Abstract
In this study, a multistep matrix solid-phase dispersion (MSPD) combining with a high-performance liquid chromatography method was developed for assaying 3 components of different polarities (mannitol, adenosine, and ergosterol) from mushroom samples. MSPD extraction was carried out using 1 g octadecyl-bonded silica as the sorbent material, 9% methanol, 20% methanol, and 100% methanol as eluting solvents for the elution of mannitol, adenosine, and ergosterol, respectively. Mannitol was separated on an NH2P-50 4E column and detected using an evaporative light scattering detector. Adenosine and ergosterol were separated on a Poroshell 120 SB-C18 column and measured at 260 nm and 283 nm, respectively. The developed method showed good linearity (R ≥ 0.9986) within the test range. The relative SD (RSD) of precisions were less than 1.4%, and the recoveries were 95.6%-97.0% (RSD ≤3.0%). Compared with the reported methods, the developed procedure could rapidly prepare components with different polarities (mannitol, adenosine, and ergosterol) from medical mushroom samples with less organic solvent and sample. The method is rapid and eco-friendly, which is helpful to improve the quality evaluation of medicinal mushrooms.
Medicinal mushrooms are an integral part of Chinese Medicine and have been used for health promotion and disease treatment in China for centuries. Modern pharmacological studies revealed that medicinal mushrooms possessed multiple bioactive effects, such as anti-inflammatory, anticancer, and antioxidant activities. 1 -3 These bioactivities are usually related to their complicated components, including hydrophilic components, such as β-glucan, mannitol, and adenosine, and hydrophobic substances, such as sterols. 4,5 Among these, mannitol, adenosine, and ergosterol are often selected as the quality control markers of medicinal mushrooms. 6 Mannitol is used to treat kidney disease and chronic stroke. 7,8 Adenosine, a nucleoside, possesses several pharmacological actions, such as anti-inflammatory, immunomodulatory, and neuroprotective effects. 9,10 Ergosterol is a biological precursor of vitamin D2, which has many bioactivities, including anticancer, anti-inflammatory, and renoprotective functions. 11 -13 Due to the difference in polarity of these 3 components, mannitol and adenosine are usually extracted with a polar solvent and ergosterol with a weakly polar solvent. 6,14 To prepare the test solution of these components, 3 medicinal mushroom samples and 3 extraction procedures were employed, which are laborious and time-consuming. 15 Therefore, developing a rapid and simple extraction method for these 3 components is helpful to improve the quality evaluation of medicinal mushrooms.
It is difficult to simultaneously extract hydrophilic and hydrophobic compounds from the same medicinal mushroom sample by traditional techniques, such as ultrasonic extraction and reflux extraction. Matrix solid-phase dispersion (MSPD) is a fast and simple extraction method, which has been successfully applied in Chinese medicine sample extraction. 16 -18 The MSPD extraction process often contains the following steps. First, the sample is disrupted and dispersed with dispersant, and then the analytes are eluted by the solvent. By using MSPD, the target analytes could be extracted and purified based on the different elution solvents. Therefore, MSPD is a feasible method for extracting components with different polarities from medicinal mushroom samples by multistep elution with different solvents. Nowadays, MSPD is mostly applied in sample extraction and purification, such as extracting flavonoids from Astragali radix and sterols from Chinese Cordyceps. 19,20 However, there is no report about extracting analytes with different polarities by multistep MSDP.
In the current study, a multistep MSPD combined with a high-performance liquid chromatography (HPLC) method was developed for the analysis of components with different polarities in medicinal mushroom samples. Mannitol, adenosine, and ergosterol were extracted from medicinal mushroom samples with different solvents and determined by HPLC.
Results and Discussion
Method Validation
The validation of the current method is summarized in Tables 1 and 2 and Supplemental Table S1. The current method showed good linearity in the tested range with correlation coefficients above 0.998. The limit of quantification (LOQs) of mannitol, adenosine, and ergosterol were 59.76, 0.16, and 0.28 µg/mL, respectively. The relative SDs (RSDs) of intraday and interday precision were less than 0.9% and 1.4%. The recoveries of analytes were 95.6%-97.0% (RSD ≤2.9%). The RSDs of stability validation were less than 0.9%.
Linear Regression Equation, Linear Ranges and LOQ.

Abbreviation: LOQ, limit of quantification.
Precision, Stability, and Recovery.
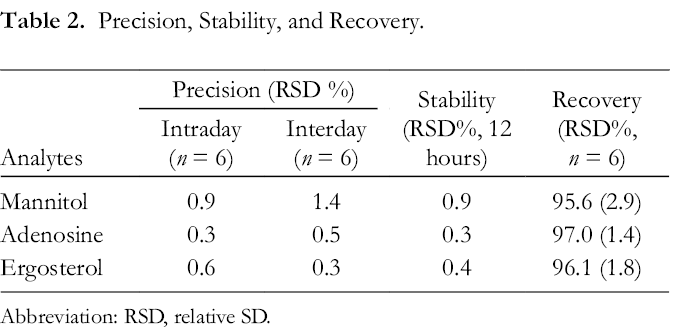
Abbreviation: RSD, relative SD.
Analysis of Medicinal Mushroom Samples
The developed method was successfully applied to 4 medicinal mushroom samples. The chromatograms of reference components and medicinal mushroom samples were shown in Supplemental Figures S1-S3, and the contents of mannitol (32.4-102 mg/g), adenosine (0.22-2.25 mg/g), and ergosterol (1.92-4.45 mg/g) in the samples are listed in Table 3.
The Content of Analytes in Samples.
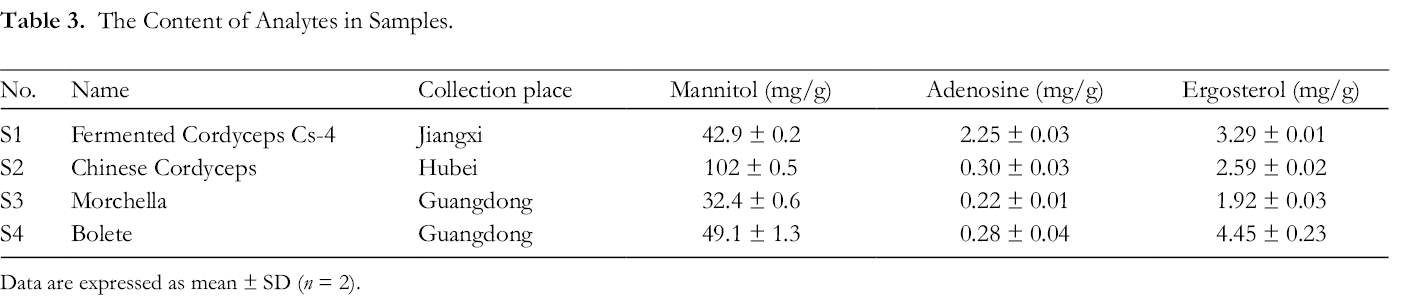
Data are expressed as mean ± SD (n = 2).
Optimization of Multistep MSPD Extraction
Sample extraction was considered time-consuming and organic solvent-consuming but a critical step in the Chinese medicine analysis process. 21 In order to explore a fast sample treatment method with low organic solvent cost, numerous efforts have been devoted. In the present study, multistep MSPD extraction of mannitol, adenosine, and ergosterol in the samples was performed. The illustration of the MSPD extraction procedure is shown in Figure 1. The conditions of MSPD, such as the ratio of dispersant to sample, and the polarity and volume of eluting solvent were optimized with a step-by-step approach. The fermented Cordyceps Cs-4 sample was used for the following tests.

Schematic diagram of multistep matrix solid-phase dispersion extraction.
First, 3 ratios of sample to dispersant (1:5, 1:10, 1:20) were compared according to the literature. 18,20 The results showed that there was no obvious difference among the content of the analyte obtained by the different ratios. For the purpose of saving materials, the ratio of 1:5 was used for sample extraction.
Second, the amounts of octadecyl-bonded silica (ODS) (1.0 g, 1.5 g, 2.0 g) were investigated, referring to the literature. 19 It was found that 1.0 g and 1.5 g ODS displayed good extraction and separation efficiency of the analytes with the same volume of elution solvent, while 2.0 g ODS needed more organic solvent to elute the analytes. Therefore, 1.0 g ODS was chosen for the MSPD because less ODS and organic solvent were used in this condition.
Third, elution solutions with different polarities were tested for the extraction and purification of analytes from the sample. Due to the different polarity of the analytes, 4 fractions were designed, the mannitol fraction, adenosine fraction, weakly polar impurities fraction, and ergosterol fraction. Mannitol, a polar compound, is often not retained on an ODS column. 22 Hence, 6 low concentration of methanol aqueous solutions (5%, 7%, 9%, 11%, 13%, and 15%) were tested for the mannitol fraction. The results showed that mannitol could be eluted by these 6 methanol solutions (Supplemental Figure S4(A)). Adenosine would be lost in the mannitol extraction in the case of eluting with 11%, 13%, or 15% methanol solution (Supplemental Figure S4(B)), and some polar impurities would be retained in the next adenosine fraction in the case of eluting with 5% or 7% methanol solution (Supplemental Figure S4(C)). Therefore, 9% methanol solution was used for mannitol extraction based on the good extraction of mannitol and with less impurities in the next adenosine fraction. Subsequently, 4 methanol aqueous solutions (15%, 20%, 25%, and 30%) were tested for adenosine extraction according to the literature in which adenosine was eluted with 15% methanol solution on an ODS column. 9 The results showed that 20%, 25%, and 30% methanol solution could elute adenosine rapidly and with less elution solvent than 15% methanol (Supplemental Figure S5). The 20% methanol solution with the minimum consumption of organic solvent was used for the experiment. In the same way, the different methanol solutions (88% to 98%) were tested for removing the weak polar impurities; the high ratio of methanol solution was good at removing the impurities. However, the 98% methanol solution eluted ergosterol and the impurities together (Supplemental Figure S6). Thus, 96% methanol solution was used for removing the weak polar impurities. Finally, ergosterol was extracted and eluted with 100% methanol, since it was usually eluted with 100% organic solvent on an ODS column in the literature. 12
Fourth, the different volumes of elution solution were compared. The elution solution was collected into tubes, each of which contained 1 sorbent bed volume (1.5 mL) of elution solution. The results (Supplemental Figure S7) indicated that 6 sorbent bed volumes (9 mL) of 9% methanol were sufficient to elute mannitol and remove the polar impurities; 3 sorbent bed volumes (4.5 mL) of 20% methanol could fully elute the adenosine, 6 sorbent bed volumes (9 mL) of 96% methanol could remove the weak polar impurities, and 6 sorbent bed volumes (9 mL) of methanol could completely elute the ergosterol.
In summary, the optimal conditions were as follows. The ratio of sample to dispersant was 1:5, and 1 g ODS was used for the extraction and purification. The sample mixture was successively eluted with 9 mL of 9% methanol solution (mannitol test solution), 4.5 mL of 20% methanol solution (adenosine test solution), 9 mL of 96% methanol solution (weak polar impurities solution), and 9 mL of 100% methanol (ergosterol test solution).
Optimization of HPLC Conditions
Mannitol is a polar compound without ultraviolet (UV) absorption, which is difficult to be analyzed by reversed-phase (RP) column separation and UV detection. According to a previously reported HPLC method for mannitol, 23 a hydrophilic interaction chromatography column (HILIC) separation coupled with evaporative light scattering detector (ELSD) was used for the HPLC analysis of mannitol. Adenosine and ergosterol from the Chinese medicinal sample were separated on a long RP column (150 or 250 mm) because of the complicated components in the sample solution, which needed more than 30 minutes. 24,25 In the current experiment, the adenosine and ergosterol sample solutions were purified by MSPD, which are easily separated on an RP column. In order to shorten the analytical time and save organic solvent, a short Poroshell RP column (30 mm) was used, which could provide rapid and efficient separation on conventional HPLC systems. 26 The detection wavelengths of adenosine and ergosterol were set at 260 nm and 283 nm based on the UV absorption characteristics of the compounds.
Comparison of Current and Reported Methods
In order to assess further the performance of the current procedure, a comparison of results between the current method, that of the Chinese Pharmacopeia (ChP) (adenosine and ergosterol), and a literature method (mannitol) was carried out using fermented Cordyceps Cs-4. 6,23 The average contents (n = 5) of mannitol, adenosine, and ergosterol in sample S1 were 42.5 ± 0.4 mg/g, 2.25 ± 0.03 mg/g, and 3.27 ± 0.02 mg/g, respectively, detected by the current method, and 44.4 ± 0.5 mg/g, 2.34 ± 0.03 mg/g, and 3.65 ± 0.07 mg/g, respectively, by the reported methods. These results showed that the data for the current method complied with that of the ChP and literature methods. In addition, the content of components obtained by the current method was similar to previously reported data. For example, the content of mannitol in Chinese Cordyceps was 102 mg/g, which is within the range of the reported data (90.4-141.4 mg/g). 14 The content of adenosine was 0.30 mg/g, within the range of the reported data of 0.14‐1.10 mg/g, 24 and the ergosterol content of bolete was 4.45 mg/g, which complied with the reported data of 3.38‐9.10 mg/g. 27 Therefore, the current developed multistep MSDP coupled with the HPLC method could be used for the determination of mannitol, adenosine, and ergosterol in medicinal mushroom samples.
The sample extraction and HPLC separation of the current method were compared with those reported. The sample extraction comparison information is summarized in Table 4. In the ChP and reported sample extraction methods, the extraction time of each analyte was more than 30 minutes, sample consumptions were 0.5‐1.0 g, and solvent consumptions were 30‐100 mL. In order to obtain these 3 analyte sample solutions, 180 mL of extraction solvent and 2.5 g of sample were consumed. In addition, 3 extractions took 210.0 minutes. In the current study, multistep MSPD was used for sample extraction, and only 0.02 g of sample, 31.5 mL of extraction solvent, and 30.0 minutes were used. Furthermore, the analyte sample solutions prepared by the ChP and literature methods were complicated and contained many impurities, causing the need for a long chromatography column for the separation of the analytes. The chromatograms of the current and reported methods are shown in Supplemental Figure S8, and the related data are listed in Table 4. The HPLC method for mannitol in the current experiment was the same as that in the literature. The HPLC separations of adenosine and ergosterol took 28 minutes by the ChP methods. By contrast, the adenosine and ergosterol sample solutions obtained from the multistep MSPD extraction were purified and separated on a short Poroshell column. The HPLC separation time for the adenosine and ergosterol sample solutions was less than 2 minutes. In summary, 2.5 g of samples, 261 mL of solvents, and a whole running time of 291 minutes were employed in the reported methods for the determination of mannitol, adenosine, and ergosterol in medicinal mushroom samples, including sample extraction and separation. However, only 0.02 g of sample, 60.5 mL of solvents, and 59.0 minutes were required for the current method. Therefore, the current method, which is rapid and requiring less solvent and sample, is an improvement for the determination of components with a large span of polarity in Chinese medicine.
Comparison of Current Method and Reported Method.

Materials and Methods
Chemicals and Material
Ergosterol (96.2%), adenosine (99.7%), and mannitol (99.1%) were purchased from National Institutes for Food and Drug Control (Beijing, China). HPLC grade ethanol from Fisher (MI, USA). HPLC grade methanol and acetonitrile from Spectrum Chemical Co., Ltd (CA, USA), and analytical grade methanol and ethanol from Xilong Scientific Co., Ltd (Sichuan, China). The deionized water was purified by a Milli-Q purification system (MA, USA). Diatomaceous earth was supplied by Sigma (Merck, Germany), and ODS by Shenzhen Kemis Technology Co., Ltd (Guangdong, China). MSPD tubes and sieve plates were purchased from Agilent Technologies (CA, USA).
Fermented Cordyceps Cs-4 (Paecilomyces hepiali) was purchased from Jiangxi province. Chinese Cordyceps (Cordyceps sinensis) was collected from Hubei province. Morchella (Morchella sextelata) and Bolete (Phlebopus portentosus) were collected from Guangdong province. The botanical origin of materials was identified by Dr Zhengming Qian. Voucher specimens were deposited at the Dongguan HEC Cordyceps R&D Co., Ltd, Dongguan, China.
Preparation of Reference Compound Solutions
Ergosterol stock solution was prepared with methanol and further diluted with methanol to obtain a series of working solutions. 11 Adenosine and mannitol stock solutions were prepared with water, and a series of individual standard working solutions were prepared by diluting the stock solutions with water. 23,24
Multistep MSPD Extraction Condition
The MSPD tube with a sieve plate at the bottom was filled with 1.00 g ODS. The MSPD tube was eluted with 9% methanol solution before extraction. Medicinal mushroom samples were pulverized and sieved through a 110 mesh sieve. One gram of sample and 5 g of diatomaceous earth were placed in a mortar and blended for 3 minutes to obtain a homogeneous mixture. Subsequently, the mixture (approximately 0.10 g) was accurately weighed and loaded onto an MSPD tube. The sample mixture was eluted with 9 mL of 9% methanol solution, 4.5 mL of 20% methanol solution, 9 mL of 96% methanol solution, and 9 mL of 100% methanol. The targeted analyte elution solutions were collected in a volumetric flask and adjusted to 5.0 mL (adenosine test solution) and 10.0 mL (mannitol test solution and ergosterol test solution). The sample solutions were filtered through a 0.45-µm membrane before HPLC analysis.
HPLC Condition
An Agilent Series 1260 LC system (Agilent Technologies, USA) was used, consisting of a quaternary pump, an autosampler, a thermostatic column compartment, a diode array detector (DAD), and an ELSD.
The HPLC conditions for mannitol were: a Shodex Asahipak NH2P-50 4E column (250 × 4.6 mm, 5 µm) was used, the column temperature was 30 °C, the mobile phase 82.5% acetonitrile with isocratic elution, and the flow rate 1 mL/min. The ELSD conditions were as follows: temperature 40 °C, nitrogen pressure 3.5 bar.
The HPLC conditions for adenosine were: an Agilent poroshell 120 SB-C18 column (4.6 mm × 30 mm, 2.7 µm) was used. The chromatographic analysis was executed in isocratic mode using 20% methanol at a flow rate of 1 mL/min. The column temperature was 25 °C, and the detection wavelength was 260 nm.
The HPLC conditions for ergosterol were: an Agilent poroshell 120 SB-C18 column (4.6 mm × 30 mm, 2.7 µm) was used. The chromatographic analysis was executed in isocratic mode using 94% ethanol at a flow rate of 1 mL/min. The column temperature was 35 °C, and the detection wavelength was 283 nm.
Method Validation
The current methods were validated for linearity, LOQ, precision, repeatability, stability, and recovery.
Linearity and LOQ
A series of concentrations in the range of 59.76‐258.95 µg/mL (mannitol), 0.84‐13.43 µg/mL (adenosine), and 4.21‐25.28 µg/mL (ergosterol) of reference component solutions were prepared for evaluation of linearity. The standard curves for adenosine and ergosterol were constructed by plotting the concentrations versus peak areas, and the standard curve of mannitol was plotted as the log 10 of calibration concentrations versus the log 10 of peak areas.
LOQs were determined by injection of reference components and calculated as analyte concentrations giving signal-to-noise (S/N) ratios of approximately 10.
Precision and repeatability and stability
The precision of the current method was determined by intraday and interday evaluations. The intraday precision was assessed by analyzing the reference component solutions 6 times within 1 day. The interday precision was evaluated by injecting the reference component solutions twice per day for 3 consecutive days.
To determine the repeatability of the current method, the MSPD extraction of the sample was performed with 6 replicates. The sample solution stability test was performed within 12 hours.
Recovery
To assess the accuracy of the method, accurate amounts of mannitol, adenosine, and ergosterol were added to a fermented Cordyceps Cs-4 sample. The mixture was then extracted by multistep MSPD and analyzed by HPLC (n = 6). The recovery rate was calculated by the following equation:
Recovery% = 100 × (found amount − original amount)/spiked amount
The relative SDs (RSD) were used to evaluate the precision, repeatability, stability, and recovery of the developed method.
Conclusions
In this study, a multistep MSPD coupled with the HPLC method was developed for the determination of mannitol, adenosine, and ergosterol in 4 medicinal mushrooms. The developed method is accurate and reliable, which is suitable for assaying the analytes with different polarities in medicinal mushroom samples. Compared with the reported method, the developed procedure is rapid, solvent saving, and low sample consumption. It could be used as an improved analytical method for the determination of mannitol, adenosine, and ergosterol in medicinal mushrooms.
Supplemental Material
Figure S1 - Supplemental material for Rapid Determination of 3 Components With Different Polarities in Medicinal Mushrooms by Multistep Matrix Solid-Phase Dispersion and High-Performance Liquid Chromatography Analysis
Supplemental material, Figure S1, for Rapid Determination of 3 Components With Different Polarities in Medicinal Mushrooms by Multistep Matrix Solid-Phase Dispersion and High-Performance Liquid Chromatography Analysis by Zhengming Qian, Zi Wu, Chunhong Li, ChangLiang Yao, Guoying Tan, Wenjia Li and De-an Guo in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Key-Area Research and Development Program of Guangdong Province (grant number 2020B1111110007) and the Science and Technology Development Fund of Macao Special Administrative Region (grant number 125/2017/A3).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
