Abstract
Hypertension is a significant public health issue that is increasing in prevalence and can have dangerous complications. This atherosclerotic disease occurs due to lifestyle choices, such as being sedentary, eating fast foods, and a lack of physical exercise. Atherosclerosis has gradually become the leading cause of death worldwide. Currently, the trend of using natural products to supplement and replace synthetic drugs is being considered in many places. This study evaluated the antihypertensive and anti-atherosclerosis effects of Nano NO plus capsules on hypertensive and atherosclerotic rat models. The results showed that Nano NO plus given to rats at doses of 420 mg/kg and 840 mg/kg lowered systolic, diastolic, and average blood pressure (BP) without affecting heart rate. Nano NO plus capsules also increased nitric oxide (NO), superoxide dismutase (SOD), glutathione (GSH), and decreased malondialdehyde (MDA) in the rats’ plasma. All of these effects were comparable to those of Losartan at 25 mg/kg. Furthermore, doses of 420 mg/kg and 840 mg/kg reduced blood lipid test indexes, including triglyceride (TG) levels, total cholesterol (TC), low-density lipoprotein-cholesterol (LDL-C), and atherogenic index (AI). At the same time, Nano NO plus capsules increased the levels of high-density lipoprotein-cholesterol (HDL-C) in the blood and protected against atherosclerotic lesions of the abdominal aorta in the studied rats. These results indicate that Nano NO plus is a potential product to treat hypertension and atherosclerotic.
Introduction
Hypertension is a common medical condition in the community, and, in Vietnam, 25% of adults suffer from this condition. 1 In 2015, 8.5 million deaths were associated with high blood pressure (BP), 88% of which were in low-income and middle-income countries. 2 High BP is a major risk factor for heart disease and stroke because it damages the lining of the arteries, making them more susceptible to the buildup of plaque, which narrows the arteries leading to the heart and brain. Hypertension is also a major risk factor for atrial fibrillation, chronic kidney disease, peripheral vascular disease, and impaired cognitive function, among other conditions.2,3
Atherosclerosis has become the main cause of death in the world. The exact cause of this disease remains unknown, but many factors are indicated to contribute to the formation of atherosclerotic plaque. Atherosclerosis is a combination of phenomena that change the structure of the intima of large and medium-sized arteries, including the local accumulation of lipids, glucide complexes, blood and blood products, fibrous tissue, and acid deposits, altering the arterial media. Atherosclerosis can occur in many vascular entities, such as the carotid artery, coronary artery, and lower extremity arteries, and can lead to a variety of related diseases. When atherosclerotic plaques rupture, they generate blood clots that fill the blood vessel, which is already narrowed by atherosclerotic plaques. 4 These almost always play a major role in cardiovascular events: myocardial infarction, cerebral infarction, and limb embolism.5,6 The risk factors for atherosclerosis include traditional risk factors such as dyslipidemia or high ‘bad’ cholesterol in the blood, which are the main risk factors for atherosclerosis. Other risk factors include hypertension, smoking, diabetes, obesity, physical inactivity, stress, and taking birth control pills.6,7
The current treatment trend is to use active compounds from natural herbal sources to supplement and replace synthetic drugs because of their effectiveness and limited side effects.8–10 In Vietnam, natural medicinal products have been widely used in traditional medicine for the treatment of cardiovascular diseases, including atherosclerosis.8,9 Among the significant medicinal products, Nano NO plus capsules were formulated based on the effects of several substances that increase NO synthesis and the transmission of NO signals in the body. They were manufactured and registered as number 1520/2021/DKSP by Nhat Hai New Technology Joint Stock Company with the name Health Protection Food NaNo NO plus. The composition of the Nano NO plus capsule includes L-arginine, L-citrulline, glutathione (GSH), coenzyme Q10 (CoQ10), curcumin, resveratrol, quercetin, and berberine.
L-arginine and L-citrulline are 2 important precursors for nitric oxide (NO) synthesis in endothelial cells. L-arginine is not only a substrate for endothelial nitric oxide synthase (eNOS) enzymes, but also increases cofactor tetrahydrobiopterin (BH4) synthesis in endothelial cells and increases eNOS activity. 11 GSH is a powerful antioxidant that eliminates free radicals and protects eNOS enzyme function and endothelial cells. 12 GSH increases NO and cyclic guanosine-3′,5′-monophosphate (cGMP) synthesis and protects NO from attack by free radicals, thereby increasing NO bioavailability. GSH has also been involved in the S-nitrosothiol (SNO) signaling pathway under oxygen deficiency. 13 Compounds CoQ10, curcumin, resveratrol, quercetin, and berberine all have antioxidant and anti-inflammatory effects which increase NO synthesis, reduce LDL and blood glucose, prevent atherosclerosis, and lower BP.14–17
Although this product has received positive feedback from users who are cardiovascular patients (high BP, myocardial ischemia), there have been no previous investigations on the impact of the product on cardiovascular disease. Therefore, this study evaluated the antihypertensive and anti-atherosclerosis effects of capsules in vivo in hypertensive and atherosclerotic rat models.
Results
Effects of Nano NO Plus on the BP and Heart Rate of Rats
Compared with the physiological control group (G1), the systolic BP of the rats in the pathological control group (G2) increased significantly, beginning at T2 (P < .05) and increasing dramatically (P < .01) at times T3 and T4. Consequently, after 2 weeks of administering N(G)-nitro-L-arginine-methyl ester (L-NAME) (0.5% w/v in distilled water), the rat's systolic BP began to increase significantly (P < .05) and increased markedly (P < .01) after 3 to 4 weeks of L-NAME oral administration (Table 2). Compared with the pathological control group (G2), the systolic BP of the rats in the Losartan group (G3) and the Nano NO plus groups (G4 and G5) decreased significantly at T3 (P<.05). Furthermore, the decline was more noticeable at T4 (P < .01 when comparing G3 and G5 to pathological control groups. Compared with G3 (using Losartan 25 mg/kg), the systolic BP of rats in groups using Nano NO plus (G4 and G5) during evaluation did not show any difference (P > .05). Therefore, Nano NO plus at doses of 420 mg/kg and 840 mg/kg demonstrated a systolic BP-lowering effect in hypertensive rat models induced by oral administration of L-NAME. Furthermore, the antihypertensive effect of Nano NO plus at the tested doses was equivalent to that of Losartan at 25 mg/kg (Table 1).
Results of Systolic Blood Pressure (BP) of Rats.
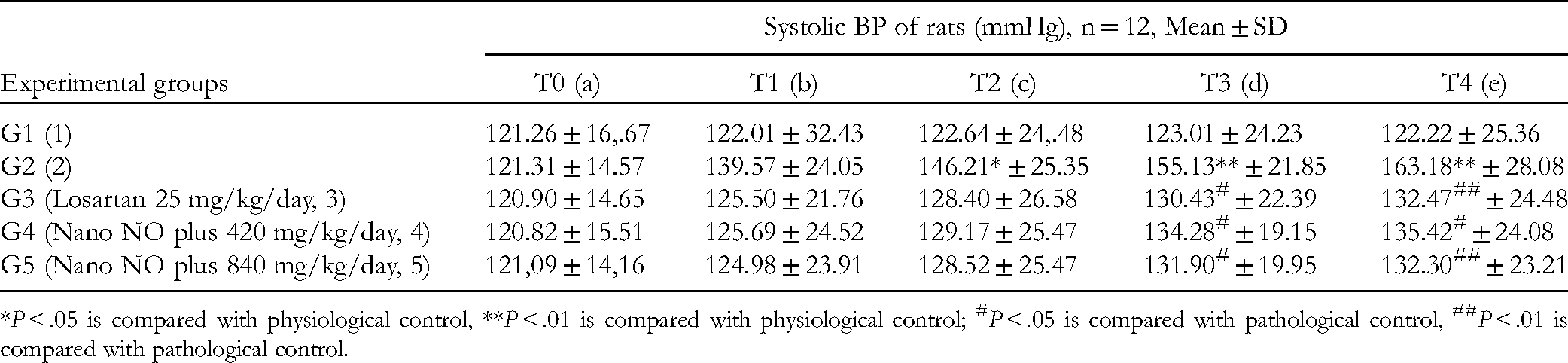
*P < .05 is compared with physiological control, **P < .01 is compared with physiological control; #P < .05 is compared with pathological control, ##P < .01 is compared with pathological control.
Results of Diastolic Blood Pressure (BP) of Rats.

*P < .05 is compared with physiological control. **P < .01 is compared with physiological control. #P < .05 is compared with pathological control. ##P < .01 is compared with pathological control.
In Table 2, compared with the physiological control group (G1), the diastolic BP of the rats in the pathological control group (G2) increased significantly, starting at time T2 (P < .05) and increasing markedly (P < .01) at times T3–T4. After 2 weeks of receiving L-NAME (0.5% w/v in distilled water), the rat's diastolic BP began to increase significantly (P< .05). Furthermore, the increase was remarkable (P < .01) after 3 and 4 weeks. The diastolic BP of the rats in the Losartan group (G3) and the Nano NO plus groups (G4 and G5) decreased significantly compared with the pathological control group (G2), starting at time T3 (P < .05). Additionally, the decrease was more remarkable at T4 (P< .01 when comparing G3 and G5 with a pathological control group). Compared with G3 (using Losartan at 25 mg/kg), the diastolic BP of rats in groups using Nano NO plus (G4 and G5) during evaluation did not show any difference (P > .05). Therefore, Nano NO plus at doses of 420 mg/kg and 840 mg/kg demonstrated a diastolic BP-lowering effect in hypertensive rat models induced by oral administration of L-NAME. Furthermore, the antihypertensive effect of Nano NO plus at the tested doses was equivalent to that of Losartan at 25 mg/kg.
In Table 3, compared with the physiological control group (G1), the mean BP of the rats in the pathological control group (G2) increased significantly, starting at time T2 (P < .05). Additionally, there was a significant increase (P< .01) at times T3 and T4. After 2 weeks of administering L-NAME (0.5% w/v in distilled water), the mean BP of rats began to increase significantly (P < .05); there was a remarkable increase (P < .01) after 3 to 4 weeks of L-NAME oral administration. Compared with the pathological control group (G2), the mean BP of the rats in the Losartan group (G3) and the Nano NO plus groups (G4 and G5) decreased significantly, starting at T3 (P < .05). Additionally, the decrease was more remarkable at T4 (P < .01 when comparing G3 and G5 with a pathological control group). Compared with G3 (using Losartan at 25 mg/kg), the mean BP of rats in groups using Nano NO plus (G4 and G5) during evaluation did not show any difference (P > .05). Therefore, Nano NO plus at doses of 420 mg/kg and 840 mg/kg demonstrated an antihypertensive effect in hypertensive rat models induced by oral administration of L-NAME. Furthermore, the antihypertensive effect of Nano NO plus at the tested doses was equivalent to that of Losartan at 25 mg/kg.
Average Blood Pressure (BP) results.
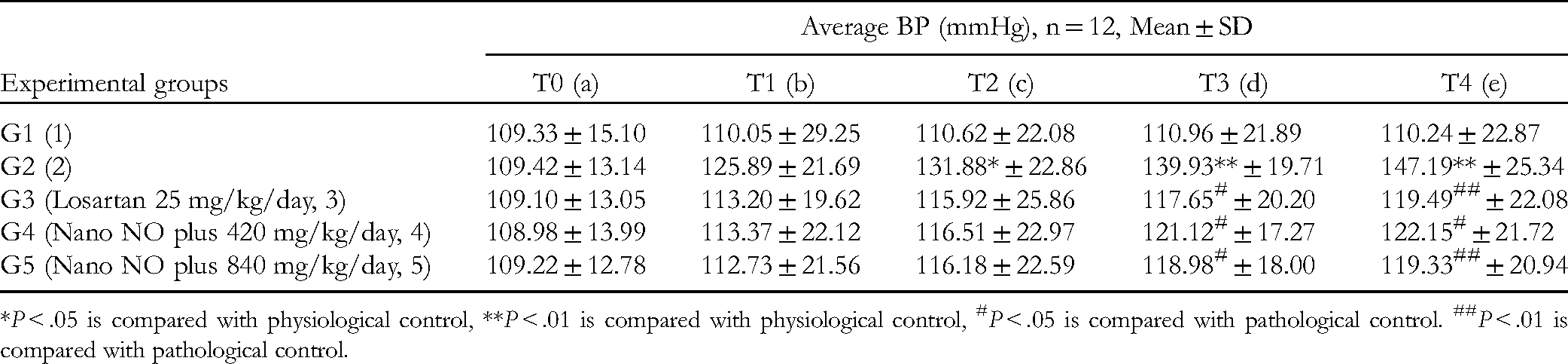
*P < .05 is compared with physiological control, **P < .01 is compared with physiological control, #P < .05 is compared with pathological control. ##P < .01 is compared with pathological control.
The heart rate of rats did not change significantly (P > .05) when groups were compared concerning the same experimental time nor when experimental times were compared within each group. Thus, the test sample did not affect the heart rate of the mice (Table 4).
Results of the Heart Rate of Rats.

Data are presented as Mean ± SD with n = 10.
**P < .01 is compared with physiological control, ##P < .01 is compared with pathological control.
Effect of Nano NO Plus on NO Concentration and Antioxidant Indexes (SOD, GSH, and MDA) in rat Plasma
In the next experiment, the effect of Nano NO plus on NO concentration and antioxidant indexes in the rats’ plasma, including superoxide dismutase (SOD), GSH, and malondialdehyde (MDA), were measured. The results are shown in Table 5. Compared with G1, G2 showed a decrease in plasma (NO, SOD, and GSH concentrations) and a significant increase in serum MDA concentration (P < .01). The NO, SOD, and GSH plasma concentrations in G3 and the Nano NO plus groups (G4 and G5) increased while serum MDA concentrations decreased significantly compared to G2 (P < .01). Compared with G3 (Losartan at 25 mg/kg), the NO, SOD, GSH, and MDA plasma concentrations in the groups using Nano NO plus (G4 and G5) during evaluation did not show any difference (P > .05). Therefore, Nano NO plus at doses of 420 mg/kg and 840 mg/kg showed a NO-increasing effect in rats given L-NAME, a NO-reducing agent. Simultaneously, Nano NO plus also demonstrated an antioxidant effect in rats given L-NAME, as indicated by the increase in SOD and GSH and decrease in MDA. These effects of Nano NO plus at the tested doses were equivalent to those of Losartan at 25 mg/kg.
Nano NO plus Affections on Nitric Oxide (NO) Concentration and Antioxidant Indexes (Superoxide Dismutase [SOD], Glutathione [GSH], and Malondialdehyde [MDA]) in rat Plasma.

Data are presented as Mean ± SD with n = 12.
**P < .01 is compared with physiological control; ##P < .01 is compared with pathological control.
Changes in TC
Changes in the total cholesterol (TC) in the rats’ blood were investigated and the results are presented in Table 6.
Total Cholesterol (TC) (Mmol/L) in rat Blood (n = 12,

▴P < .01 is compared with physiological control, #P < .05 is compared with pathological control, ##P < .01 is compared with pathological control.
After 8 weeks of feeding the high-fat diet, G2–G5 had significantly high levels of TC compared with the physiological control group (P < .01). After 14 days and 28 days of treatment, G4 (treated with the Nano NO plus at doses of 420 mg/kg), G5 (treated with the Nano NO plus at doses of 420 mg/kg), and G3 (treated with reference drug—simvastatin at a dose of 3 mg/kg/24h) exhibited significantly decreased TC compared to G2 (P < .05 and P< .01, respectively). After 14 days and 28 days of treatment, the total blood cholesterol levels had increased slightly in G4 (P> .05) and decreased slightly in G5 (P < .05) compared to G3.
Changes in TG Levels
At 0 days of treatment, G2–G5 had blood triglyceride (TG) levels that were significantly higher than those in the physiological control group (G1; p < 0.01). After 14 days of treatment, G4 and G5 showed significantly decreased blood TG levels compared with G2 (p < 0.01), and G3 showed a significant decrease with P < .05. After 14 days and 28 days of treatment, G3–G5 showed a significant decrease in TG levels in blood compared with G2 (P< .01). After 14 days and 28 days of treatment, the total blood TG levels had increased slightly in G4 (P > .05) and decreased slightly in G5 (P< .05) compared to G3 (Table 7).
Triglyceride (TG) Levels (Mmol/L) in rat Blood (n = 12,
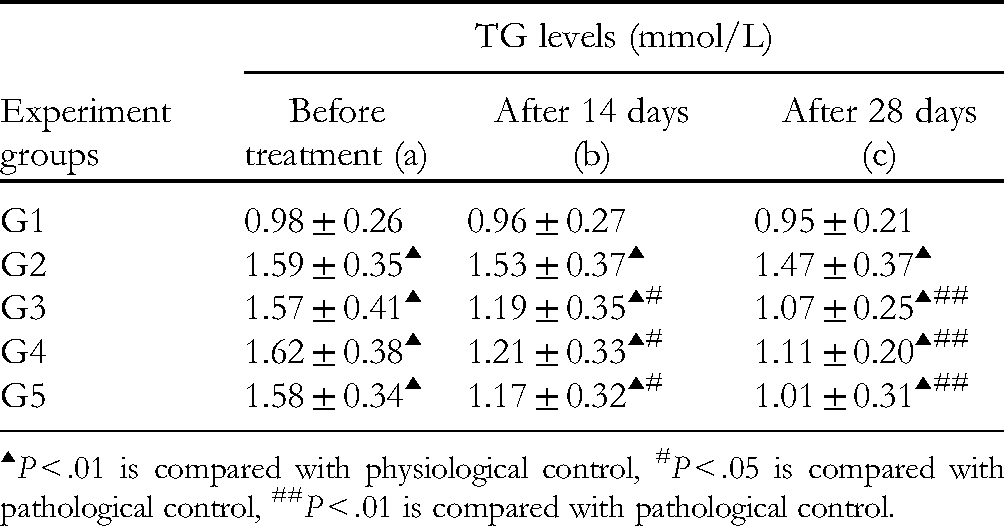
▴P < .01 is compared with physiological control, #P < .05 is compared with pathological control, ##P < .01 is compared with pathological control.
Changes in HDL-C Levels
At 0 days of treatment, G2–G5 showed a slight decrease in blood high-density lipoprotein-cholesterol (HDL-C) compared with the physiological control group (G1; P > .05). After 14 days and 28 days of treatment, G3–G5 showed significantly increased blood HDL-C levels compared to G2 (P < .05). After 14 days and 28 days of treatment, the total blood HDL-C levels had increased slightly in G4 and G5 (P > .05) compared to G3 (Table 8).
High-Density lipoprotein-Cholesterol (HDL-C) Levels in the Blood (Mmol/L) of Rats (n = 12,
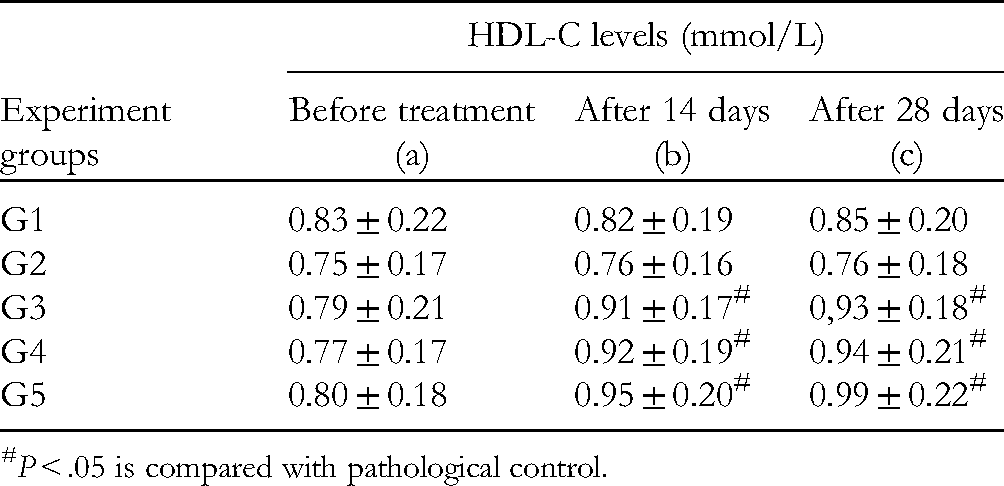
#P < .05 is compared with pathological control.
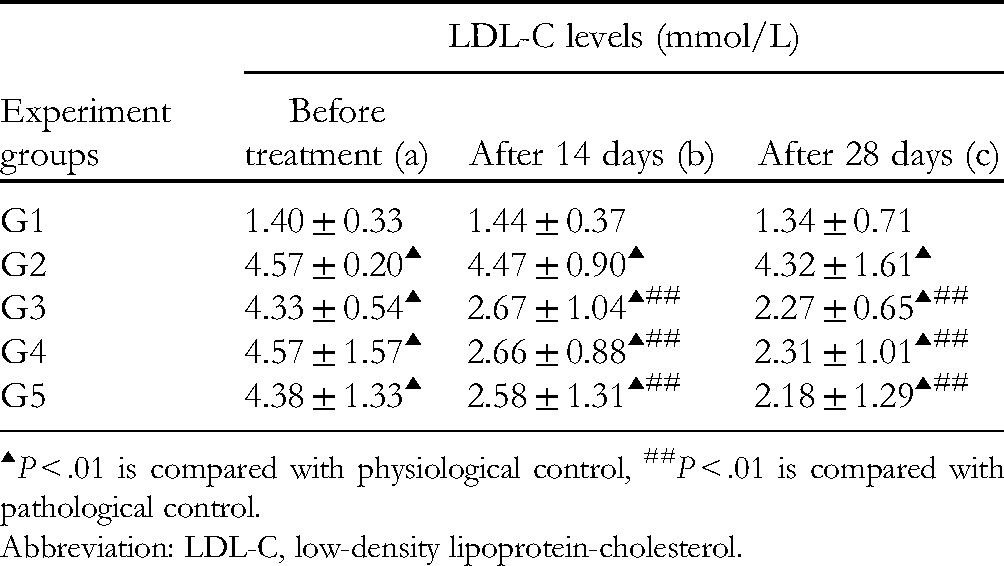
Changes in LDL-C Levels
At 0 days of treatment, the blood low-density lipoprotein-cholesterol (LDL-C) level in G2–5 had increased compared to the physiological control group (G1; P < .01). G4 and G5 had a significant decrease in blood LDL-C levels compared to the model group (P < .01), while G3 showed a significant decrease with P< .05. In addition, G3–5 had significantly decreased blood LDL-C levels in the blood compared to G2 (P < .01), and G4–5 also had significantly decreased blood LDL-C levels in the blood compared to G3 (P < .01) after 14 days and 28 days of treatment (Table 9).
Low-Density lipoprotein-Cholesterol (LDL-C) Levels in the Blood (Mmol/L) of Rats (n = 12,

▴P < .01 is compared with physiological control, #P < .05 is compared with pathological control, ##P < .01 is compared with pathological control.
Changes in AI
At 0 days, the atherogenic index (AI) of G2–5 had increased significantly compared to the physiological control group (G1; P < .01). After 14 days and 28 days of treatment, G3–5 exhibited a significant decrease in AI (P < .01) compared to G2 (P < .01). The AI had increased slightly in G4 and decreased slightly in G5 (P< .01) compared to G3 (Table 10).
Atherogenic Index (AI) (n = 12,
▴P < .01 is compared with physiological control, ##P < .01 is compared with pathological control.
Abbreviation: LDL-C, low-density lipoprotein-cholesterol.
Assessment of Abdominal Aortic Atherosclerosis
Histopathology specimens of the rats’ abdominal aortas were interpreted at the Department of Pathology, Military Hospital 103. Histopathological images of the rats’ abdominal aortas, representing each rat group, are presented in Figure 1.

Microscopic images of rats’ abdominal aortas. (A) Physiological control group; (B) model group: The vessel wall is rough and foam cells (black arrow) appear in the submucosal layer, which is the basis of the atheroma growth (red arrow) forming above; (C) simvastatin reference group; (D) experimental extract dose 1 group; and (E) experimental extract dose 2 group (HE × 400). (C), (D), and (E): Vessel wall thickness was normal. Intimal endothelium structure and submucosal layer were no different when compared with the physiological control group.
The results of the microscopic image analysis of the rats’ abdominal aortas showed that the histopathology of the rat abdominal aorta of G1 is thin and smooth and the muscle cells are oriented horizontally (Figure 1A). However, in G2, the vessel wall is rough and foam cells (black arrow) appear in the submucosal layer, which is the basis of the atheroma growth (red arrow) forming above (Figure 1B). Interestingly, histopathological images of the abdominal aorta in G3–5 were similar to those of the physiological control group, G1. The vessel walls of these groups were slightly rougher than those of G1, with no expression of foam cells or atheroma (Figure 1C, D, and E).
Discussion
The best way to avoid issues related to high BP is to keep BP under control. To control BP, patients must take medication daily for the rest of their lives. Choosing the right drug for each patient is crucial. Many groups of new drugs with active forms alone or in combination have good effects in the treatment of hypertension such as angiotensin-converting enzyme (ACE) inhibitors, angiotensin II AT1 receptor blockers, diuretics, Ca2+ channel blockers, and direct-acting vasodilators, among others. 18 However, drug resistance and side effects caused by long-term administration reduce the effectiveness of treatment and cause complications. The quest for novel drugs to help patients control their BP is of interest to researchers due to the benefit of being able to maintain safe BP for a long period without generating side effects. In our experiments, after 2 weeks of administering L-NAME to rats (0.5% w/v in distilled water), their systolic, diastolic, and mean BP began to increase significantly (P < .05). There was a remarkable increase (P < .01) after 3 to 4 weeks of L-NAME oral administration (Tables 1-3). This proves the success of our experiment using rat models in which hypertension had been induced. Our results are similar to those reported by other authors. Nano NO plus at doses of 420 mg/kg and 840 mg/kg demonstrated antihypertensive effects (systolic, diastolic, and mean BP) in hypertensive rat models induced by oral administration of L-NAME. Additionally, the antihypertensive effect of Nano NO plus at the tested doses was equivalent to that of Losartan at 25 mg/kg (Tables 1-3). In addition to the antihypertensive effect, the product did not affect heart rate, indicating that it is a potential product for the treatment of hypertension (Table 4). The antihypertensive effect of the product may be the result of NO-increasing and antioxidant effects (Table 5). These are 2 important effects on cardiovascular disease in general and hypertension in particular due to vascular disease, which causes a decrease in NO production and damage due to oxidative stress in blood vessels. Our findings regarding this product are consistent with the effects of increasing NO release and anti-oxidation of the component compounds that have been studied by many authors. Vasodilators include NO, prostacyclin (PGI2), and endothelium-derived hyperpolarizing factors (EDHF); vasoconstrictors include thromboxane (TXA2); and endothelin-1 (ET-1). Among them, NO is the most important vasodilator and plays a particularly important role in cardiovascular health and BP. 19
Atherosclerosis is a chronic inflammatory disease with lipid deposition, oxidative stress, and foam cell formation. The persistence of cholesterol-engorged macrophages (foam cells) in the artery wall fuels the development of atherosclerosis. Many studies have focused on the inhibition of foam cell formation, which plays an important role in the early onset of atherosclerosis.20,21 Dyslipidemia is the most important risk factor for atherosclerosis. LDL-C and non-HDL-C are pro-atherogenic. Delayed clearance of TG-rich lipoproteins results in the formation of small dense LDL-C (sdLDL-C) which is associated with insulin resistance and postprandial hyperlipidemia. HDL-C plays an important role in reverse cholesterol transport as well as having anti-inflammatory and antioxidative effects.22,23 In atherosclerotic rat models, the effects of the studied drug were often evaluated for both atherosclerosis and dyslipidemia, suggesting a strong association of drug efficacy in improving these 2 symptoms.24,25 In our experiments, rats were dyslipidemic after 8 weeks of feeding the high-fat diet, with increased TC, LDL-C, TG, and AI, and reduced levels of HDL-C (Tables 6-10). Microscopic images of rat abdominal aortas at G2 showed atherosclerotic and pre-atherosclerotic lesions (Figure 1B). Nano NO plus at doses of 420 mg/kg and 840 mg/kg reduced TC, LDL-C, TG, and AI compared to G2, and increased HDL-cholesterol (Tables 6-10). Microscopic images of rat abdominal aortas at G4 and G5 showed no atherosclerotic and pre-atherosclerotic lesions (Figure 1D and E). The anti-atherosclerotic effect of Nano NO plus is also attributed to antioxidant effects, increasing NO, and regulating dyslipidemia. Antioxidants have a crucial role in the prevention and treatment of atherosclerosis through different mechanisms: the inhibition of LDL oxidation, the reduction of reactive oxygen species (ROS) generation, the prevention of atherosclerotic plaque formation and platelet aggregation, improvement of endothelial dysfunction, and vasodilation, the augmentation of NO bioavailability, and suppression of foam cell formation.26,27 NO has a number of intracellular effects that lead to vasorelaxation, endothelial regeneration, inhibition of lipid peroxidation, inhibition of leukocyte chemotaxis, and platelet adhesion, which are important mechanisms in the prevention of atherosclerosis.28,29 Besides, CoQ10, curcumin, resveratrol, quercetin, and berberine have an effect on regulating dyslipidemia via different mechanisms and contribute to the anti-atherosclerotic effect of Nano NO plus.30–34
To increase NO production in endothelial cells, it is necessary to modify a high-protein diet to provide NO-producing precursors, including L-arginine and L-citrulline, or to supplement these amino acids directly. Additionally, several NO-releasing drugs such as organic nitrates and nitro and nitroso compounds have been used to treat angina attacks, congestive heart failure due to ischemic-reperfusion, and pulmonary hypertension.35,36
GSH is a soluble tripeptide composed of 3 amino acids, cysteine, glycine, and glutamic acid. GSH is an important antioxidant that eliminates lipid peroxides and free radicals. GSH stimulates the loss and compensation process of total L-arginine and increases eNOS enzyme activity, leading to an increase in NO synthesis. 36 GSH also protects NO from attack by free radicals, increasing the bioavailability of the NO signaling molecule. According to McKinley-Barnard et al, the combination of GSH and L-citrulline supplementation not only increases NO synthesis in endothelial cells but also increases the synthesis of nitrite and the important molecule cGMP, which has vasodilatory effects on the NO-soluble guanylate cyclase-cGMP (NO-sGC-cGMP) signaling pathway. 37
CoQ10 or ubiquinone is a lipid-soluble substance and endogenous antioxidant in the body. CoQ10 is effective in eliminating free radicals, reducing OxLDL formation, and enhancing the expression of the eNOS enzyme, which leads to an increase in NO synthesis in endothelial cells, and decreasing the expression of inducible NOS. 14 CoQ10 supplements have clinical significance in reducing cardiovascular risk factors such as regulating dyslipidemia, lowering BP, and preventing atherosclerosis. 38
Among the natural compounds, curcumin is a polyphenol with antioxidant, anti-inflammatory, antifungal, and inhibitory effects on cancer cells and has many beneficial effects on cardiovascular disease and BP.15,16 Curcumin has been shown to have antihypertensive effects on smooth muscle cells by decreasing the expression of angiotensin II Type 1 Receptor. 15 As curcumin has a strong antioxidant capacity, it protects NO from free radicals, leading to an increase in the bioavailability of NO in the NO signaling pathway. As a result, it dilates blood vessels and increases blood flow, aiding blood circulation. 16 In addition, curcumin prevents endothelial dysfunction, smooth muscle cell proliferation and migration, and foam cell formation, and modulates macrophage polarization, regulates dyslipidemia, and protects against atherosclerotic plaque formation. 39
Resveratrol is a polyphenolic phytoalexin with strong antioxidant properties and many beneficial effects on cardiovascular health. Some results of preclinical studies in animal models showed that resveratrol can eliminate active free radicals and increase cofactor BH4 production, thereby increasing eNOS enzyme activity and NO synthesis in endothelial cells. 40 Resveratrol enhances eNOS expression in endothelial cells by activating the promoter of the gene encoding the active eNOS enzyme. Additionally, resveratrol also has the effect of activating phosphorylation of eNOS enzymes at the Ser1177 position, which leads to an increase in the activity of eNOS enzymes. 41 Administration of resveratrol prevented high-fat diet and LPS-induced dysfunction of serum lipids including TC, TG, LDL-C, and HDL-C, ameliorated the thickened coronary artery wall, and decreased the areas of atherosclerotic lesion on aortas. 42
Quercetin is a flavonoid with anti-inflammatory, anti-cancer, antioxidant, and cardioprotective activities. As quercetin has free radical-scavenging and OxLDL inhibitory effects, it protects eNOS from free radical attacks and increases Ca2+ concentration in endothelial cells. Additionally, quercetin increases the activity of eNOS, leading to an increase in NO synthesis in endothelial cells. 43 Furthermore, quercetin has been shown to increase the synthesis of cGMP − a key molecule in the NO-sGC-cGMP signaling pathway that helps dilate blood vessels and increases blood flow. 17 According to a study by Gormaz et al, patients with high BP who were treated with quercetin at a dose of 1 g/day for 28 days had lower and more stable BP compared with the control group using a placebo. 17 In addition, quercetin can reduce the formation of atherosclerotic plaque through anti-inflammatory, antioxidant, regulation of lipid metabolism disorders, and other pharmacological effects. 44
Berberine is an isoquinoline alkaloid with a yellow color and bitter taste. 45 According to Kang et al, testing berberine on a rat aortic rings model (in vitro) showed that berberine, at a concentration of 10 µg/Ml, causes endothelial cells to release NO and cGMP—2 important molecules in the NO signaling pathways that help dilate blood vessels and aid blood circulation. 46 As a supplement to endothelial cell culture medium in a rat aortic rings model with an IC50 = 42 µg/mL (125 µM), berberine inhibited ACE activity, resulting in an antihypertensive effect. 47 Additionally, berberine, with anti-oxidation and cholesterol-lowering effects, inhibited both the proliferation and apoptosis of vascular smooth muscle cells induced by mechanical stretch stress simultaneously, all contributing to the anti-atherosclerotic effect. 48
Overall, Nano NO plus capsules at doses of 420 and 840 mg/kg lowered the systolic, diastolic, and average BP of hypertensive rats. Nano NO plus also increased NO in the plasma of rats and had an antioxidant effect, as shown by the index of increasing SOD and GSH and decreasing MDA in the plasma of white rats with hypertension induced by oral administration of L-NAME. Furthermore, Nano NO plus capsules at doses of 420 and 840 mg/kg also exhibited good effects in treating atherosclerosis, equivalent to the reference drug simvastatin at a dose of 3 mg/kg/day. The experimental capsules reduced the lipid test indexes in the blood, including TG levels, TC, LDL-cholesterol, and the AI, increased blood HDL-cholesterol, and inhibited foam cell or plaque formation. Hypertension is one of the major risk factors for cardiovascular diseases, while atherosclerosis underlies many of the common causes of cardiovascular deaths, including stroke and heart attack. 49 Furthermore, hypertension not only enhances the development of atherosclerosis but also destabilizes the plaques, increasing cardiac death. 50 With lowering BP and anti-atherosclerotic effects, Nano NO plus has potential value in the prevention and treatment of cardiovascular diseases in general, and hypertension and atherosclerosis in particular. Further studies on lowering BP, anti-atherosclerosis, and other cardiovascular effects such as angina, coronary heart disease, and heart failure need to be conducted to better understand the effect of Nano NO plus on cardiovascular disease.
Materials and Methods
Nano NO Plus Capsule
The capsule containing 550 mg Nano NO plus was manufactured and supplied by the OIC Company, meeting Vietnam's basic standards. The composition of each Nano NO plus capsule is comprised of L-arginine (150 mg), L-citrulline (300 mg), GSH (50 mg), CoQ10 (10 mg), curcumin (10 mg), resveratrol (10 mg), quercetin (10 mg), and berberine (10 mg).
The expected human dose of Nano NO plus is 3000 mg/person/day. For a person weighing 50 kg, the dose is 60 mg/kg/day. According to the conversion coefficient from humans to experimental animals, the dose expected to be effective in rats (conversion factor 7) is 420 mg/kg/day. This was the base dose (dose 1) used to evaluate the effects of the Nano NO+ capsules. The high dose level (dose 2) used in the study was twice as high as dose 1, ie, 840 mg/kg/day.
Experimental Animals
One hundred and twenty healthy male Wistar rats weighing 200 to 220 g were used in this study. The healthy rats were evaluated according to the criteria of smooth hair, clear eyes, dry anus, activity, normal movement, normal eating habits, and normal waste production. Experimental animals were provided by the Board of Animal Husbandry at the Vietnam Military Medical University and raised in the laboratory for at least 1 week before the experiment. Animals were provided a standard laboratory diet and free access to clean cooled boiled water. The experimental protocols (anti-atherosclerosis and antihypertensive) were approved by the Vietnam Military Medical University, Hanoi, Vietnam (Permission number IACUC-2202/21 issued on February 22, 2021).
Evaluation of the Antihypertensive Effect of Nano NO Plus
Evaluation of the antihypertensive effect of Nano NO plus on the L-NAME-induced hypertensive models was according to the method described by Bilanda et al 51
Male Wistar rats were divided into 5 groups of 12 animals each:
- Group 1 (G1, physiological control): non-induced hypertension + drinking distilled water. - Group 2 (G2, pathological control): induced hypertension + drinking distilled water. - Group 3 (G3, reference drug): induced hypertension + Losartan at a dose of 25 mg/kg/day. - Group 4 (G4, dose 1): induced hypertension + Nano NO plus at a dose of 420 mg/kg/day. - Group 5 (G5, dose 2): induced hypertension + Nano NO plus at a dose of 840 mg/kg/day.
Hypertension was induced in the rats by oral administration of L-NAME (0.5% w/v in distilled water) at a dose of 50 mg/kg/day for 4 consecutive weeks. Rats were simultaneously given distilled water, reference drugs, or reagents (forced drinking using a special curved needle). The systolic BP, diastolic BP, and heart rate of the rats in all groups were measured using a noninvasive BP measurement system on the rats’ tails at the following time points: T0 (before drug administration), T1, T2, T3, and T4 (after 1 week, 2 weeks, 3 weeks, and 4 weeks of taking the drug). To acclimate to BP recording conditions, rats’ movement was restricted using a confinement device for 10 to 20 min per day for 5 days before taking BP measurements. BP was recorded at the base of the tail using the tail-cuff technique. Rats were warmed for 30 min at 28 °C in a thermostatically controlled heating cabinet (Ugo Basile, Italy) to better detect caudal artery pulse. Then, the tail was passed through the cuff and the tail-cuff sensor was connected to the amplifier (ML 125 NIBP, AD Instruments). To minimize stress-induced BP variations, all measurements were taken by the same person in the same quiet environment at the same time of day. The mean BP of rats was calculated according to the formula below
52
:
Evaluation of the Anti-Atherosclerosis Effect of Nano NO Plus
Wistar rats, eligible for the experiment, were randomly assigned to 5 groups with 12 animals in each group.
+ Group 1 (G1, physiological control): no atherosclerosis + distilled water. + Group 2 (G2, pathological control): induced atherosclerosis + distilled water. + Group 3 (G3, reference drug): induced atherosclerosis + oral administration of simvastatin at 3 mg/kg/24 h. + Group 4 (G4, dose 1): induced atherosclerosis + Nano NO plus at a dose of 420 mg/kg/day. + Group 5 (G5, dose 2): induced atherosclerosis + Nano NO plus at a dose of 840 mg/kg/day.
Atherosclerosis was induced according to the method described by Yurina et al
54
The rats were fed a high concentration of fried fats for 8 weeks. Fatty food for rats was prepared according to the following formula: cornmeal 42.7%, soybean oil 7%, casein 20%, sucrose 10%, cellulose 5%, gelatin 3% (heated at 190 °C for 24 h) 10%, cholic acid 0.3%, and cholesterol 2%. The treatment was given daily for 28 days at 8 am after the completion of 8 weeks of feeding a diet high in fried fats. Blood samples were obtained from rats at 0, 14, and 28 days after treatment began to examine blood lipid indicators: TG, TC, HDL-C, and LDL-C. The AI is used to understand and assess the risk of development of atherogenicity in vessels. It was calculated according to the formula below
55
:
Statistical Analysis
The data in this experiment were processed with the biomedical statistics method using SPSS 22.0 software. Figures are shown as
Footnotes
Acknowledgements
The authors are thankful to the management, the principal of Department of Pharmacology – Military Medical Academy, and Department of Pathology and Forensic Medicine, 103 Military Medical Hospital, Vietnam for providing the necessary infrastructure and conducting the experiments.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was conducted under the research grant received from Nhat Hai New Technology Joint Stock Company (2021).
Ethical Approval
It is applicable for this article.
Statement of Human and Animal Rights
This study contains animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
