Abstract
Cervical cancer is the fourth most common female malignancy with high morbidity and mortality, which urgently needs novel anti-cancer drugs. Accumulating investigations have focused on the antitumor activity of betulinic acid (BA), which is a natural compound with low toxicity and high efficiency. Although the effect of BA on SiHa cells is obvious, the specific mechanism is seldom studied. Target identification is an important part of research on the internal mechanism of action. In this current study, an integrated method based on literature collection, target prediction, enrichment analysis, network analysis, and western blotting experiments was performed to identify the potential key targets of BA-induced apoptosis. Then, combined with the identified potential key targets, the specific mechanism of BA-induced cervical cancer SiHa cells apoptosis was elucidated. Our present study demonstrated that BA significantly reduces the viability of cervical cancer SiHa cells in a dose- and time-dependent manner. In addition, 8 potential key targets (AKT1, CASP8, LMNA, TNF, BCL2, CASP3, PARP1, and XIAP) were obtained through our integrated target identification method. Meanwhile, western blotting showed that within a certain concentration range, the expression of cleaved-caspase 3, cleaved-PARP, and cytochrome c increased with the BA concentration, while XIAP was almost unchanged. Therefore, the effect of BA on cervical cancer is noticeable. BA-induced SiHa cells apoptosis is a multi-molecule coordinated process. In this process, BA is not only a participant in either the extrinsic or intrinsic pathways, but also a regulator of apoptosis effector molecules of the CASP3/PARP1 axis.
Introduction
Cervical cancer is the fourth most common female malignancy worldwide and represents a major global health challenge. 1 Each year, more than half a million women are diagnosed with cervical cancer and the disease resulted in over 300,000 deaths in the whole world. 2 At present, the clinical treatment of cervical cancer is mainly based on surgery, radiotherapy, and chemotherapy. 3 In addition, immunotherapies have received regulatory approval and therapeutic vaccines are currently being explored.4,5 Meanwhile, a large number of natural products have been studied in anti-cervical cancer research. For example, quercetin suppresses cervical cancer aggressiveness by reversing epithelial–mesenchymal transition signaling 6 ; curcumin inhibits invasion and proliferation of cervical cancer cells via impairment of NF-kB and Wnt/β-catenin pathways 7 ; and formononetin represses cervical tumorigenesis by interfering with the activation of PD-L1 through MYC and STAT3 downregulation. 8 Despite advances in the treatment of cervical cancer, its high morbidity and mortality still pose many threats to women. Therefore, the development of novel natural anti-cancer drugs may provide more treatment options.
Betulinic acid (BA) is a naturally occurring pentacyclic triterpene with various pharmacological activities such as antitumor, anti-inflammatory, anti-angiogenesis, and immune regulation.9,10 In particular, its antitumor effect has received widespread attention. Currently, studies have shown that BA has extensive specificity for a variety of tumor types, such as melanoma, leukemia, colon carcinoma, lung carcinoma, prostate carcinoma, multiple myeloma, and cervical cancer.11-13 In addition, the cytotoxicity of BA only affects tumor cell lines, but not normal cells.14,15 In vivo studies with rats also showed that even when the BA concentration reached 500 mg/kg, there was no obvious toxicity. 16 It can be seen that BA has the potential to be developed into a low-toxic and high-efficiency antitumor agent.
The effect of BA on cervical cancer is worthy of attention. Some studies have shown that 30 μM BA had a 50% to 100% inhibitory effect on HeLa cells,17,18 and 100 or 200 mg/kg BA effectively suppressed tumor growth in cervical carcinoma (U14) tumor-bearing mice . 19 These results confirmed the therapeutic potential of BA for cervical cancer. However, the specific mechanism of BA against cervical cancer has been little studied, and inducing apoptosis may be an important part of its anti-cervical cancer activity. According to existing research, BA could induce apoptosis by regulating PI3K/Akt signaling, and the endoplasmic reticulum stress (ERS) and reactive oxygen species (ROS)-medicated mitochondrial pathways in human cervical cancer cells.18,20,21 Although this research has pointed the way to a certain extent, the mechanism of BA-induced apoptosis of cervical cancer cells still needs to be further improved.
Choosing the right biological target is a critical first decision in new drug development. 22 Natural products usually have more than 1 biological target, and the key target is often the core of its mechanism of action. 23 Target identification aims to identify key targets and even discover new targets through a series of bioinformatics, computational simulations, experiments, and statistics.24-26 Surprisingly, target prediction of active natural products has gradually become an indispensable part of target identification due to its low cost and high speed, although wet experiments are more convincing. 27 Currently, target prediction can be roughly classified into 2 methods, bioinformatics-based and computer-aided. Bioinformatics-based target prediction is more inclined to the application of network pharmacology, and computer-aided target prediction is more based on molecular docking theory. However, the possibility of false positives is inevitable, no matter which prediction method you choose. Perhaps, the integration of multiple prediction methods combined with experimentation is an alternative to target identification.
We have studied the effects of different concentrations of BA on cervical cancer SiHa cells, and clarified its mechanism of inducing apoptosis through a target identification-based method. Further work will provide an experimental basis for the development of BA as a potential natural medicine for the treatment of cervical cancer.
Materials and Methods
Reagents and Antibodies
The human cervical cancer cell line SiHa was obtained from the American Type Culture Collection (ATCC, USA), and given by the Zhejiang University School of Medicine. BA (purity≥98.0%) was purchased from MedChemExpress, USA, Dulbecco's modified eagle's medium (DMEM), fetal bovine serum (FBS), and 0.25% trypsin-EDTA from Gibco, USA, cell culture flasks, 6-well plates, 96-well plates, and Pasteur pipettes from Corning, USA, phosphate buffer saline (PBS) from Northendbio, China, the cell counting kit-8 (CCK-8), phenylmethylsulfonyl fluoride (PMSF), BAC protein assay kit, RIPA lysis buffer, and Tris-buffered saline containing Tween 20 (TBST) from Biosharp, China, the TUNEL kit from Beyotime, China, and the XIAP polyclonal antibody, active caspase-3 monoclonal antibody, CYCS monoclonal antibody, and cleaved-PARP-1 polyclonal antibody from ImmunoWay Biotechnology.
Preparation of BA Solution
BA was dissolved in dimethyl sulfoxide (DMSO) to prepare a 10 mM stock solution and stored away from light. Gradient dilution with culture medium was prepared as needed.
Cell Cultures and Treatments
SiHa cells were cultured in DMEM high-glucose medium, which contained 10% FBS. They were cultured in a humidified incubator with 5% CO2 at 37 °C (Thermo Fisher Scientific), and passaged twice a week during the culturing period.
Cell Viability Assays
The viability of SiHa cells was quantified using the CCK-8 assay system. In brief, SiHa cells were plated into 96-well plates at a density of 5 × 103 cells/well with 100 μL culture media. After 24 h, the culture medium was replaced by a fresh medium containing different concentrations of BA (20, 40, 60, 80, and 100 μM), and then SiHa cells were cultured for 24 and 48 h, respectively. Subsequently, to each well 10 μL CCK-8 solution was added for incubation for 1.5 h. Eventually, the optical density (OD) values at 450 nm were measured. Cell viability (%) = (OD value in experimental group – OD value in blank control group)/(OD value in control group – OD value in blank control group) × 100%. Each group contained 6 wells at least and was repeated 3 times.
Time Curve of BA on SiHa Cells
The concentrations of IC10, IC50, and IC90 were calculated by the cell survival curve at 48 h. After allowing the cells to settle down for 24 h in 96-well plates, the culture medium was replaced by a fresh medium containing different concentrations of BA (IC10, IC50, and IC90). Then SiHa cells were cultured for 0, 6, 12, 24, 48, and 72 h, respectively, and the cell viability was measured by CCK-8 assay. Eventually, the time curve of BA was drawn using GraphPad Prism (Version 7.0.2).
TUNEL Staining
SiHa cells were plated onto glass slides in a 12-well plate at a density of 1 × 105 cells per well, and incubated with various concentrations of BA (IC10, IC50, and IC90). Subsequently, a TUNEL kit was used according to the instructions. The cells were washed with PBS, proteinase K working solution was added, immersed in blocking buffer, fixed, rinsed, and infiltrated with 0.1% Triton X-100, followed by fluorescein isothiocyanate (FITC) end labeling of apoptotic DNA fragments. The nuclei were counterstained with DAPI. Finally, the FITC-labeled TUNEL-positive cells were observed under a fluorescence microscope, and the TUNEL-positive cells were counted in at least 6 fields of view.
Collection and Prediction of Potential Targets
The literature on BA and cervical cancer were searched on PubMed database (https://pubmed.ncbi.nlm.nih.gov/), and the related proteins were obtained as the targets of literature collection. Meanwhile, an integrated screening strategy was used for target prediction: First, 300 potential targets obtained from the PharmMapper database (http://www.lilab-ecust.cn/pharmmapper/) were molecularly docked with BA using AutoDock Vina software (Version 1.1.2), and the targets with a docking score less than the experimental ligand were screened; second, the proteins related to cervical cancer were collected through DisGeNET (https://www.disgenet.org/), OMIM (https://omim.org/), KEGG (https://www.kegg.jp/), TTD (http://db.idrblab.net/ttd/), and RGD (https://rgd.mcw.edu/) databases; third, the targets obtained from the molecular docking and the proteins related to cervical cancer were intersected to obtain the predicted targets of BA against cervical cancer. In addition, the names of all the targets were unified through the GeneCards database (https://www.genecards.org/) for standardization.
Enrichment and Network Analysis
The potential targets of BA against cervical cancer were enriched in the biological pathway and process of P < .05 through the Metascape database (https://metascape.org/), and the enriched targets were considered as important targets for BA to regulate the pathway or process. Subsequently, the protein-protein interaction network of potential targets was constructed by Bisogent software (Version 3.0.0) and String database (https://string-db.org/), and the network was visualized and analyzed by Cytoscape software (Version 3.7.2). According to the results of network analysis, potential key targets were further screened from the enriched targets.
Western Blotting
The cells were plated in 6-well plates at a density of 5 × 105 cells/well in 2.5 mL culture media. After 24 h, 2.5 mL new culture medium was added to the control groups, and to the experimental groups the IC10, IC50, and IC90 concentrations of BA solution were added. After 48 h of treatment, cells were gently washed with iced PBS, twice, protein lysis buffer was added, and debris was eliminated by centrifugation at 12,000 rpm for 10 min at 4 °C.
Following addition of sample loading buffer, the proteins were separated according to their molecular weight by electrophoresis and then electrotransferred onto polyvinylidene fluoride (PVDF) membranes. The blots were blocked for 1 h at room temperature with fresh 5% nonfat milk in TBST and then incubated with specific primary antibodies diluted in TBST, for 16 to 18 h, at 4 °C. Following 3 washes with TBST, the blots were incubated with respective secondary antibodies for 30 min and the immunoreactive bands were visualized using an enhanced chemiluminescence kit. The bands were quantified using Image-Pro Plus software (Version 6.0).
Statistical Analysis
For evaluation of the significant differences between groups, SPSS software (version 16.0) was used for statistical analysis, and the data were analyzed using an independent sample t-test. P < .05 was considered statistically significant. Results were reported with mean ± SD.
Results
Effects of Exposure to BA on SiHa Cells
SiHa cells were treated with different concentrations of BA (0, 20, 40, 60, 80, and 100 μM) for 24 and 48 h. The variations in SiHa cell viability with increases in BA concentration are shown in Figure 1. BA inhibited SiHa cell proliferation in an obvious dose-dependent manner after 24 and 48 h. The calculated IC50 values of SiHa cells treated with BA for 24 and 48 h were 40.98 ± 2.97 μM and 29.82 ± 0.89 µΜ, respectively.

Cell viability variation at different concentrations. The circles and triangles represent 24 and 48 h after betulinic acid (BA) treatment, respectively.
Time Curve of BA on SiHa Cells
The IC10, IC50, and IC90 concentrations were 18.70 ± 1.09, 29.82 ± 0.89, and 47.59 ± 1.26 μM, respectively. SiHa cells were treated with these 3 concentrations for 0, 6, 12, 24, 48, and 72 h, respectively. As shown in Figure 2, BA inhibited the proliferation of SiHa cells in a time-dependent manner. Compared with 0 h, the cell viability of the IC10 treated cells was significantly decreased in the first 24 h of BA treatment, and then there was no difference. With the passage of time, the cell viability decreased significantly within 72 h after treatment with BA at IC50 and IC90 concentrations, but, except at 24 h with the IC50 concentration, the curves of IC50 and IC90 showed a downward trend.

Cell viability variation at different times. The square, circle, and triangle represent betulinic acid (BA) treatment with IC10, IC50, and IC90 concentrations, respectively. *, #, and + are PIC10, PIC50, and PIC90 respectively, compared with the control group, *P < .05, **P, ##P, ++P < .01.
TUNEL Apoptosis Assay Results
SiHa cells were treated with different concentrations (0, IC10, IC50, and IC90) of BA for 48 h. Intuitively, DAPI staining revealed the decrease of cells with the increase in BA exposure (Figure 3). The levels of apoptosis in each group were determined using TUNEL staining. As shown in Figure 3, TUNEL-positive cells in the control group could hardly be observed. The No. of TUNEL-positive cells in the IC50 and IC90 groups was obviously higher than that in the control group, which was highest in the IC90 group. The above results revealed that BA-induced apoptosis in SiHa cells in a dose-dependent manner.

TUNEL staining. SiHa cells were treated with different concentrations (0, IC10, IC50, and IC90) of betulinic acid (BA), followed by TUNEL staining and visualization under a fluorescent microscope. Scale bar: 250 μm (green: TUNEL-positive, blue: DAPI).
Collected and Predicted Potential Targets
We have obtained 50 targets of BA against cervical cancer from the literature in PubMed database. In target prediction, 98 BA targets were obtained through virtual screening, 1076 cervical cancer-related proteins were collected through multiple disease databases, and 32 predicted targets were finally obtained through the intersection of the first two.
Enriched Biological Pathways and Processes
After deleting duplicates, 81 potential targets were obtained from the literature collection and target prediction, and then these targets were used for pathway and process enrichment analysis. The top 20 enriched biological pathways and processes are shown in Figure 4. The enrichment results showed that apoptosis was an important biological process of BA in anti-cervical cancer. In the KEGG apoptotic pathway, 19 important targets were enriched, of which 12 were collected from the literature and 7 were predicted.
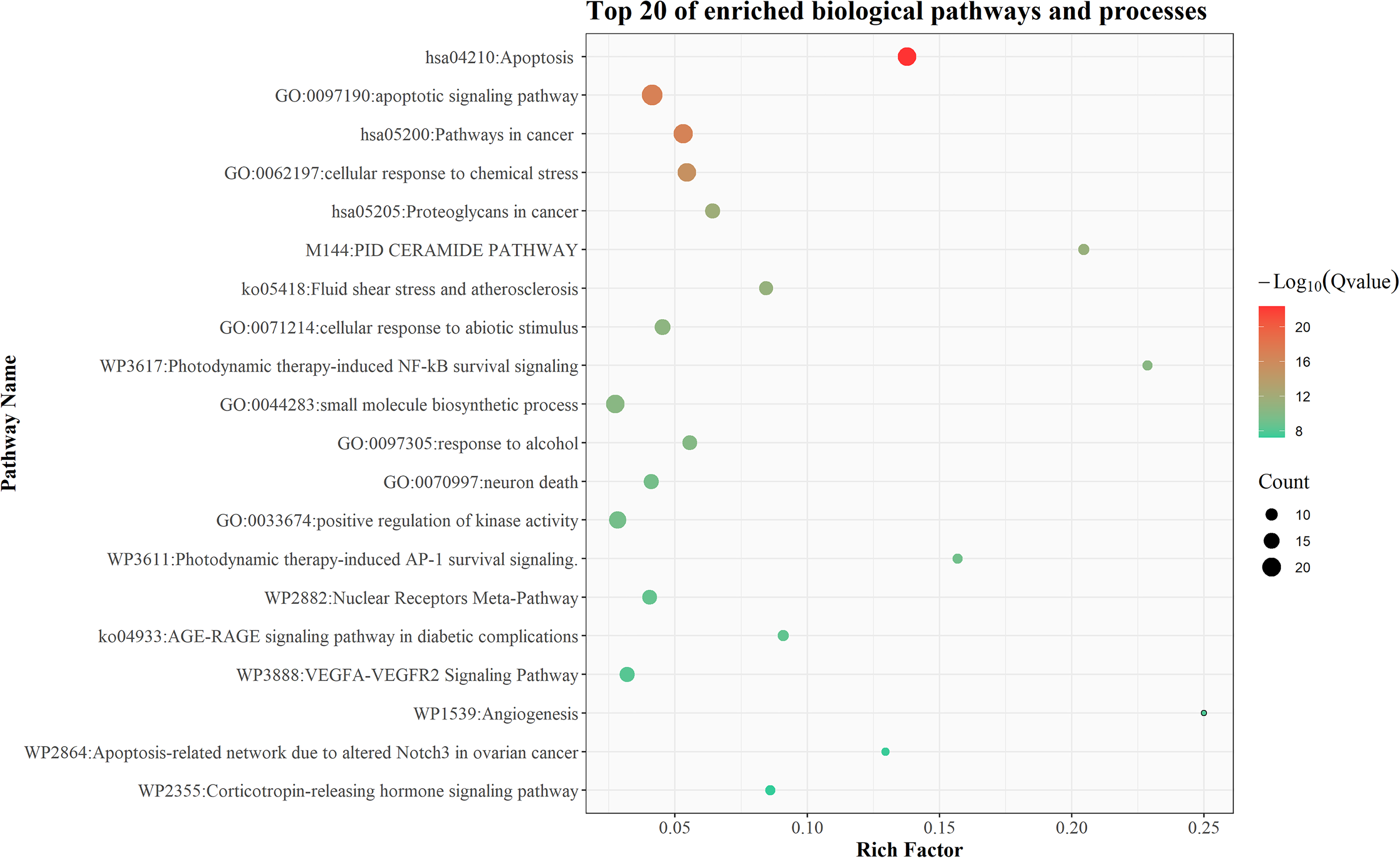
The top 20 enriched biological pathways and processes. The x-axis represents the enrichment factor, and the y-axis represents the enriched item. The bubbles are colored by Q-value, and the size is drawn by count.
Target Network and Potential key Targets
The 81 potential targets of BA against cervical cancer were constructed into a protein-protein interaction network. As shown in Figure 5, the network had a total of 81 nodes and 183 edges, of which 69 nodes form a large cluster, and 12 nodes were scattered. Each node represents a potential target for BA to regulate cervical cancer, and each edge represents the interaction between the 2 targets. The size of the node reflects the impact of the corresponding target on the biological process. In addition, the 12 orange nodes in the figure represent the enriched targets from the literature collection, the 7 green nodes represent the enriched targets from the target prediction, and the remaining gray nodes represent the targets that were not enriched.

Interaction network of 81 potential targets. The orange, green, and gray nodes represent collected enriched targets, predicted enriched targets, and other unenriched targets, respectively. The larger the node size, the greater the degree of the node.
As shown in the network analysis results of Table 1, there were 8 enriched targets with a node degree greater than 10, including 5 collected and 3 predicted. It could be seen that these 8 targets play a vital role in biological networks. Therefore, these 8 were considered potential key targets for BA to regulate cervical cancer. In our study, the 3 predicted targets (proteins encoded by CASP3, PARP1, and XIAP genes) have been verified by western blot experiment.
The Network Analysis Results of Enriched Potential Targets.

Effect of BA on the Expression of c-Casp-3, c-PARP, XIAP, and CytC
Western blot analysis demonstrated that after treatment with different concentrations (IC10, IC50, and IC90) of BA for 48 h, the expression of cleaved-caspase 3 increased with BA concentration, but was almost unchanged at high concentration (Figure 6A and B); the expression of cleaved-PARP was unchanged at low concentration, but decreased at high concentration (Figure 6A and C); and the expression of XIAP and cytochrome c remain largely the same compared with the control group (Figure 6A, D, and E).

Effect of betulinic acid (BA) on the expression of cleaved-caspase 3, cleaved-PARP, XIAP, and cytochrome C. (A) Cleaved-caspase 3, cleaved-PARP, XIAP, and cytochrome C expression in SiHa cells after treatment with different concentrations (IC10, IC50, and IC90) of BA for 48 h were analyzed via western blotting. (B-E) The band intensities of cleaved-caspase 3, cleaved-PARP, XIAP, and cytochrome C were, respectively, quantified by normalizing to GAPDH. Compared with the control group, *P < .05.
Discussion
Cervical cancer treatment might involve radical hysterectomy, chemoradiation, or both. 2 Therefore, less treatment-related injury is very urgent. Preliminary results with novel natural products have shown promising results so far, and BA is a good example of low toxicity and high efficiency. Previous studies have shown that BA was highly effective against human cervical cancer cell lines, such as HeLa and SiHa.20,21 Our present study demonstrated that BA significantly reduced the viability of cervical cancer SiHa cells in a dose- and time-dependent manner. In fact, the inhibition of SiHa cells under BA exposure was in good agreement with a previous study. 20 However, how BA inhibits the proliferation of SiHa cells at the molecular level is still a question worthy of interest.
Target identification is an important part of research on the internal mechanism of action. In this study, the potential targets of BA against cervical cancer came from literature collection and target prediction. From the literature, collection is known and reliable targets can be obtained, and target prediction can explore the new potential mechanisms. Here, we obtained 50 collected targets and 32 predicted targets, but the mechanism was still unknown. Therefore, we conducted an enrichment analysis of these potential targets. The results showed that induction of apoptosis was an important biological process of BA in anti-cervical cancer. In order to understand how BA induces apoptosis, the importance of these enriched targets needed to be further evaluated to determine the key regulatory sites of BA. Fortunately, network analysis can help us quickly find these potential key targets. In network analysis, a high degree node is generally considered to be important because of its extensive connectivity. 27 Based on this, the 8 enriched targets were regarded as potential key targets for BA-induced apoptosis in cervical cancer SiHa cells. They were 5 collected targets encoded by TNF, CASP8, AKT serine/threonine kinase 1 (AKT1), BCL2, and LMNA genes and 3 predicted targets encoded by CASP3, PARP1, and XIAP genes, respectively.
The 3 predicted potential key targets were further validated by western -blot analysis. Among them, caspase 3 is the most common molecule involved in apoptosis, 28 which is usually cleaved into cleaved-caspase 3 to produce activity in mammalian cells.29,30 Our results revealed a significant increase in the expression of cleaved-caspase 3 after treatment with BA. However, the levels of cleaved-caspase 3 did not increase at high BA concentration (IC90). PARP1 is a DNA damage sensor, 31 and participates in various DNA repair pathways and in the maintenance of genomic stability. 32 Cleaved-PARP is the hallmark of apoptosis, which is produced by activated caspase 3 to cleave PARP1.33,34 To our surprise, there was hardly any change in the level of cleaved-PARP when we treated SiHa cells with BA at the IC10 concentration, but this was decreased at higher BA concentrations (IC50 and IC90). These unexpected results prompted us to consider further whether BA acted on SiHa cells through more than apoptosis. In fact, some views showed that cell death could be classified into 2 major categories: apoptosis and necrosis. 35 Therefore, our results seem to suggest the involvement of necrosis after treatment with high concentrations of BA. The current findings were much in agreement with those of Jian Ping Yuan et al, who demonstrated the coexistence of necrosis and apoptosis in HeLa cells. 36 Also, some studies have indicated that PARP1 could repair DNA if low damage occurs, be cleaved through caspase-3 activation when apoptosis occurs, and become over-activated, resulting in ATP/NAD depletion and necrosis if high damage occurs. 37 This may make sense of the reduction of cleaved-PARP, because when the concentration of BA was too high, it did not pass through the apoptosis of the CASP3/PARP axis. XIAP is an extremely potent suppressor of apoptosis, and these effects are partly mediated by its ability to suppress caspases. 38 However, the present study indicated that the inhibitory effect of BA was independent of XIAP, because the expression of XIAP was almost unchanged within a series of concentrations of BA incubation.
Inducing cell apoptosis may be an important process of BA against cervical cancer. As a multi-target molecule, BA probably regulates multiple sites of SiHa cell apoptosis. As shown in Figure 7, apoptosis can be triggered in a cell through either the caspase-mediated extrinsic or intrinsic pathways. 39 The extrinsic apoptotic pathway is initiated by the interaction between the cell surface exposed death receptors and their specific ligands. 40 Tumor necrosis factor-alpha (TNF-α) is one of the initiation factors of the extrinsic apoptosis pathway.41,42 BA can significantly improve the level of TNF-α in cervical carcinoma U14 tumor-bearing mice. 19 The intrinsic apoptotic pathway is mediated by disruption of the mitochondrial outer membrane permeability (MOMP) leading to the diffusion of cytochrome c. 43 Apoptosis regulator Bcl-2 is an important inhibitory molecule of Bax-induced MOMP.44,45 BA can significantly downregulate Bcl-2 and upregulate Bax expression in cervical cancer cells.19,21,46 AKT1 is a bridge molecule to connect the survival signal and intrinsic apoptosis,47-49 and it enables cells to withstand apoptotic stimuli by indirectly promoting the activity of Bcl-2. 50 BA can participate in the intrinsic apoptosis of cervical cancer by suppressing AKT1 phosphorylation. 18 These 2 apoptotic pathways then activate initiator caspases separately to amplify the caspase signaling cascade, and converge at the level of effector caspases.51,52 Caspase-8 and 9 are the initiator caspase of extrinsic and intrinsic apoptosis, respectively.53,54 BA can activate the effector caspases in cervical cancer cells through enhancing cleavage and expression of caspases-8 and 9.18,19,55 The effector caspases are responsible for the proteolytic cleavage of a broad spectrum of cellular targets, which ultimately leads to cell death. 56 A-type lamins (lamin A/C), which are target proteins responsible for effector caspase-3, are closely related to the integrity of the nuclear membrane, and the loss of which can lead to apoptosis.57,58 The expression level of cleaved lamins was increased significantly (P < .05) in BA-induced HeLa cells. 21 In addition, our western blot analysis also showed that within a certain concentration range, BA could regulate the level of the CASP3/PARP1 axis and induce apoptosis of SiHa cells. Although there was no evidence that these potential key targets were the actual targets of BA, the regulation of BA on these sites was obvious. This regulation by BA might be the result of direct action, or the synergistic effect of biological networks.

Process of betulinic acid (BA) inducing cell apoptosis for 48 h at IC50 concentration. The process of BA-induced apoptosis is a multi-molecule coordinated process. BA is not only a participant in either the extrinsic or intrinsic pathways, but also a regulator of apoptosis initiation and effector molecules. The blue and red proteins represent the collected and predicted targets, respectively. Asterisks indicate potential key targets, red indicates upregulation, blue indicates downregulation, and gray remains unchanged.
Conclusions
In short, the effect of BA on cervical cancer is noticeable. Combined with the identified potential key targets, we speculate that BA-induced apoptosis of cervical cancer SiHa cells is a multi-molecule coordinated process. In this process, BA is not only a participant in either the extrinsic or intrinsic pathways, but also a regulator of apoptosis effector molecules of the CASP3/PARP1 axis.
Footnotes
Data Availability Statement
The data from the databases used in this study are available at the following links, and the other data used to support the findings of this study are available from the corresponding author upon request. PubMed (https://pubmed.ncbi.nlm.nih.gov/); DisGeNET (https://www.disgenet.org/); OMIM (https://omim.org/); KEGG (https://www.kegg.jp/); TTD (http://db.idrblab.net/ttd/); RGD (https://rgd.mcw.edu/); GeneCards (https://www.genecards.org/); Metascape (https://metascape.org/); and String (![]() ).
).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Zhejiang Province (grant number LYQ20H280002).
