Abstract
High-performance liquid chromatographic-tandem mass spectrometric (HPLC-MS-MS) methods were developed for the analysis of 13 bioactive constituents of Zizyphi Spinosae Semen (ZSS), and for samples of ZSS fried at different temperatures and from different production areas. The results showed that the content of bioactive constituents increased with the increase in frying time from 65 to 80 min at 150 °C; among the 14 samples of ZSS from different production areas, those from Shanxi Changzhi, Shandong Jining, and Hebei Xingtai had a higher content of bioactive constituents. This analytical method can accurately and quickly analyze the bioactive constituents of ZSS and provide a scientific basis for its quality control.
Keywords
Introduction
Zizyphi Spinosae Semen (ZSS) are mature seeds of Ziziphus jujuba Mill. var. spinosa (Bunge) Hu ex H.F. Chow (Rhamnaceae). They have been used for the treatment of insomnia and anxiety. 1 The seeds of ZSS are white or light yellow and rich in oils. They have a slight odor and light taste. 2 The seeds are rich in saponins, flavonoids, and alkaloids, as well as other pharmacologically active constituents with antioxidant functions. They have been mainly used for sedation and hypnosis.3–10 Numerous studies have also reported other pharmacological effects of ZSS, including cardiovascular system protection, regulation of immune function, and antioxidant effects.11–13
Previous studies of the medicinal components of ZSS have been performed using either crude components or single compounds. Studies performed on pentobarbital mouse models have demonstrated that ZSS could shorten sleep latency and have a significant effect on deep sleep. 14 Tests by Poindexter et al 15 showed that high concentrations of saponins have toxic effects on neonatal rat cardiomyocytes, but not on adult rat cardiomyocytes, and can have cardiotonic effects. Due to stress in the environment, plants produce compounds that can moderately activate cellular stress response mechanisms at sub-toxic levels in humans, which may enhance tolerance to severe dysfunction or disease. 16 In addition, the sedative and hypnotic effects of saponins and flavonoids from ZSS have been shown to reduce the movement of mice and increase sleep time. 17 Cao et al and others have demonstrated that saponins can prolong the sleep time of normal rats and enhance the hypnotic activity of pentobarbital. 18 Furthermore, several studies have shown that the flavonoid, spinosin, may act as a post-synaptic 5-HT 1A receptor antagonist to reduce the effects of the agonist 8-OH-DPAT to prolong non-rapid eye movement sleep, rapid eye movement sleep, and total sleep time.19–21 Jujube seed alkaloids constitute apofen and cyclic peptide alkaloids. Studies have shown that these alkaloid components could shorten pentobarbital-induced sleep episodes and prolong sleep time, while the total alkaloids could significantly reduce the exercise ability observed in mouse models.22–24 ZSS, a traditional Chinese medicine, has been shown to calm the nerves and improve sleep function. These findings provide abundant modern pharmacological evidence for its function.
We investigated methods of cooking and sautéing to determine whether they affected the bioactive constituents of raw and fried ZSS. The efficacy was then evaluated using raw and fried ZSS. Previous studies have evaluated raw and fried ZSS, but, in this study, we also evaluated the bioactive constituents of ZSS using different processing methods and times. Dynamic changes in the bioactive constituents during the processing steps were observed. These results could explain the changes in the composition of the bioactive components when processing ZSS.
We also observed that the bioactive constituents of ZSS from various production areas were different. To determine the quality of ZSS from the various areas, the composition of ZSS from 14 production areas was measured using high performance liquid chromatography-evaporative light scattering detection (HPLC-ELSD) and HPLC-MS-MS. By measuring the total effective components, the highest content for the bioactive constituents was determined. This study provides the basis for the screening of high-quality ZSS medicinal material and the development of functional ZSS products.
Experimental
Instrumentation
Quantitative analysis of ZSS was performed using HPLC-MS-MS on an Agilent 1200 HPLC series and 6410 triple quad MS (USA). The HPLC system consisted of a binary pump, a degasser, a thermostat column compartment, and an automatic sampler. The ion source was electrospray ionization (ESI) and was used in the positive mode with a nebulizer pressure of 15.0 psi, a capillary voltage of 4000 V, and a capillary temperature of 300 °C. The mass spectra were recorded and analyzed using Agilent Masshunter workstation acquisition software. Sample analysis was performed on a Prorshell 120 EC-C18 column (50 × 2.1 mm, 4 μm) at a temperature of 30 °C. The mobile phase consisted of a 0.1% formic acid aqueous solution at a flow rate of 0.2 mL/min. All samples were filtered into the injection bottle with an organic filter membrane with an aperture of 0.22 μm. The volume before HPLC analysis was adjusted with methanol. Multiple reaction monitoring (MRM) was selected as the scan mode for quantitative analysis for triple quad MS.
Chemicals and Materials
Standards for puerarin, magnoflorine, spinosin, 6‴-feruloylspinosin, jujuboside A, jujuboside A1, jujuboside B, jujuboside B1, nuciferine, lysicamine, saponarin, isovitexin, and swertisin (HPLC≥ 98%) were purchased from Shun Bo Bioengineering Co. Ltd (Shanghai, China). HPLC-grade methanol was obtained from Duksan Platech Co. Ltd (Korea), and ZSS from Medicinal Materials Market in Anguo Yanqing, Beijing, Xingtai, Hebei, Zanhuang, Hebei, Zhangjiakou, Hebei, Jiaozuo, Henan, Chaoyang, Liaoning, Jining, Shandong, Pingyi, Shandong, Taian, Shandong, Lvliang, Shanxi, Changzhi, Shanxi, Qinling, Shanxi, Shangluo, Shanxi, Kashi, and Xinjiang. Ultra-pure water was prepared using a Millipore-Q purification system (USA). Analytical grade ethanol was purchased from Beijing Chemical Works Co. Ltd (Beijing, China), and 30 to 60 °C boiling point light petroleum from Shanghai Macklin Biochemical Co. Ltd (Shanghai, China). Reagent-grade light petroleum and ethanol were used for ZSS extraction.
Preparation of Fried ZSS
An induction cooker was set on the cooking mode, with the power set at 1300 W. ZSS (500 g) was fried in a preheated iron pan at 150 °C. In the ancient and modern concoction industry, there has always been a theory of “frying every son must be fried,” in which frying is mostly based on a slow fire, with the temperature mostly controlled at 130 to 200 °C, and the degree of frying is mostly based on the observation of the properties and odor of the herbs. Taking into account the experimental operation and production reality, the frying temperature was controlled at 150 °C in this experiment. At 0, 5, 30,40, 50, 65, 80, 95, and 130 min, 50 g of ZSS was removed for extraction.
Extract Preparation
ZSS extraction was performed as follows: 50.00 g dried powder of ZSS (which was passed through a 60-mesh sieve) was wrapped in filter paper and then extracted in a Soxhlet apparatus at 60 °C with 200 mL of light petroleum (30-60 °C boiling point). After 6 h, the defatted ZSS powder was placed in a fume hood until completely air-dried. Then, 20.00 g of ZSS dry residue was extracted using a solvent reflux method. The optimized extraction conditions were as follows: ethanol volume fraction was 50%, the material–liquid ratio was dried powder-50% ethanol (1:20, m/v), extraction temperature 80 °C, and an extraction time of 3 h. The solution was centrifuged at 10 000 r/min for 10 min, the supernatant collected, filtered, the filtrate rotary evaporated at 40 to 50 °C, and then freeze-dried.
Preparation of Standard and Sample Solutions
Puerarin, nuciferine, spinosin, 6-feruloylspinosin, magnoflorine, jujuboside A, jujuboside A1, jujuboside B, jujuboside B1, lysicamine, saponarin, isovitexin, and swertisin were weighed (0.003 g of each) and dissolved in methanol. Magnoflorine, nuciferine, lysicamine, jujuboside A, jujuboside A1, jujuboside B, and jujuboside B1 were formulated into solutions at concentrations of 10, 8, 6, 5, 4, 2, and 1 μg/mL. Puerarin, spinosin, 6‴-feruloylspinosin saponarin, isovitexin, and swertisin were formulated into solutions at concentrations of 100 50, 25, 10, 5, 2, and 1 μg/mL. Extract of 0.01 g of ZSS from 14 producing areas was ultra-sonicated with 1 mL MeOH. All solutions were stored at 4 °C until required.
Calculation of Bioactive Constituents in ZSS
HPLC-MS-MS was used to determine the peak area of puerarin, magnoflorine, spinosin, 6‴-feruloylspinosin, jujuboside A, jujuboside A1, jujuboside B, jujuboside B1, nuciferine, lysicamine, saponarin, isovitexin, swertisin, and N-nornuciferine in the extracts. The content of each bioactive constituent was calculated using the following formula:
Statistical Analysis
Statistical analyses were performed using SPSS 12.0.
Results and Discussion
Optimization of HPLC-MS-MS Parameters
MRM in positive mode with ESI was used for improved specificity and sensitivity. Collision energies (modifying their value from 1 to 100 eV) were optimized for each analyte (listed in Table 1). These parameters were optimized to acquire higher intensity.
MS Parameters of the Thirteen Compounds.
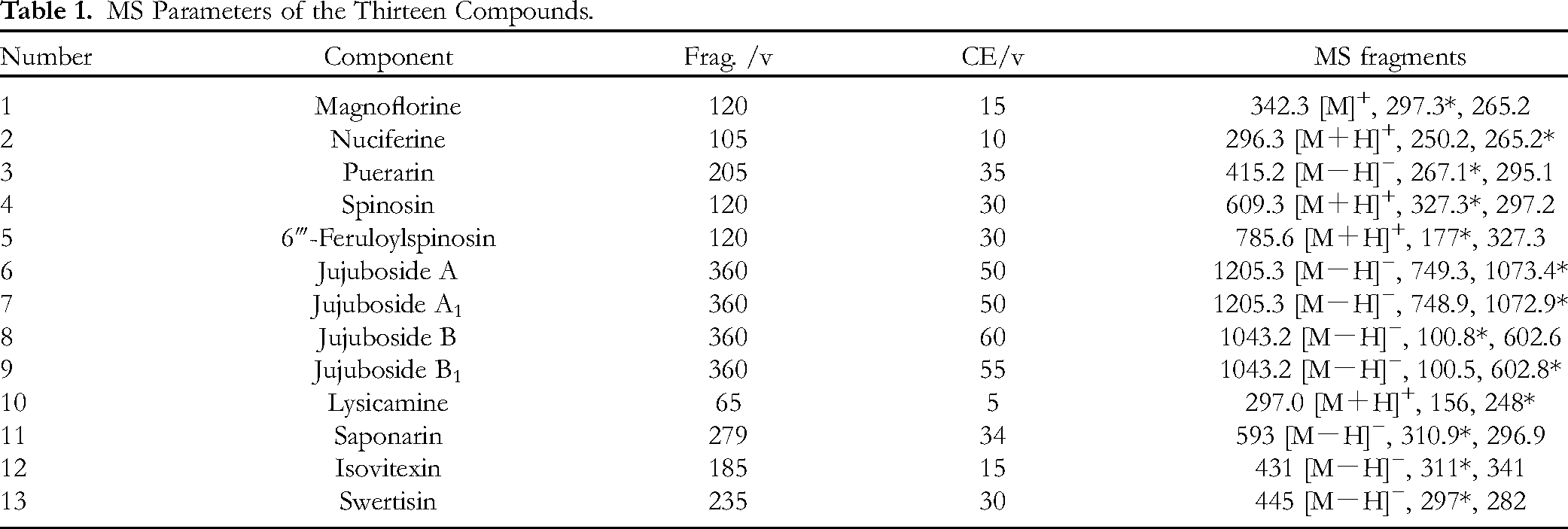
Formic acid was used as a mobile phase modifier to significantly reduce peak tailing and improve peak shape. It was easier to clean and use in the MS detector as it is volatile and leaves no residues. In this study, 0.05% and 0.1% aqueous formic solutions, 0.1% triethylamine solution, 0.05% and 0.1% acetic acid solution, and an aqueous solution without formic acid in the mobile phase were evaluated. To obtain the optimal elution conditions for the separation and determination of the bioactive constituents, various linear gradients of 0.05% aqueous formic acid solution were evaluated. Finally, gradient elution was optimized as follows: 0 to 10 min 20% B to 56% B, 10 to 14 min 56% B to 59% B, 14 to 20 min 59% B to 75% B, 20 to 25 min 75% B to 96% B, 25 to 30 min 96% B to 100% B, and 30 to 35 min 100% B.
Method Validation
Linearity, LOD, and LOQ
Standard curves were generated using 7 different concentrations and 3 injections were used for the HPLC-MS-MS conditions (section “Optimization of HPLC-MS-MS Parameters”). In addition, samples were also evaluated. A good linear relationship for the 13 bioactive constituents was obtained with correlation coefficients between 0.993 and 0.9999 (Table 2). The detection limits (LOD) and quantification limits (LOQ) were estimated at a signal-to-noise (S/N) ratio of 3 and 10, respectively, with values in the range from 0.01 to 0.75 μg/mL and 0.03 to 2.5 μg/mL.
Linear Ranges, Correlation Coefficients, LOD and LOQ of the 13 Bioactive Constituents.

Repeatability and Recovery
Three consecutive injections of a mixture of ZSS samples were performed to evaluate the precision. The contents of puerarin, nuciferine, and lysicamine in ZSS were lower than the detection limit. Results were expressed as relative standard deviations (RSD). RSD values ranged from 0.8% to 1.93% (Table 2) for the tests and were lower than 10%, indicating good precision. Recovery tests were performed using 14 samples that were in the range of the calibration curve (three replicates each). Recoveries ranged from 96.9 to 98.9 (Table 2), with the recovery rates of the standards meeting the detection requirements.
Identification of the Constituents of the ZSS Extracts
HPLC-MS-MS was used to identify magnoline, spinosin, 6‴-feruloyl-spinosin, jujuboside A, jujuboside A1, jujuboside B, jujuboside B1, puerarin, nuciferine, lysicamine, saponarin, isovitexin, and swertisin in the ZSS extracts. Flavonoids, saponins, and alkaloids were identified. The analysis method for the bioactive constituents in ZSS extracts provided a fast and effective procedure for their identification and quantitation. Figure 1 shows the chromatograms of samples from the 14 producing areas, that is, Yanqing, Beijing (BY), Xingtai, Hebei (HX), Zanhuang, Hebei (HZ), Zhangjiakou, Hebei (HZh), Jiaozuo, Henan (HJ), Chaoyang, Liaoning (LC), Jining, Shandong (SJ), Pingyi, Shandong (SP), Taian, Shandong (ST), Lvliang, Shanxi (SL), Changzhi, Shanxi (SC), Qinling, Shanxi (SQ), Shangluo, Shanxi (SS), Kashi, and Xinjiang (XK). Puerarin, nuciferine, and lysicamine were not detected due to their low abundance.
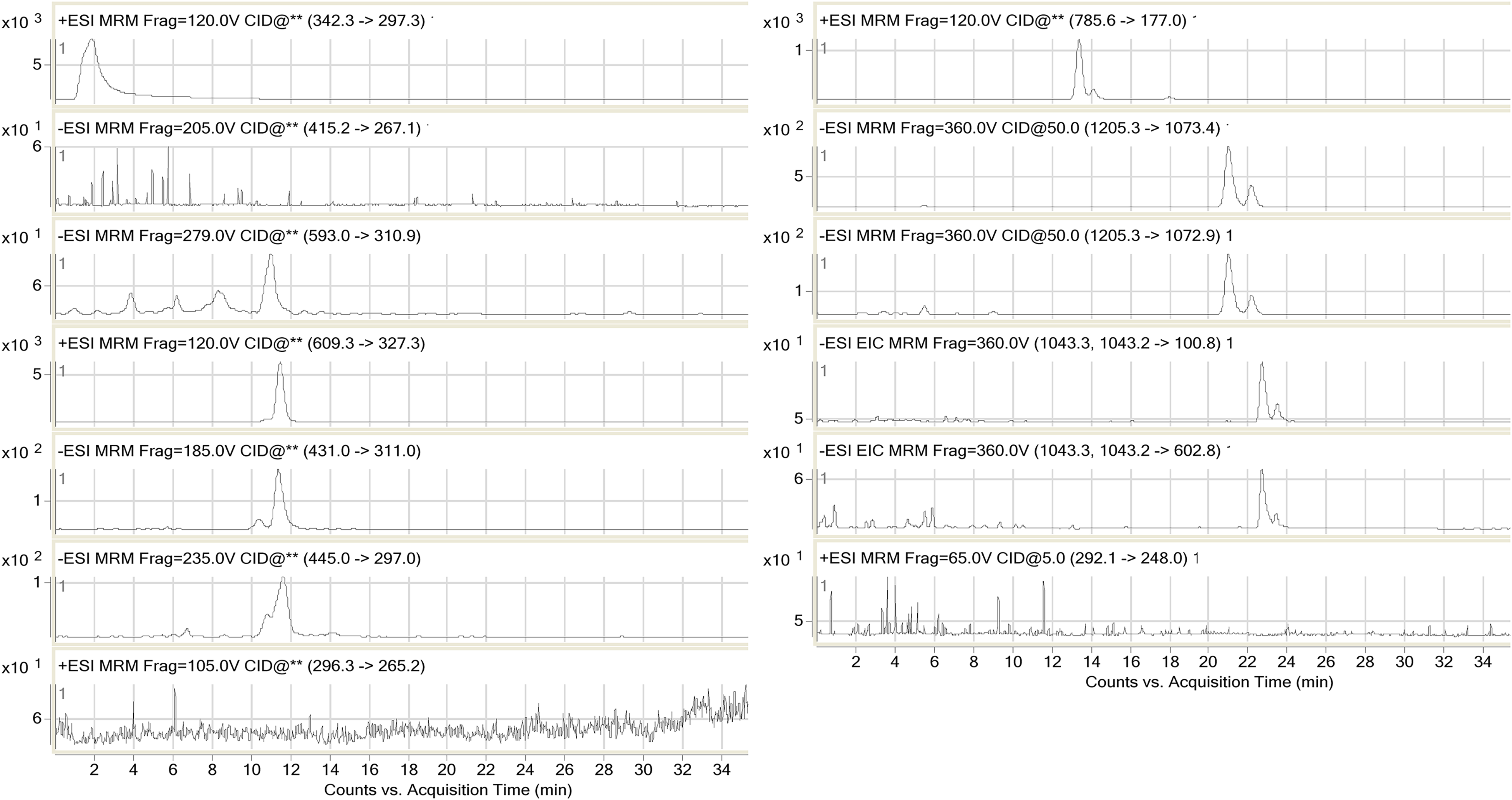
Multiple reaction monitoring (MRM) chromatograms of the samples of the ZSS bioactive constituents.
Fried State of ZSS
ZSS was gradually cooked from 0 to 50 min of frying. After fried for 65 min, ZSS started to produce an oil, and its surface had an oily appearance, accompanied by a slight aroma. ZSS contains a large amount of fatty and volatile oils, and the fatty oil will separate out after heating, and the volatile oil will evaporate and produce an aroma. After 80 min, ZSS was shiny and started to become thick, and after 130 min of frying, it was charred.
The changes in bioactive constituents in ZSS at different stir-fried levels are shown in Figure 2.

Bioactive constituent levels in ZSS at the different time points (A: content of flavonoids in ZSS with different degrees of ripeness; B: content of saponins in ZSS with different degrees of ripeness; C: content of alkaloids in ZSS with different degrees of ripeness).
Our results showed that the levels of total jujubosides, total flavonoids, and magnoflorine in ZSS decreased in sequence during the 0 to 50 min of cooking. Jujubosides and flavonoids are easily soluble in water and ethanol aqueous solutions. The alkaloids magnoflorine and sanjoinine A are both water-soluble compounds. ZSS began to produce oil after 65 min of frying. After 80 min, the surface of ZSS appeared shiny. At this time, the levels of bioactive constituents in ZSS increased, likely because the oil was favorable to the bioactive constituents of ZSS. After dissolution, the levels of the bioactive constituents obtained after subsequent solvent reflux extraction also increased accordingly. ZSS became pasty after 95 min of frying, and after 130 min, it was charred. During this period, the levels of the various bioactive constituents in the ZSS extracts were reduced, presumably due to prolonged high temperatures. Because the bioactive constituents in the ZSS were reduced, the levels were also reduced in the extracts.
Analysis of Bioactive Constituents in ZSS From the 14 Producing Areas
The content of bioactive constituents in ZSS from the 14 different producing areas is shown in Figure 3 and Table 3. Puerarin, spinoretin, 6‴-feruloyl spinosin, isovitexin, and swertisin are flavonol glycosides; jujuboside A, jujuboside A1, jujuboside B, and jujuboside B1 are saponins; and magnoline and nuciferine are alkaloids. The level of flavonol glycosides from Xingtai Hebei was higher compared to the other producing areas. The producing area with the highest content of saponins was Jining Shandong. The amounts of alkaloids from Jining Shandong were higher compared to the other producing areas. The producing area with the highest total bioactive constituents was Jining Shandong, while the lowest was Kashi Xinjiang. It was previously demonstrated that ZSS flavonoids could improve the learning and memory ability of mice. 23 ZSS from the Zanhuang area of Hebei has been used as a traditional Chinese medicine to improve memory. The alkaloids from ZSS have been shown to possess anti-convulsion properties 24 and have been considered an anti-epileptic drug in Chinese medicine. Wang et al 25 demonstrated that saponins could significantly increase the expression levels of GABA receptor subtype mRNA in hippocampal neurons to improve sleep. The levels of total saponins in ZSS were the highest in the Shandong Jining production area and could be suitable for the development of sleep-improving medication.
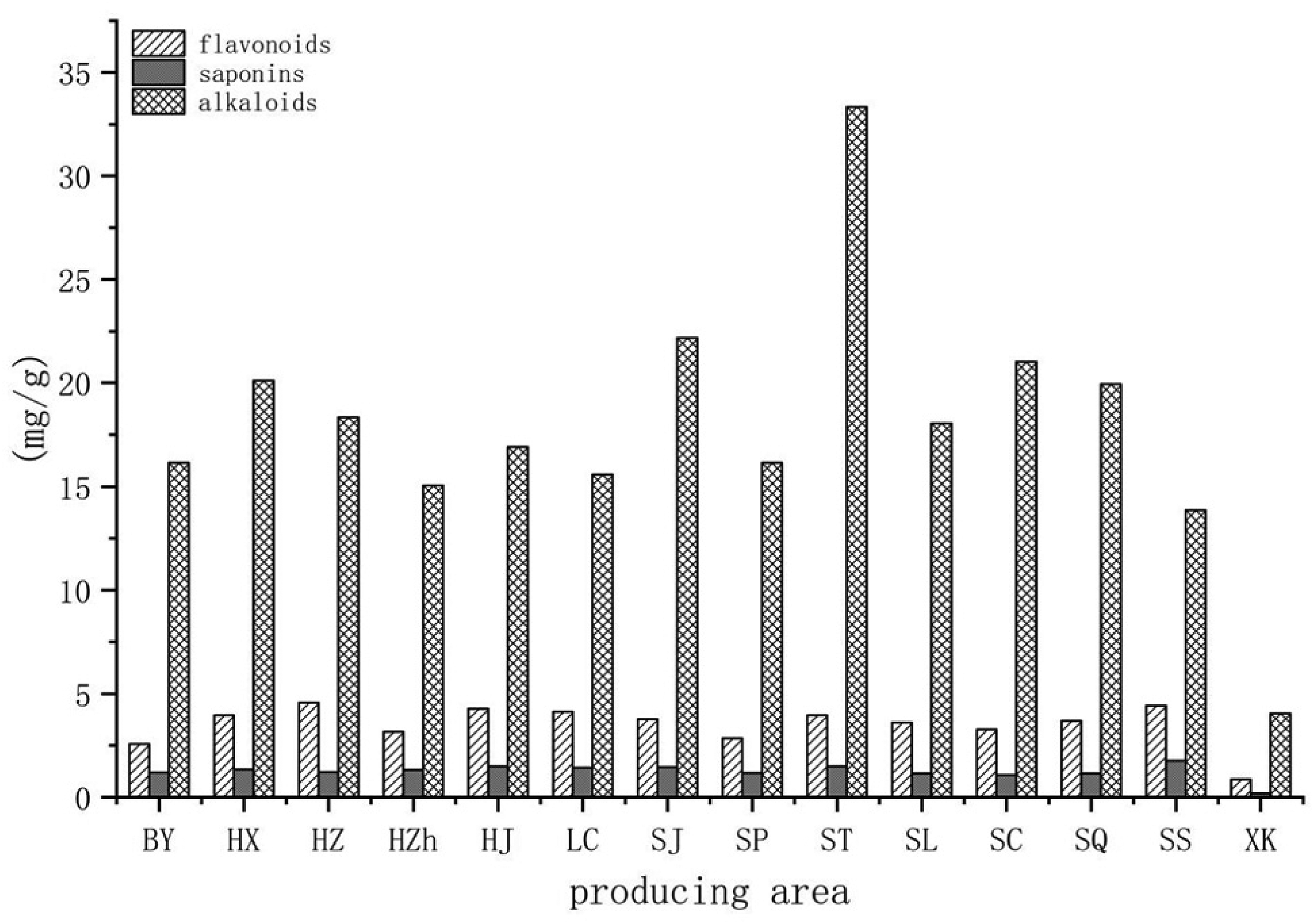
Bioactive constituents in ZSS from the 14 producing areas. Yanqing, Beijing (BY), Xingtai, Hebei (HX), Zanhuang, Hebei (HZ), Zhangjiakou, Hebei (HZh), Jiaozuo, Henan (HJ), Chaoyang, Liaoning (LC), Jining, Shandong (SJ), Pingyi, Shandong (SP), Taian, Shandong (ST), Lvliang, Shanxi (SL), Changzhi, Shanxi (SC), Qinling, Shanxi (SQ), Shangluo, Shanxi (SS), and Kashi, Xinjiang (XK).
Bioactive Constituents in ZSS From the 14 Producing Areas.

a-j represent significant differences.
Conclusions
In this study, a simple and reliable analytical method for the determination of 13 bioactive constituents of ZSS by HPLC-MS-MS was developed, and parameters such as linear range and recovery were calculated with recoveries ranging from 96.9 to 98.9 and RSDs from 1.12% to 1.93%. Compared with the traditional method, the analytical method developed in this study increased the analysis of multiple bioactive constituents, which made the identification of ZSS components more accurate. Moreover, the changes in bioactive constituents of ZSS with different frying times were analyzed using this analytical method, and it was found that the surface of ZSS started to be oily and accompanied by a slight aroma at the frying temperature of 150 °C after 65 to 80 min, and the content of bioactive constituents during this time was found to increase with the increase in frying time, as determined by the analytical method, which provides a process reference for the preparation of ZSS herbal products. In addition, this study evaluated ZSS from 14 different production areas in China using total bioactive constituents as the assessment index and screened out the production areas with higher total bioactive constituents. The 3 production areas, Shanxi Changzhi, Shandong Jining, and Hebei Xingtai, had higher active ingredient contents of ZSS than the other production areas, which provides a reference value for selecting high-quality ZSS raw materials.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This work was supported by the Beijing Natural Science Fund (KZ201710020014) and the National Natural Science Foundation of China (31601658).

