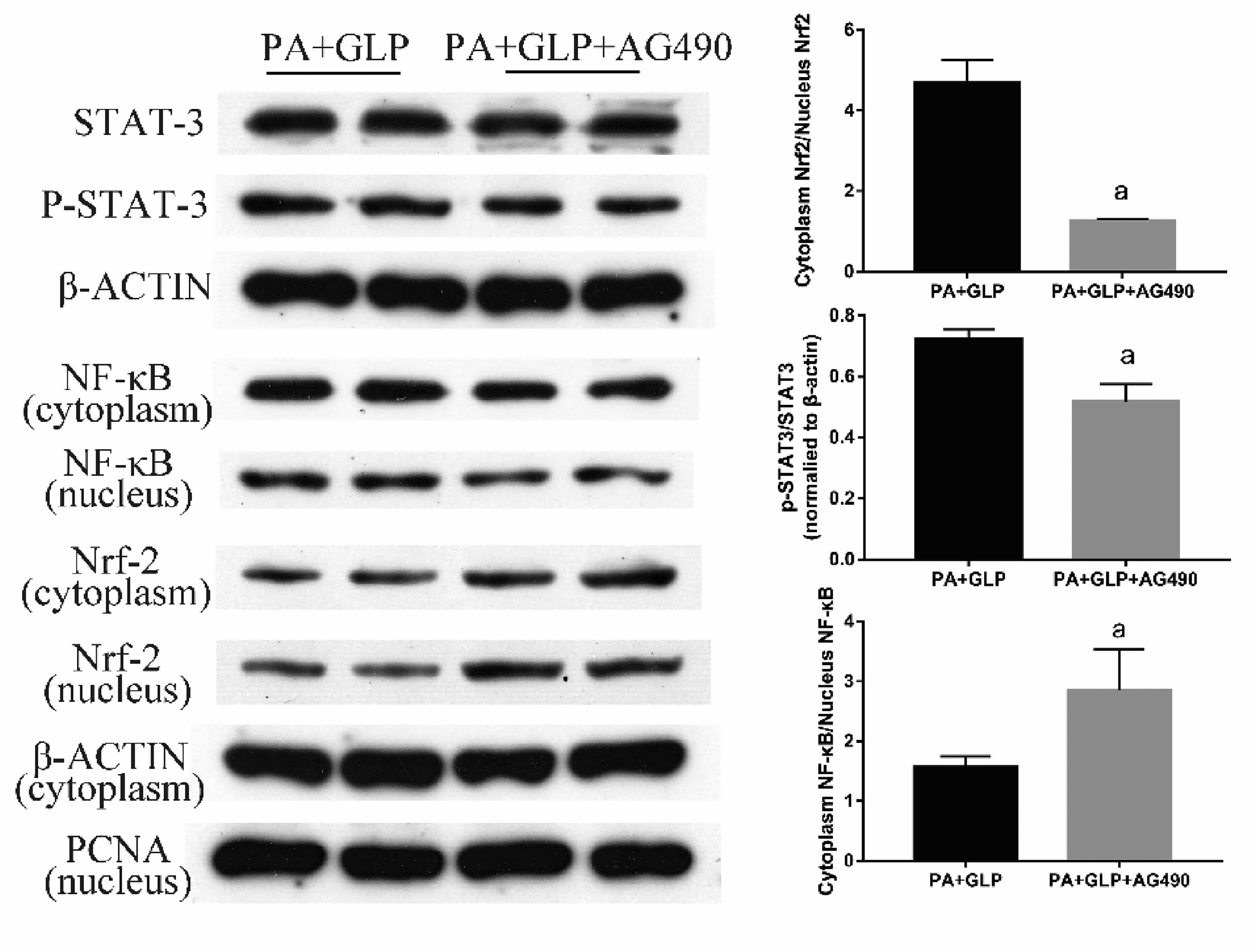
AG490 reversed the effect of GLP on apoptosis-related proteins in PA-treated IPEC-J2 cells. Cells were pretreated with 50 µM AG490 for 1 h and then incubated with 600 µM PA or 0.6 mg/mL GLP for 12 h. Then, the relative protein levels of STAT3 and p-STAT3 and the expression levels of Nrf2 and NF-κB in the cytoplasm and nucleus were quantified by Western blotting. β-actin and PCNA were used as a loading control. Data are presented as the mean ± SD. A P < .05 and a P < .01 versus PA + GLP treatment.
Discussion
The pathogenetic role of the STAT signaling pathway has been tested recently in several models of intestinal injury, such as myocardial ischemia-reperfusion injury, 26 intestinal inflammation (eg, enterocytes and intestinal stem cells), 27 apoptosis, 28 and bacterial infection. 29 However, the possible role of the STAT signaling pathway in the protective effect of GLP on enterocyte cell damage induced by high-fat components has not been extensively investigated. Recent studies have proposed that different medicines contribute to protection from intestinal injury by a mechanism involving the modulation of the STAT pathway, at least in part via the phosphorylation of STAT3. Using pyruvate-peritoneal dialysis solution, peritoneal resuscitation protects against intestinal injury by inhibiting the STAT pathway in rats with hemorrhagic shock. 30 Li et al 31 demonstrated that the mTOR/STAT3 signaling pathway is activated by lipopolysaccharide (LPS) followed by the induction of apoptosis and inflammation in rat intestine epithelial cells, and echinacoside could alleviate the toxic effect of LPS. Some downstream targets (cyclin E, cyclin-dependent kinase 2, cyclin D1, and c-Myc) of the STAT pathway were confirmed to play important roles in cell cycle distribution and progression in cutaneous T-cell lymphoma HUT78 cells. 32
However, the possible role of STAT has not been previously investigated in the protection of GLP against PA-induced IPEC-J2 cell injury. In the present study, we investigated whether the STAT pathway participates in the suppression of apoptosis and cell cycle arrest-induced GLP in PA-treated IPEC-J2 cells. We observed that PA exposure could induce cell apoptosis and cell cycle arrest through upregulation of STAT1 and STAT2 expression and downregulation of the p-STAT3/STAT3 ratio, while GLP significantly mitigated this effect and protected IPEC-J2 cells. We further evaluated the biological significance of the STAT3 pathway in GLP-induced protection of IPEC-J2 cells from the toxicity of PA. The pharmacological JAK2 inhibitor AG490 was used to selectively deplete STAT3 signaling. Our results indicated that the increase in cell growth triggered by GLP can be inhibited by AG490 treatment of PA-treated IPEC-J2 cells. Pretreatment of IPEC-J2 cells with AG490 blocked the capacity of GLP to reduce cell apoptosis induced by PA. Moreover, AG490 increased the S phase cell cycle arrest of IPEC-J2 cells, as demonstrated by the marked decrease in cells in the G1 and G2 phases in the presence of PA and GLP. These findings suggest that AG490 promoted the population of S phase cells in PA + GLP-treated IPEC-J2 cells and that stimulation of the STAT3 pathway might be attributed to the GLP-mediated protective effect against cell damage caused by PA. The results are similar to those of a previous study conducted by Fuke et al. 33
STAT activation in some instances seems to depend on reactive oxygen species (ROS) signaling, including the accumulation of H2O2, O2−, and OH. 34 ROS are thought to be a second messenger to modulate STAT action. 35 H2O2 is a byproduct of various oxidase and superoxide dismutase reactions and is a key regulator in many oxidative emergency reactions, the reduction of which regulates intracellular ROS levels. 36 Stimulation of the STAT1 and STAT3 pathways is accompanied by regulation of the cell cycle proteins p21 and cyclin D1 in fibroblast cells, resulting in transient cell cycle arrest after H2O2 accumulation. 37 Conversely, STATs promote autophagy, apoptosis, and ROS levels. 38 These studies strongly suggest that STAT and ROS generate a positive ROS-STAT feedback loop, but the details are still not well understood. Ca2+ iron is essential for the induction of cell death and the regulation of many cellular processes and functions. Pathophysiological and pathological conditions can be affected by even slight changes in any of the intracellular Ca2+ signals; thus, they must be strictly regulated in amplitude, space, and time. 39 An increase in Ca2+ concentration can result in phosphorylation of STAT1 in NIH3T3 cells and lymphocytes. Moreover, decreases in Ca2+ flux lead to suppression of STAT1 phosphorylation and a lack of STAT1-dependent gene activation. 40 A recent finding emerged that the transcription factor STAT3 also affects the Ca2+ signaling pathway modulating apoptotic responses. 41 These results indicate that there is a specific interaction among STAT, ROS, and Ca2+. In this study, our data showed that PA disrupted the intracellular Ca2+ and ROS hemostasis and induced increases in intracellular Ca2+ and H2O2 concentrations in IPEC-J2 cells, while GLP reversed the PA influence in a dose-dependent manner, concluding that GLP stimulated the STAT pathway through inhibition of intracellular Ca2+ and H2O2 production, thereby relieving PA-induced IPEC-J2 cell damage.
Nrf2 levels directly or indirectly influence NF-κB, both of which are thought to be relevant to pathological reactions, as they are stimulated by external and internal stimuli that involve oxidative/xenobiotic stress, apoptosis, and various diseases. Numerous studies have proposed that the Nrf2 and NF-κB signaling pathways interact to negatively regulate the release of inflammatory mediators, including chemokines (eg, CCL2 and CCL3) and cytokines (eg, tumor necrosis factor-α, IL-1, and IL-6). 42 Genetic and pharmacological studies have proven that there is signaling crosstalk between NF-κB and Nrf2. Inhibition of NF-κB-induced transcriptional activation via Nrf2-dependent processes is likely to take place in the early phase of inflammation as inferred from NF-κB regulation in proinflammatory synthesis. 43 The link to the STAT signaling pathway should be of great interest to developmental biologists because STAT family members are crucial components of many morphogenetic signaling pathways that directly bind to receptors and other proteins. Recent experimental evidence suggests that the STAT pathway and Nrf2 are connected via STAT1 and STAT3. 44 Our results confirmed that GLP not only inhibited the nuclear translocation of Nrf2 but also increased the nuclear translocation of NF-κB in PA-induced IPEC-J2 cells. Furthermore, inactivation of the STAT3 pathway by AG490 exposure mediated suppression of the NF-κB pathway and activation of the Nrf2 pathway in the presence of GLP and PA treatment. We speculate that GLP activates the STAT pathway, thereby reducing the promotion of Nrf2 nuclear translocation and exacerbating the decrease in NF-κB nuclear translocation mediated by PA. All of these results, directly or indirectly, validated the importance of STATs and the NF-κB and Nrf2 signaling pathways in GLP against PA-induced IPEC-J2 cells. However, there are still some limitations in the research such as whether there is a connection between Nrf2 and NF-κB.
Conclusions
The present study suggests that GLP has protection against PA-induced IPEC-J2 cell injury by promoting cell vitality, counteracting cell apoptosis, and relieving cell cycle arrest in PA-induced IPEC-J2 cells. Moreover, GLP reversed the PA damage by blocking intracellular Ca2+ and H2O2 productions and the nuclear translocation of Nrf2 and NF-κB. These results also show that GLP reversed STAT3-induced NF-κB activation and Nrf2 inactivation in the injured intestines, which may therefore promote proliferation and protection against apoptosis in intestinal epithelial cells. These prove that GLP is an effective agonist of the STAT signaling in intestinal epithelial cells as well as the potential approaches to optimize treatment with high-fat diet-induced intestines disease.
Footnotes
Authors’ contributions
LZ and YS contributed to conceptualization and project administration; YS contributed to supervision; Z-HY and YW contributed to methodology and validation; YW contributed to the investigation; LZ and Z-HY contributed to data curation; LZ contributed to writing—original draft preparation; Z-HY, YS, and J-JG contributed to writing—review and editing; Z-HD and L-LX contributed to visualization. All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was funded by the National Natural Science Foundation of China (grant number 31501484); the National Key Research and Development Project (grant number 2020YFD1001101) the Research and Development Program in Key Areas of Hunan Province (grant number 2019NK2041).
Ethical Approval
The animals were cared for in accordance with the Guide for the Care and Use of Laboratory Animals in China. All experimental procedures were approved by the Animal Care and Use Committee of Hunan Agricultural University (No. 43321503).
Statement of Human and Animal Rights
None.
Informed Consent
None.
