Abstract
Rutin dietary supplements may offer pharmacological benefits as anticancer and antiinflammatory properties. This study aimed to investigate the inhibitory and protective effect of rutin on signaling pathways of mammary gland carcinogenesis expermintally induced in female rats by 7,12-di-methyl benz (a) anthracene (DMBA). Results showed that rutin administration ameliorated DMBA toxicity and carcinogic effect on kidney and liver revealed by a significant decrease of urea and creatinine levels, and the activity of the liver enzymes alanine aminotransferase (ALT) and alkaline phosphatase (ALP). The antioxidant state indicated by the total antioxidant capacity (TAC) was significantly increased accompanied by a reduction in the inflammatory markers of interleukin-1β (IL-1B), interleukin-6 (IL-6), and tumor necrosis factor (TNF-α) with induction of apoptosis indicated by a significant increase in caspase-3 level. Rutin significantly reduced the levels of the tumor markers carcinoma antigen 15-3 (CA 15-3) and proto-oncogene tyrosine-protein kinase Src1 (Src1). along with downregulation of nuclear factor-kB (NF-κB), heat shock protein 90 (HSP 90), and inducible nitric oxide synthase (iNOS) gene expression. The present study demonstrated the beneficial anticancer activity of rutin as a protective and therapeutic agent. Rutin induces its antitumor activity through elevation of the antioxidant state, inhibition of inflammatory cytokines, downregulation of oncogenes expression, and stimulation of apoptosis.
Introduction
Worldwide 19.3 million new cases of cancer were estimated and about 10 million deaths occurred in 2020. Female breast malignancy is the most diagnosed cancer, with 11.7% of new cases and a 6.9% death percentage of total cancer deaths. Due to rising risk factors connected with globalization and a developing economy, the worldwide cancer burden is anticipated to reach 28.4 million cases in 2040, up 47% from 2020. 1
Chemical-induced carcinogenesis in rodents is frequently used in cancer research because of its close resemblance to human tumors. Polycyclic aromatic hydrocarbons, a class of carcinogenic substances found in the environment, include 7,12-dimethylbenz(a)anthracene (DMBA), which is utilized to develop breast tumor in animal models, having both carcinogenic and immunosuppressive effects due to increased prostaglandin E2 synthesis. 2 The carcinogenicity of DMBA has been proposed to be due to different mechanisms that involve DNA damage. The genes that are important for cell growth and survival are disrupted, including point mutation, activation of the ras gene family, along with a disturbance in the cellular oxidant–antioxidant balance,2,3 which plays an essential role in breast cancer development and progression. 4 DMBA-induced mammary carcinoma was found to be stimulated by different hormones exhibited with positive estrogen receptor (ER) and progesterone receptor (PR). 5
New compounds to be employed in cancer therapy either alone or as an addition to current medications, as a combination therapy are highly encouraged in order to maximize efficacy and decrease negative effects. 6 Because they target important pathways in a characteristically synergistic or additive manner, a combination of anticancer medications improves efficacy compared to monotherapy. This strategy could diminish medication resistance while also delivering therapeutic anticancer effects, including slowing tumor growth and metastatic potential, stopping mitotically active cells, lowering cancer stem cell populations, and inducing apoptosis. 7
Natural substances are of significant interest to prevent related side effects in cancer treatment since they are potential multitargeted medicines. 8 According to growing research, natural products’ cytostatic effects are derived from their ability to modulate oxidative stress, inflammation, autophagy, and apoptosis, resulting in the prevention/reduction of their associated toxicity.9,10 Flavonoids are known to have antioxidant and radical scavenging activities, which are the main mechanisms against several diseases such as atherosclerosis and certain cancers, mainly caused due to an imbalance of antioxidant/oxidant revealed oxidative stress. 11 Rutin is of a glycoside include rutinose (α-L-rhamnopyranosyl-(1→6)-β-D-glucopyranose) linked to OH-3 of quercetin (3,3′,4′,5,7-pentahydroxyflavone). 12 It is known as vitamin P and found in many plants, fruits, and especially in citrus fruits (grab fruit, lemon, and orange), black tea, apple peel, and buckwheat.13,14 Rutin has been shown to have antioxidant and anti-inflammatory properties, as well as cytoprotective effects associated with antiaging and anticancer properties. TLR2 has been considered a player in anticancer immunity Rutin's antioxidant effect is revealed by its binding to Fe2+, preventing it from attaching to hydrogen peroxide. 15
High dosages of rutin have been demonstrated to stimulate enzyme systems with protective and adaptive capabilities. 16 Rutin has a protective effect against procarcinogens in HTC hepatic cells 17 and breast cancer cells. 18 In the tumor cells, rutin's anti-inflammatory and anticarcinogenic effect is exhibited through modulating the signaling pathways of JAK/STAT, Wnt, EGF, AP-1, NF-κB, and Akt, with apoptotic stimulatory effect. 19 Rutin is a host inhibitor for tall-like receptors (TLR) mediating the inflammatory response 12 and can be used as anticancer immunotherapies. 20 Furthermore, rutin controls tumor growth through its antiangiogenic effect, which is exhibited by controlling multiple signaling pathways including Ras/Raf, PI3K/Akt, MAPK, and TGF-2/Smad2/3Akt/PTEN, which are involved in tumorigenesis and apoptosis at the cellular level of many types of cancers, including prostate, cervical, gastric, colon, lung, and neuroblastoma.21,22
The current study goal was to assess the protective and therapeutic effects of rutin against breast carcinogenesis in female albino rats induced by 7,12-dimethyl benzanthracene (DMBA) in an attempt to restrict the development, angiogenesis, and metastasis of mammary gland carcinoma.
Materials and Methods
Chemicals
DMBA was obtained from Sigma Chemicals, and rutin (C27 H30 O16.3 H2O; 97%) from Alfa Aesar.
Animals
Fifty young virgin Swiss albino female rats weighing 80 to 90 g were provided by The El-Nile Pharmaceutical Company and Chemical Industries Company, Cairo, Egypt. Rats were housed at the animal house of the National Center for Radiation Research and Technology (NCRRT). During the experiment, the animals were kept in a room with a controlled temperature (24 ± 3 °C) and were kept under typical laboratory circumstances (12 h of light and 12 h of darkness). Rats were given free access to tap water and commercial 21% protein meals. 23
Experimental Design
Rats were divided randomly into 5 groups (n = 10):
- Group 1 (control): rats received daily, by oral gavage, 1 mL of physiological saline. - Group 2 (rutin): rats received daily, for 60 days, by oral gavage, 1 mL of rutin solution (100 mg of rutin/kg. b. wt., dissolved in warm distilled water).
24
- Group 3 (DMBA): rats received, by oral gavage, DMBA (50 mg/kg. b. wt., dissolved in sesame oil)
25
twice through 6 months. - Group 4 (DMBA/rutin) treatment group: rats received by oral gavage DMBA (50 mg/kg. b. wt.) twice through 6 months, then received rutin (100 mg/ kg. b. wt.) for 60 days. - Group 5 (rutin/DMBA) protective group: rats received by oral gavage rutin (100 mg/kg. b. wt.) for 60 days, then received DMBA (50 mg/kg. b. wt.) twice through 6 months with continuous administration with rutin.
The prolonged treatment with rutin was designed to evaluate its toxicity to be recommended as a nutritional supplement. At the end of the experiment, blood was collected by cardiac puncture from anesthetized rats, and serum samples were separated. Also, mammary glands of all groups were dissected.
Biochemical Assay
Serum urea and creatinine levels were determined by using commercial kits of Diamond Diagnostics Company, Cairo, Egypt, and serum alanine aminotransferase (ALT), and alkaline phosphatase (ALP) activities were determined using commercial assay kits of Spectrum Diagnostics, Egypt. Total antioxidant capacity (TCA) in serum was measured using kits of Biodiagnostic Company, Egypt. The level of serum tumor necrosis factor (TNF-α) Cat.#: MBS2507393, interleukin-1β (IL-1B) Cat.#: MBS825017, interleukin-6 (IL-6) Cat.#: MBS355410, mammary gland tissue caspase-3 (Casp-3) Cat.#: MBS018987, carcinoma antigen 15-3 (CA 15-3) Cat.#: MBS2502096, and proto-oncogene tyrosine-protein kinase Src (Src) Cat.#: MBS9928603 were assayed by ELISA using commercial kits of MyBioSource, Inc. USA.
Quantitative Real-Time Polymerase Chain Reaction (qRT-PCR) of NF-kB, HSP90, ER-α, and iNOS Genes
Determination of relative gene expression of nuclear factor-kB (NF-κB), heat shock protein 90 (HSP90), and inducible nitric oxide synthase (iNOS) in the tumors and mammary glands was accomplished by qRT-PCR.
RNA extraction: RNA was isolated from mammary gland tissue by using Qiagen kits, according to the kit instructions. The extracted RNA absorbance ratio of A260/A280 was 1.7 to 2.2. Complementary DNA (cDNA) was synthesized by transcriptase enzyme using the kit of Qiagen, Germany.
Quantitative real-time polymerase chain reaction (qRT-PCR): RT-PCRs were conducted in a thermal cycler step one plus (Applied Biosystems) by Sequence Detection Software (PE Biosystems). The oligonucleotide primers of NF-kB, ER-α, HSP90, iNOS, and GAPDH are listed in Table 1. The RT-PCR reaction mixture contained 25 mL SYBR Green PCR Master Mix (Applied Biosystems), 900 nM of each primer, and 2 µL of cDNA. Relative expression of NF-kB, ER-a, HSP90, iNOS, and GAPDH mRNA was calculated using the comparative threshold cycle method. 26 Data were analyzed with ABI Prism sequence detection system software. The relative quantification was then calculated by the expression 2−ΔΔCt. Ct is the value used for the analysis where the PCR curve crosses the threshold, in the linear part of the curve. As an endogenous control gene, all results were normalized to the GAPDH gene.
Sequence of Primers Used for Real-Time PCR.

Histopathological Study
Autopsy samples from the mammary glands of the different groups were obtained and preserved in 10% formalin for 48 h for histopathological investigations. Tissue was rinsed with tap water before being dehydrated with ethyl alcohol in repeated dilutions. The samples were embedded in blocks made of paraffin. Tissue sections (3-4 mm) were stained with hematoxylin and eosin. 27
Statistical Analysis
Data were subjected to statistical analysis using the Statistical Package for Social Science (SPSS) software package version 20 (Armonk, NY: IBM Corp). Data were represented by the mean and standard deviation (mean ± SD). Differences between various treatment groups were determined by one-way analysis of variance (ANOVA), Least significant difference (LSD) and post hoc test (Tukey). Data were significant when P values were ≤.05 and highly significant when P values were ≤.001. Significance was denoted by superscript letter a: significance versus control group, b: significance versus DMBA group.
Results
Antioxidant State and Liver and Kidney Parameters
Total antioxidant capacity (TAC) was evaluated in the serum as an indicator of the antioxidant state. Results obtained revealed a significant (P ≤ .001) reduction of TAC, by 61.1% in DMBA rats compared to the control group (Table 2). Administration of rutin significantly (P ≤ .001) elevated the TAC level in the treatment group with a percentage change of 61.0%. In the protective group, rutin markedly elevated TAC by 95.8% compared to the DMBA group.
Effect of DMBA and Rutin on Total Antioxidant Capacity, and Liver and Kidney Parameters.

Each value is expressed as mean ± SD for six rats in each group. Data are significant at P ≤ 0.05 and highly significant at P ≤ 0.001. a Significance versus control group. b Significance versus DMBA group.
Serum levels of ALT and ALP activity were evaluated in all groups as an indicator of liver function. Intoxication with DMBA produced rises (P ≤ .001) in ALT and ALP activities by 189.0% and 87.8%, respectively, compared to the control group (Table 2). Rutin administration significantly reduced (P ≤ .001) ALT activity in the treatment group by 31.8% and in the protective group by 36.5% compared to the DMBA group. At the same time, rutin significantly decreased (P ≤ .001) ALP activity in the treatment group by 25.3% and in the protective group by 39.6% in comparison to the DMBA group.
Introduction of DMBA induced a significant increase (P ≤ .001) in creatinine and urea levels in the serum by 330.8% and 189.0%, respectively, when compared to the control group (Table 2). Rutin administration to DMBA rats significantly (P ≤ .001) reduced the creatinine level in the treatment group by 44.6% and in the protective group by 46.4%, when compared with the DMBA group. In the same manner, rutin significantly (P ≤ .001) reduced the urea level in the treatment group by 31.8% and in the protective group by 36.5% compared to the DMBA group.
Breast Tumor Markers of CA 15-3, ER-α, and Src
Serum level of CA 15-3 was evaluated as an important breast tumor marker. Results revealed a significant elevation (P ≤ .001) of CA 15-3 level by 1448.1% in response to DMBA treatment compared to the control (Figure 1A). Rutin introduction to DMBA rats significantly reduced the CA 15-3 level in the treatment group by 64.0% and in the protective group by 65.5% in comparison to the untreated DMBA group.
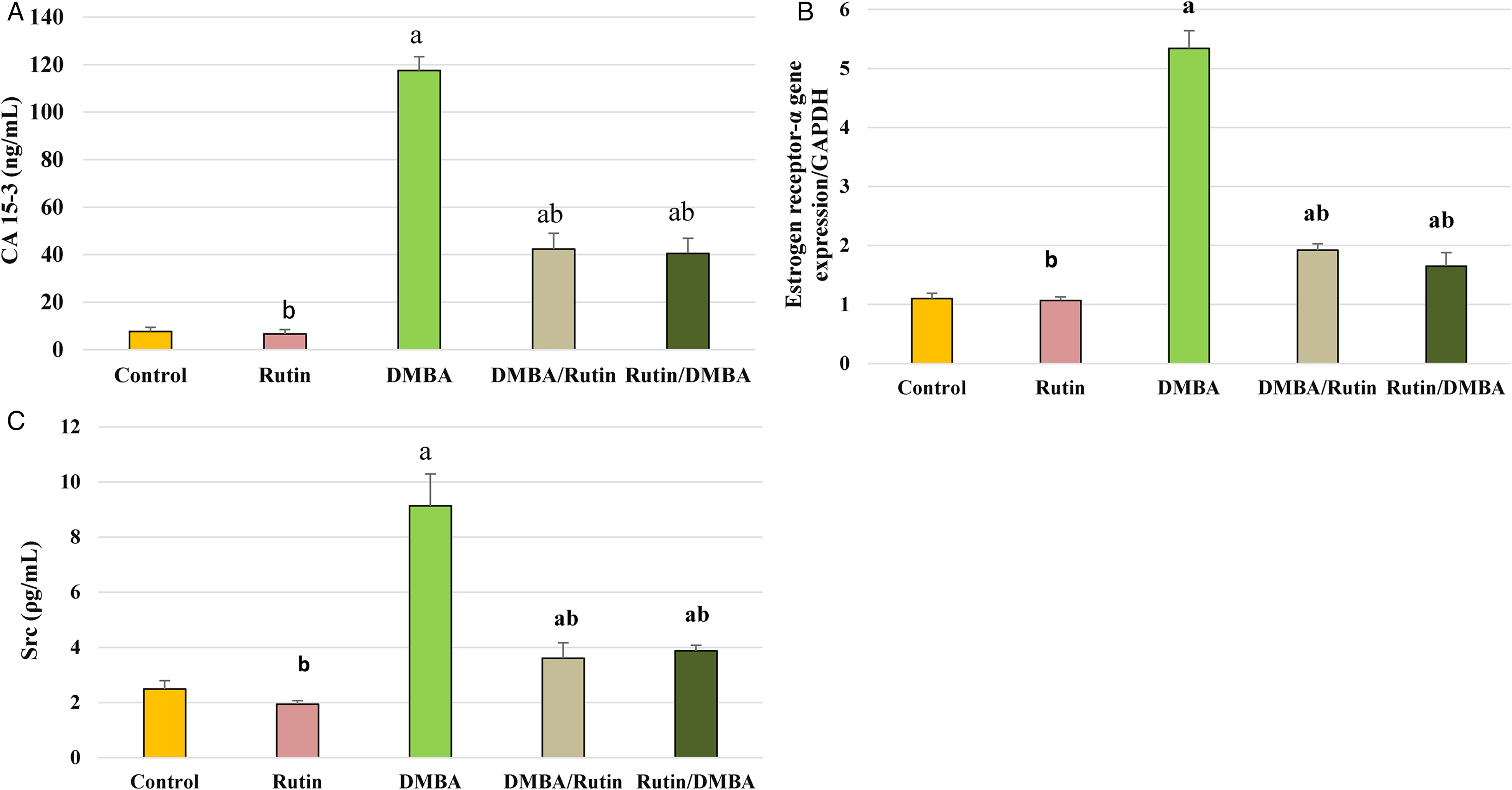
(A) Ca 15-3 level changes with DMBA and rutin. Each value is expressed as mean ± SD. Results are significant at P ≤ .05.aSignificance versus control group, bSignificance versus DMBA group. (B) qRT-PCR analysis of estrogen receptor-α in experimental groups. Each value is expressed as mean ± SD. Results are significant at P ≤ .05. aSignificance versus control group, bSignificance versus DMBA group. (C) Proto-oncogene c-Src (Src) level changes with DMBA and rutin. Each value is expressed as mean ± SD. Results are significant at P ≤ .05. aSignificance versus control group, bSignificance versus DMBA group.
Gene expression of ER-α is a tumor marker for hormone-related breast cancer. qRT-PCR analysis of ER-α gene expression in the mammary gland tissue indicated a significant (P ≤ .001) upregulation of ER-α by 4.9-fold in DMBA-treated rats compared to the control (Figure 1B). Rutin intake by DMBA rats significantly downregulated ER-α expression in the treatment group by 0.35-fold and in the protective group by 3.1-fold compared to the DMBA untreated group.
The level of Src was evaluated in the mammary gland tissue, and the results revealed a significant increase (P ≤ .001) in Src level by 267.1% in DMBA-treated rats compared to the control group (Figure 1C). Rutin intake significantly (P ≤ .001) reduced the Src level in the treatment group by 60.5%. Simultaneously, rutin significantly reduced the Src level by 57.5% in the protective group compared to the DMBA group.
Antiproliferation and Apoptosis
The effect of rutin on tumor growth and regression was evaluated through proliferation and apoptosis markers. Statistical analysis of qRT-PCR data revealed a significant (P ≤ .01) elevation of NF-κB gene expression in the DMBA group by 7.7-fold compared to the control group (Figure 2A). Rutin introduction to the treatment group produced a significant inhibitory effect on NF-κB expression by 0.62 fold. In the protective group, rutin significantly (P ≤ .01) reduced NF-κB expression by 0.49-fold compared to the untreated DMBA group.

(A) qRT-PCR analysis of NF-κβ in experimental groups. Each value is expressed as mean ± SD. Results are significant at P ≤ .05. aSignificance versus control group, bSignificance versus DMBA group. (B) Caspase-3 level changes with DMBA and rutin treatment. Each value is expressed as mean ± SD. Results are significant at P ≤ .05. aSignificance versus control group, bSignificance versus DMBA group.
Evaluating caspase-3 in the different groups indicated an apoptotic stimulatory effect of rutin in the tumor tissue of rutin treated groups. DMBA treatment significantly (P ≤ .001) elevated caspase-3 level by 50.0% in the DMBA rats when compared to the control (Figure 2B). In comparison to the DMBA group, rutin intake significantly (P ≤ .001) elevated the caspase-3 level by 387.7% in the treatment group and by 342.5% in the protective group.
Inflammatory Cytokines
Rats treated with DMBA resulted in a marked elevation of the inflammatory markers IL-1β, IL-6, and TNF-α. DMBA treatment significantly (P ≤ .001) stimulated the secretion of IL-1β by 272.9%, IL-6 by 190.7%, and TNF-α by 433.6% compared to the control. Rutin administration as either a treatment or protective agent showed significant (P ≤ .01) inhibition of the elevated inflammatory markers IL-1β, IL-6, and TNF-α compared to the untreated DMBA rats. Compared to the DMBA group, rutin administrated as a treatment showed significant (P ≤ .001) decreases in IL-1β by 33.7%, IL-6 by 39.7%, and TNF-α by 49.3%. In the protective group, rutin significantly (P ≤ .001) inhibited IL-1β by 36.5%, IL-6 by 47.7%, and TNF-α by 55.9% (Figure 3).

IL-1b, IL-6, and TNF-α l level changes with DMBA and rutin. Each value is expressed as mean ± SD. Results are significant at P ≤ .05. aSignificance versus control group, bSignificance versus DMBA group.
HSP-90 and iNOS Gene Expressions
DMBA tumorigenic activity was detected by HSP-90 gene expression (Figure 4). DMBA significantly upregulated HSP-90 gene expression by 5.1-fold compared to the control group. However, rutin significantly (P < .001) downregulated HSP-90 expression by 0.49-fold in the treatment group and by 0.42-fold in the protective group compared to the DMBA group.

qRT-PCR analysis of HSP90 and iNOS gene expression. Each value is expressed as mean ± SD. Results are significant at P ≤ .05. aSignificance versus control group, bSignificance versus DMBA group.
DMBA induces its carcinogenic activity through induction of NO beside ROS. DMBA induced significant (P < .001) upregulation of iNOS expression by 4.9-fold when compared to the control group. However, rutin introduction significantly downregulated iNOS gene expression by 0.49-fold in the treatment group and by 0.46-fold in the protective group compared to DMBA-treated animals.
Histopathological Examination
The histopathology of mammary gland tissue sections showed normal mammary gland features of average ducts with average epithelial lining, and average acini embedded in the average fibro-fatty stroma (H&E, ×200) (Figure 5A). The rutin-treated rats showed a normal histopathological structure with average ducts with average epithelial lining and preserved outer myoepithelial layer, average acini, and average fibro-fatty stroma (H&E, ×200) (Figure 5B). The DMBA-treated group showed both types of mammary gland cancer, ductal and solid carcinoma. Ductal carcinoma showed a ductal arrangement of the cells with the absence of the basement membrane, and the cells show criteria of malignancy including hyperchromasia, increased nucleus/cytoplasmic ratio, and pleomorphism. The solid carcinoma revealed an arrangement of cells in a solid tumor mass, and the cells showed criteria of malignancy like that of ductal carcinoma (H&E, ×400) (Figure 5C). The mammary gland tissue of the DMBA/rutin group showing dilated ducts with polyp formation and the presence of a basement membrane, dilated congested blood vessels, fatty tissue, and regression of the criteria of malignancy from ill differentiated cells to moderately differentiated cells (H&E, ×400) (Figure 5D). The mammary gland tissue of the rutin/DMBA treated rats showing an improvement in the mammary structure, along with desquamated epithelial lining of ducts, dilated congested blood vessels, and excess periductal fibrous tissue and tumor regression (H&E, ×200) (Figure 5E).

Effect of rutin on mammary gland carcinogenesis induced by DMBA. (A) Section of the mammary gland of the control group showing normal mammary ducts with intact basement membrane and healthy epithelial lining cells (H&E, ×200). (B) Section of mammary gland of rutin-treated rats showing normal mammary ducts with intact basement membrane and healthy epithelial lining cells (H&E, ×200). (C) Section of mammary gland of DMBA treated rats showing ductal carcinoma; the arrangement of malignant cells in ducts with absence of basement membrane and cells showing criteria of malignancy including: hyperchromacia, increased nucleus/cytoplasmic ratio, and pleomorphism (H&E, ×400). (D) Section of mammary gland tissue of DMBA/rutin (treatment) group showing improvement of mammary tissue, and the presence of the basement membrane together with the lining cells (H&E, ×400). (E) Section of mammary gland tissue of rutin/DMBA (protective) group showing improved mammary tissue structure compared to DMBA group with congested blood vessels, and mammary ducts with intact basement membrane (H&E, ×200).
Discussion
DMBA is a powerful carcinogenic compound, which has been used extensively to induce breast carcinoma in animal models. DMBA induces its carcinogenic effect through mutagenic, immunosuppressor, and oxidative stress activities through reactive oxygen species, formation of a DNA adduct, and altering the activities of serum enzymes. 27 In this work, DMBA introduction to female rats significantly depressed liver and kidney activities and induced an oxidative stress state, which was markedly observed in reduced TAC, and elevated ALT, ALP, creatinine, and urea levels. Rutin showed protective and therapeutic effects against DMBA toxicity by ameliorating TAC level, liver enzymes, and kidney parameters.
Liver and kidney impairment in cancer-bearing animals could be attributed to induced DMBA toxicity and to distant metastasis.28,29 Propagation of oxygen and nitrogen reactive species induces DNA damage and cellular alterations and stimulates carcinogenesis. This damage could be prevented or ameliorated by using compounds with antioxidant activity, such as polyphenols, vitamins, or some cofactor metals such as selenium or zinc.30,31
Many cancer cells show serum TAC depression, and this is considered to be an important biochemical marker in breast cancer patients.32,33 Natural antioxidants may enhance the conventional therapy effect against cancer. 30 Previous work has shown that rutin possesses an antioxidant potency that increases glutathione resulting in oxidative stress reduction. 34 Rutin decreases the formation of reactive oxygen species (ROS), NO, glutathione disulfide (GSSG), malondialdehyde (MDA), and inducible nitric oxide synthase (iNOS) activity. Moreover, rutin attenuates mitochondrial damage, increases the glutathione (GSH)/GSSG ratio, enhances the activities of superoxide dismutase, glutathione peroxidase, peroxidase, and glutathione-disulfide reductase, elevated glutathione contents and decreased oxidative stress.35,36
Several studies showed a correlation between inflammation and several types of cancer. Inflammation plays a role in malignant cell proliferation and resistance to cancer therapy. 29 TNF-α, IL-1 β, and IL-6 are essential pro-inflammatory cytokines found in breast cancer patients affecting tumor cell proliferation and survival and may play a pro-tumorigenic role.37,38 Pro-inflammatory cytokine–induced cancer cell invasiveness is mediated by a STAT3-regulated mechanism. IL-6 and TNF-α promote cancer stemness and telomerase activity via phosphorylating transcription factors STAT3 and activate hTERT gene playing a role in telomerase activation in human cancers. 39
IL-1b cytokine is an important mediator of the inflammatory response, activating monocytes/macrophages and inducing IL-6 production mediated by the phosphatidylinositol 3-kinase (PI3K)-dependent AKT/IκB kinase α pathway. IL-1 triggers many cellular pathways including the activation of mitogen-activated protein kinases (MAPKs) involved in the activation of AP-1 and IκB kinases (IKKs) included in the activation of NF-κB. 40 In cancer cells, IL-1β promotes tumor development and spread through neo-angiogenesis and antiapoptotic signaling. 41 IL-1β stimulates the synthesis of IL-6, a critical regulator of immunosuppression in cancer, as well as increased invasiveness and metastasis. 42 It also plays a role in breast cancer therapeutic resistance. 29 IL-6 expression is induced by various stimuli: TNF, platelet-derived growth factor, bacterial lipopolysaccharide, acute viral infections, and transforming growth factor (TGF-β). 43 Inhibition of IL-6 production reduces the risk of chemically induced carcinoma. 44 Inflammatory mediators play a major role in cancer induction through activation of the NF-kB pathway. The IL-6/NF-κB pathway and TNF-α/NF-κB pathways link inflammation to cancer and maintain cells in a transformed state.45,46 In the same way, TNF-α is one of the major cancer inflammatory mediators, involved in inflammation, immunity, cellular homeostasis, tumor promotion, and progression. TNF-α is upregulated in breast cancer and increases stem-like cells, which elevate the chances of cancer recurrence.47,48
As inflammation plays an important role in carcinogenesis, anti-inflammatory drugs can inhibit tumor growth in breast cancer, emphasizing the relevance of the inflammatory microenvironment for tumor growth.27,29 In this work, the increase in pro-inflammatory cytokines: TNF-α, IL-1β, and IL-6 detected in DMBA treated rats was markedly depressed by both preventive and therapeutic treatment with rutin. Many studies have detected the anti-inflammatory and antitumor activity of natural compounds by blocking cytokine signaling and inhibiting their production. Polyphenols have been used as adjuvant therapy for their anti-inflammatory and antioxidant effects. 49 Rutin has been reported to demonstrate numerous pharmacological properties, including antioxidant and anti-inflammatory through inhibition of IL-6 secretion and NF-κB protein expression,50,51 to modulate the production of pro-inflammatory cytokines by decreasing TNF-α and IL-1β generation in microglia, 35 and to show hepatoprotective activity via enhancing the antioxidant effect through amelioration of oxidative stress genes. 51
Rutin may induce its anti-inflammatory effect through downregulating NF-kB expression and inhibiting the NF-κB signaling pathway. NF-κB is an important transcription factor, promoting inflammatory cytokine gene transcription of IL-1B, TNF, and IL-6. It is also involved in the regulation of cell differentiation, proliferation, and apoptosis. 52
CA15-3 level is also an indicator for breast cancer treatment assessment, with alkaline phosphatase being correlated with breast cancer recurrence. 53 The significant elevation of CA15-3 level in this work indicated the occurrence of breast cancer, while the reduction in CA15-3 through rutin intake in the treatment and protective groups indicates rutin's efficiency as a chemotherapeutic and protective agent for breast cancer. 54
Estrogens play an important role through binding to ER in attenuating oxidative stress by preventing ROS production either by regulating ROS generating enzymes or augmentation of ROS eliminating mechanisms. 55 Estrogen receptor-α contributes to carcinogenesis and tumor progression as an oncogene. 54 The present work showed the stimulatory effect of DMBA on ER-α gene expression.5,56 Rutin ameliorated this elevation when introduced as a protective and therapeutic agent. Many studies proved the inhibitory activity of natural products on the expression of ER-α, known as an antiestrogenic effect for cancer management. Antiestrogenic phytochemicals, such as ellagic acid, EGCG, green tea catechin, graviola, and genistein, were found to potentially inhibit the expression of ER-α.56,57
Estrogen binding to its receptors activates the p85 regulatory subunit of class I phosphoinositide 3-kinase (PI3K), mitogen-activated protein kinase (MAPK), and Src. These signal transduction pathways activated by estrogen result in stimulating cell survival and proliferation signals. 58
The iNOs enzyme was found to be involved in cancer promotion and inhibition. It has been evident that nitric oxide (NO) signaling is implicated in the tumorigenesis and progression of many cancer types, including breast cancer. Increased NO generation is associated with tumor grade and expression of various signaling components important for cancer development, such as ER and VEGF. 59 Our results detected significant upregulation of iNOS gene expression in the breast tissue as a result of exposure to DMBA, which was depressed by rutin administration as either a treatment or protective agent. Upregulation of iNOS expression produces NOs, which promote cancer formation and progression.60,61 Rutin is a potent antioxidant polyphenol that reduces inducible nitric oxide synthase (iNOS) activity; attenuates mitochondrial damage; increases the glutathione (GSH)/GSSG ratio; enhances the activities of superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPx); and modulates the production of pro-inflammatory cytokines by decreasing TNF-α and IL-1β generation in microglia. 35
Src kinase is activated by nitric oxide generation, but this activation could be abolished with a nitric oxide scavenger. 62 Src kinases are key regulators for cell proliferation playing a role in tumor progression and development of metastasis and represent a logical target for the treatment of prostate and breast cancer. 63 The significant elevation of Src observed in the present study in DMBA rats is in line with that of human breast cancer which expressed a significantly higher level of about 46% of Src-1 compared to the control. 64 Inhibiting Src-1 represents a logical target for the treatment of prostate and breast cancer as Src targeting agents are of high interest. 65
Src induces HSP 90 production in cancer cells as Src are the transcription clients of the HSP 90 chaperone. 66 HSP 90 inhibition induces cell death and enhances chemotherapy activity also, induces cell cycle arrest with a decrease in cyclin-dependent kinase 4 and 6, and triggers apoptosis through caspase-dependent and caspase-independent methods in Hodgkin's lymphoma cell. Furthermore, inhibition of Src and HSP 90 depresses cancer cell growth and survival through depleted cellular contents of AKt and extracellular signal-regulated kinase (ERK) and inhibition of NF-κB signaling. 67 Rutin exhibited an apoptotic effect through enhancing Bax expression and decreasing Bcl-2 expression, releasing cytochrome c into the cytosol, followed by caspase cascade activation with cleavage of caspase-3, caspase-8, and caspase-9 in human cancer cells. 68
Conclusion
In conclusion, rutin showed marked antitumor activity as a protective and therapeutic agent. Rutin induces its antitumor properties through antioxidant, anti-inflammatory, antiproliferative, and apoptotic activity. Rutin has a modulatory effect on oncogenes, various cancer signaling pathways (TNF-α/NF-kB, iNOs/Src/HSP90), and stimulates apoptosis. We can recommend it as preventive medicine as prolonged intake of rutin exhibited no toxicity.
Footnotes
Author Contributions
The research idea: Magda K El-Din Ezz, Nashwa K Ibrahim, Sawsan M El-Sonbaty. Experimental work: Sawsan M El-Sonbaty and Seham S M Youssef. Structure elucidation and writing: Sawsan M El-Sonbaty, Magda K El-Din Ezz, Nashwa K Ibrahim, Seham S M Youssef.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Experimental procedures were performed in compliance with the standards and guidelines of the National Research Center Ethics Committee, Approval Serial no.: 83A/21.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animals Rights
This work was performed on animal model under the standards and guidelines of the National Research center Ethics Committee.
Statement of Informed Consent
There are no human subjects in the article and informed consent is not applicable.
