Abstract
Lipoxygenase (LOX)-mediated lipid hydroperoxides (LHPOs) are converted to physiologically active mediators. Thus, decreasing hydroperoxides is important for diminishing various health-related risks. In this study, decreasing compounds of LOX-catalyzed LHPOs were investigated and classified by mechanism using a modified triple LOX-indamine dye formation (IDF) method. The compounds 2,3-dibromo-4,5-dihydroxybenzyl methyl ether (1) and fucoxanthin (2) were isolated from algae as compounds that can decrease hydroperoxides. Along with previously isolated pheophytin a (3), a chlorophyll-related compound, and purchased nordihydroguaiaretic acid (4), a well-known lipoxygenase inhibitor, all the compounds were examined to determine the mechanisms responsible for decreasing LHPOs by using a modified triple LOX-IDF method. Compounds 1 and 4 were found to be LOX inhibitors, compound 2 was a decomposer of the produced LHPOs, and compound 3 was both an inhibitor and a decomposer of the produced LHPOs.
Keywords
Introduction
Lipid hydroperoxides (LHPOs) are generated through chemical and enzymatic processes.1,2 The chemical process is initiated with free radicals and reactive oxygen species (ROS). Free radicals withdraw hydrogen radicals from lipids to generate lipid radicals. Subsequently, molecular oxygen is incorporated to make a peroxyl radical, and then the radical is converted to racemic LHPO by withdrawing hydrogen radical from another lipid.2,3 ROS-like singlet oxygen is also incorporated into lipids to form various racemic LHPOs.1,2 The enzymatic process is triggered by various lipoxygenases (LOXs) and an α-dioxygenase.1,2 They catalyze enantiomeric incorporation of molecular oxygen into lipids at various positions. The produced LHPOs are converted to physiologically active mediators, such as leukotrienes, which are involved in inflammatory diseases. 4 LHPO-derived products such as aldehydes and polymeric materials show cytotoxic and genotoxic effects. 2 Lipid peroxidation products exist in human atherosclerotic lesions and act as signaling messengers. Thus, decreasing produced LHPOs is important for diminishing various health risks.
Decreasing LHPOs is achieved by their reduction and conversion to adducts, anti-oxidation of lipids, and/or inhibition of the LOX reaction.2,5 LHPOs are converted to lipid hydroxides (LHOs) or other adducts by enzymatic and nonenzymatic reactions. The enzymes glutathione peroxidase5,6 and peroxiredoxin5,7 catalyze the reduction in LHPOs to their corresponding LHOs while glutathione S-transferase 8 catalyzes the formation of glutathione adducts. These enzymes help to quench hazardous ROS that is biologically harmful. Additionally, LHPOs are decreased by enzymatic and nonenzymatic conversion to aldehydes, ketones, divinyl ethers, α- and γ-ketols, and carotenoid adducts.1,9 Most of these conversion products belong to the oxylipin family and are known as signaling molecules. 10
LOX inhibitors have been isolated from terrestrial plants11,12 and algae.13‐16 Terrestrial plant-derived inhibitors are classified into various groups such as fatty acids, polyphenols, amides, and polyacetylene. In our previous search for LOX inhibitors derived from algae, algal extracts were screened using a LOX-indamine dye formation (IDF) method.17,18 We have isolated two highly unsaturated hydrocarbons, (6Z,9Z,12Z,15Z,18Z)-1,6,9,12,15,18-henicosahexaene and (9Z,12Z,15Z,18Z)-1,9,12,15,18-henicosapentaene, 15 a chlorophyll-related compound, pheophytin a, 15 and a phlorotannin, fucophlorethol C, 16 and identified them as LOX inhibitors by employing the conventional LOX-IDF method. The compounds obtained by our group were chemically diverse. Therefore, it was important that the compounds identified from the results of the conventional LOX-IDF method were either confirmed as LOX inhibitors or were determined to be involved in the decomposition of the produced LHPOs or indamine dye.
In the present research, a known bromophenol, 2,3-dibromo-4,5-dihydroxybenzyl methyl ether (
Results and Discussion
Isolation of LHPO-Decreasing Compounds
Compounds
Determination of the Mechanism Responsible for Decreasing LOX-Catalyzed LHPO Using a Modified Triple LOX-IDF Method
All the compounds examined showed the ability to decrease LOX-catalyzed LHPOs. A modified triple LOX-IDF method was used to determine the detailed mechanism responsible for the decrease in LOX-mediated LHPOs. The results are shown in Figure 2. None of the compounds

Compounds that decrease lipoxygenase-catalyzed lipid hydroperoxides. (1) 2,3-dibromo-4,5-dihydroxybenzyl methyl ether; (2) fucoxanthin; (3) pheophytin a, derived from algae, and (4) nordihydroguaiaretic acid, a well-known lipoxygenase inhibitor.
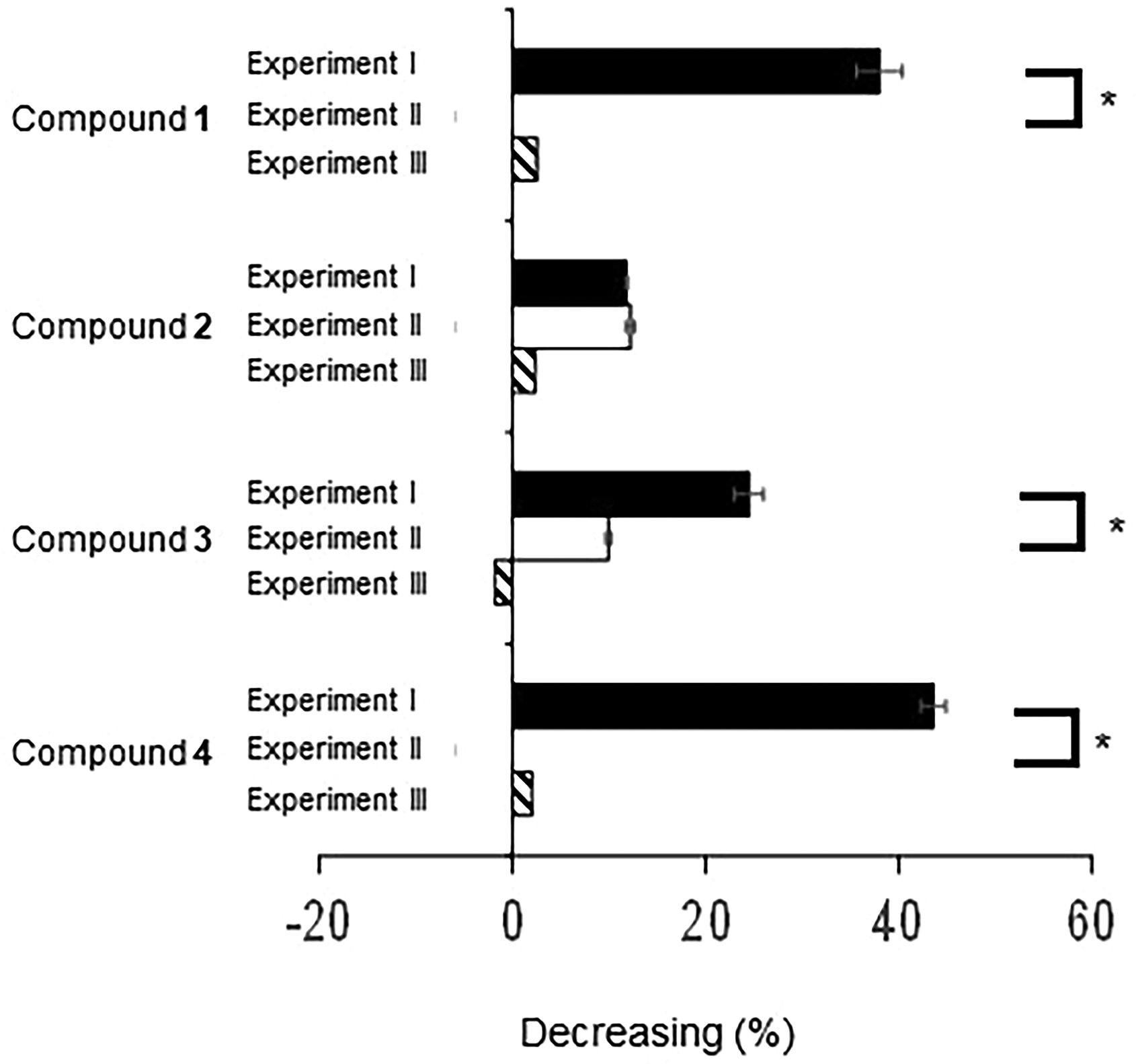
Determination of the decreasing ratios of compounds
Phenolic compounds
Although fucoxanthin
Pheophytin a (
LOX-catalyzed LHPOs are trigger compounds of biosynthetic pathway cascades of leukotrienes. It is important to decrease the production of LOX-catalyzed LHPOs and also reduce already produced LHPOs for the prevention of leukotriene-related diseases. The LOX-A234 method can screen only for LOX inhibitors, not LHPO decomposers. The conventional LOX-IDF consists of 3 steps. In the first enzyme reaction step, soybean LOX catalyzes the oxidation of linoleic acid to the corresponding LHPO. In the second indamine dye formation step, the produced LHPO is converted to an indamine dye in the presence of 3-methyl-2-benzothiazolinone hydrazone (MBTH) and 3-dimethylaminobenzoic acid (DMAB). In the final measurement step, the resulting solution is homogenized by adding sodium dodecyl sulfate (SDS) and measured its absorbance. Thus, the conventional LOX-IDF method can screen for both LOX inhibitors and decomposers of LHPOs, but not distinguish between the inhibitors and the decomposers. Therefore, both the previous methods cannot determine the detailed mechanism(s) of active compounds. However, the novel modified triple LOX-IDF method can screen for and distinguish between the inhibitors and the LHPO, already generated by LOX, decomposers simultaneously. The modified method would become a powerful tool for screening LOX inhibitors and LHPO decomposers (reductants) because the compounds showing decreasing LHPO are rapidly determined decreasing mechanisms.
Conclusion
Two compounds, bromophenol (
Experimental
General
Soybean LOX (lipoxidase Type I-B), linoleic acid, and MBTH were purchased from Sigma-Aldrich Chemical Company (St. Louis, MO, USA), Kanto Chemical Co., Inc. (Tokyo, Japan), and Nacalai Tesque, Inc. (Tokyo, Japan), respectively. Pheophytin a, DMAB, and Tween 20 were purchased from Wako Pure Chemical Industries (Osaka, Japan). Other reagents were purchased from Kanto Chemical Co., Inc. and Wako Pure Chemical Industries. High-performance liquid chromatography (HPLC) was performed using a Shimadzu LC-10ATVP chromatograph equipped with a Mightysil RP-18 column (internal diameter 4.6 mm, length 250 mm, Kanto Chemical Co., Inc.) and a Shimadzu SPD10-AVVP UV-VIS detector. Mass spectrometry (MS) was recorded on a JEOL JMS-T100GCV mass spectrometer (Akishima, Japan). Thin-layer chromatography was performed on TLC silica gel 60 F254 plates (Merck KGaA, Darmstadt, Germany). Nuclear magnetic resonance (NMR) spectra were recorded on a Bruker AMX500 NMR spectrometer (Karlsruhe, Germany). Absorbance was measured on a JASCO V-630 spectrophotometer (Hachioji, Japan).
Isolation and Identification of Compounds That Decrease LOX-Mediated LHPOs
The red alga C. pinnulatus (1340 g, air-dried) and the brown alga S. confusum (1070 g, air-dried) were collected from the coast of Hakodate, Japan (41°56′11.5″N, 140°56′56.9″E). They were washed with tap water and independently extracted with methanol (ca. 5 L) at room temperature for several days.
The methanolic extract (36.5 g) of C. pinnulatus was separated by organic solvent partitioning to obtain an ethyl acetate soluble fraction (663 mg). The fraction was chromatographed on silica gel (diameter 3.0 cm, height 30 cm) eluted with n-hexane:ethyl acetate (1:3, vol/vol) followed by preparative TLC developed with chloroform:methanol (9:1, vol/vol) to obtain a semi-purified fraction. This fraction was finally purified by HPLC eluted with acetonitrile: 0.1% formic acid (6:4, vol/vol) as the eluent to afford compound
The methanolic extract (29.6 g) of S. confusum was separated into an ethyl acetate soluble fraction (1.30 g) and chromatographed on silica gel (diameter 3.5 cm, height 45 cm) eluted with chloroform:methanol (39:1, vol/vol) to obtain a semi-purified fraction. This fraction was finally purified by HPLC eluted with acetonitrile: 0.1% formic acid (9:1, vol/vol) as the eluent to afford compound
Modified Triple LOX-IDF Method
Preparation of Reagent Solution
Solutions A and B and the substrate solution in the LOX-IDF method were prepared as described previously 15 with slight modification. Solution A contained 20 mM DMAB in 100 mM borate buffer (pH 9.0), and solution B contained 10 mM MBTH and 5.0 mg/mL hemoglobin in 50 mM phosphate buffer (pH 5.0). The substrate solution was prepared from 140 mg linoleic acid and 280 mg Tween 20 in 1 N NaOH (0.60 mL) and water (5.0 mL) and then was mixed with a Pasteur pipette. The mixture was made up to 20.0 mL with water. The substrate solution was divided into 2 mL aliquots in plastic vials, flushed with argon gas, and stored at −30 °C until use. Test compound was dissolved in methanol at concentration of 1.00 mg/mL.
Experiment I (the Conventional Method)
Experiment I was the conventional method 15 by the addition of the test compound before pre-incubation. Test compound solution (0.020 mL) was added into solution A (0.910 mL) and 128000 U/mL soybean LOX in 100 mM borate buffer (pH 9.0) (0.020 mL). This solution was preincubated at 27.5°C for 5 min. The LOX reaction was initiated by adding the substrate solution (0.050 mL) under the final test compound concentration of 20 μg/mL, incubated at 27.5°C for 5 min, and terminated by adding solution B (1.00 mL) to form the colored product. After 5 min, 1% SDS (1.00 mL) was added to the reaction mixture, and it was mixed vigorously. The absorbance at 598 nm was measured and compared with that obtained in the experiment performed by adding methanol instead of adding test compound solution.
Experiment II
Experiment II was the modified method by adding the test compound after the LOX reaction. The experimental procedure was similar to that of experiment I except each test compound was added after the LOX reaction at the same time of adding solution B. The absorbance at 598 nm was measured and compared with that obtained in the experiment performed by adding methanol instead of adding test compound solution.
Experiment III
Experiment III was the modified method by adding the test compound after LOX-IDF formation. The experimental procedure was similar to that of experiment I except each test compound was added after the IDF reaction. The absorbance at 598 nm was measured and compared with that obtained in the experiment performed by adding methanol instead of adding test compound solution.
Estimation of Mechanism
The 3 decreasing (%) values, DExI, DExII, and DExIII, were calculated from the decrease of the absorbance of the indamine dye solution compared with those of the control solutions as equation (1):

The modified triple lipoxygenase-indamine dye formation method. Enzyme, soybean lipoxygenase in borate buffer (pH 9.0); substrate, linoleic acid, Tween 20, sodium hydroxide in water; solution A, 3-dimethylaminobenzoic acid (DMAB) in borate buffer (pH 9.0); solution B, 3-methyl-2-benzothiazolinone hydrazone (MBTH) and hemoglobin in phosphate buffer (pH 5.0); SDS, sodium dodecyl sulfate in water. Decreasing ratio (%) values, DExI, DExII, and DExIII, represent as decreasing ratios (%) in experiments I, II, and III, calculated by equation (1).
Statistical Analyses
The data obtained for the modified triple LOX-IDF method represent the mean ± standard error of the mean. A significant difference between experiments I and II was determined by the Student t test (P < .05).
Supplemental Material
sj-pptx-1-npx-10.1177_1934578X221115249 - Supplemental material for Classification of Marine Algae–Derived Compounds by the Mechanism Responsible for Decreasing Lipoxygenase-Catalyzed Lipid Hydroperoxides
Supplemental material, sj-pptx-1-npx-10.1177_1934578X221115249 for Classification of Marine Algae–Derived Compounds by the Mechanism Responsible for Decreasing Lipoxygenase-Catalyzed Lipid Hydroperoxides by Hideyuki Kurihara, Kenta Yachiyama and Chihiro Morimoto in Natural Product Communications
Footnotes
Acknowledgements
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

