Abstract
The aim of this study was to confirm the anti-inflammatory effect and explore the adverse effects and underlying mechanisms of Prunella vulgaris L., which has been extensively used for hundreds of years in East Asia. Network pharmacology studies predicted that glucocorticoids (GCs), GC-targeting molecules, and brain-derived neurotrophic factor (BDNF) were intensively involved in the anti-inflammation and glucose intolerance. To attest the effects and underlying mechanisms, C57 male mice were randomly divided into 5 groups, control (C), dexamethasone (Dex), water extract of P. vulgaris (PE 35 or 70 mg), and PE (70 mg) + mifepristone (PEM). After a 3-week treatment, acetic acid-induced writhing and hot plate tests confirmed the peripheral and central analgesic effects, respectively. Plasma GCs and BDNF were significantly increased. Coincidently, plasma pro-inflammatory cytokines, including IL1β, IL6, and IL10, were decreased by PE treatment, which were blocked by the application of mifepristone (P < 0.5). Western blots confirmed GC receptor (GR) translocation, and decreased cyclooxygenase 2 in the lumber spine by PE treatment. Food intake was impeded after a 4-week PE treatment, but the ratio of bodyweight gain to food intake was increased in a time-dependent manner. An intraperitoneal glucose tolerance test disclosed that PE treatment impaired glucose disposal in mice. Quantitative polymerase chain reaction (PCR) showed that hepatic GC-responsive genes such as GC-induced leucine zipper protein and glucose-6-phosphatase catalytic subunit 1 were up-regulated, and hypothalamic neuropeptide Y and agouti-related protein expressions were decreased by PE treatment. Hypothalamic BDNF was up-regulated, whereas hepatic BDNF was down-regulated. The regulation of these genes by PE was reversed by mifepristone administration. In conclusion, PE treatment plays analgesic and glucose regulation roles simultaneously through GC-induced signaling pathways, and P. vulgaris may provide a natural ligand of GR for the treatment of inflammation with glucose dysregulation.
Introduction
Prunella vulgaris L. is a perennial plant of the Labiatae (Lamiaceae) family. It has a broad range of beneficial effects including immune modulation, anti-viral, anti-allergy, anti-inflammatory, and antioxidant activity1-7 and has a wide distribution over the world. 3 Studies using aqueous or organic fractions of P. vulgaris (PE) revealed similar effects such as anti-allergy, 8 anti-microbial,1,2 and anti-inflammation properties. 3 Aqueous extracts of PE were also reported to inhibit infection. 9 In addition, P. vulgaris is used, together with other herbal medicines, like Pinellia ternata Thunb., for some classic decoctions in Chinese medicine for dispersing hard lumps and abscesses in tissues, 10 or as an over-the-counter medication for hypertension. 11 For example, Xia Sang Ju formula, a Chinese drug and herbal tea made up of Prunellae spica, Mori folium, and Flos Chrysanthemi Indici, has commonly been used in the clinic for fever, headache, and sore throat since 1814. 11 The aerial part of P. vulgaris is also directly used as tea in some areas of China to relieve a variety of ailments. 12
The widespread clinical use of P. vulgaris indicates that it may have numerous components, or some potent components to activate critical regulatory pathways. To date, chemical analysis found that P. vulgaris contains at least 60 active constituents including oleanolic acid, rosmarinic acid, rutin, ursolic acid, tannins volatile oil, and β-carotene.2-5,13 These components can be obtained from aqueous and ethanol/methanol extracts, but are not limited to any single extract.1-3,13 The identification of these compounds links them to the treatment of 86 related diseases, some of which have been validated (https://tcmsp-e.com). For example, the high concentration of oleic acid (30 μM) stimulates glucagon secretion in the presence of glucose at 2.8-8.3 mM. 14 The secretion of glucagon is modulated by brain-derived neurotrophic factor (BDNF) activity, 15 which is one of the main factors associated with metabolic regulatory function. 16 Oleanolic acid may bind to the glucocorticoid (GC) receptor to activate GC-signaling pathways. 17 Similarly, rosmarinic acid, a featured compound in plants of the Labiatae, is able to attenuate repetitive viral infection stress through up-regulation of the GC receptor (GR).18,19 In addition, stigmasterol in P. vulgaris may be able to interact with the GR playing anti-inflammatory effect, 20 but the effects and mechanisms are not fully clarified.
GCs are well known as stress hormones generating an increased blood glucose level to provide enough energy rapidly to respond to environmental challenge, while producing analgesic, anti-nociceptive, depressive, and many other therapeutic effects. 21 They induce nuclear translocation of GR in target cells, and the activated GR turns on transcription of the genes containing GC response elements (GRE) to fulfill the above functions. 22 Therefore, they are widely used in clinical practice and have positive effects on the control of inflammation, metabolism, and mediation of immune response at physiological concentrations. However, prolonged GC administration is often accompanied by severe side effects characterized by the development of diabetes and iatrogenic Cushing syndrome, increasing the risk of serious opportunistic infections, drug resistance, and immune system disorders.23,24
Side effects of the clinical use of P. vulgaris have been gradually recognized by some physicians in China, though they are not well documented. These side effects include the loss of appetite, weakness, and diarrhea after use for several weeks. Some patients may have immune system disorders, or a gradual loss of kidney/liver function (private correspondence with Professor Shuke Cui, Henan University of Chinese Medicine). To our knowledge, there is limited research on the causal factor(s) and underlying mechanisms for these side effects. Our preliminary studies using network pharmacology approaches revealed that at least 547 genes are relevant to both analgesic and glucose intolerance, which may help to find clues to understand the side effects of P. vulgaris application. However, only 6 constituents of P. vulgaris have been identified to target 8 molecules associating with analgesic effect, and 6 that target 9 molecules relevant to glucose intolerance.
The current investigation adopted network pharmacology approaches to predict the involved molecules, including GCs, GC-responsive genes, and BDNF when aqueous extract of P. vulgaris spica was applied for analgesic purposes. Experimental studies confirmed that PE treatment significantly increased plasma GC level, and induced GR translocation, which consequently up-regulates GC-induced leucine zipper protein (GILZ) and glucose-6-phosphatase catalytic subunit 1 (G6PC1) for anti-inflammation and glucose intolerance, respectively. PE treatment also raised plasma BDNF level with the subsequent down regulation of hypothalamic neuropeptide Y (NPY) and agouti-related protein (AgRP). The coincidental elevation of plasma BDNF and GCs indicates that PE treatment may relieve the symptoms through fine tuning the therapeutic effects and adverse reactions.
Materials and Methods
Reagents and Apparatus
The assay kits for the evaluation of plasma IL1β (EH3655), IL6 (EH11330), and IL10 (EH3264) were purchased from Wuhan Fine Biotech Co., Ltd (Wuhan, China); mouse GC (CK-E10092M) and BDNF (CK-E20084M) ELISA kits from Shanghai Jijin Chemistry Technology Co., Ltd (Shanghai, China); dexamethasone (50-02-2), mifepristone (RU-486) (84371-65-3), 2-hydroxypropyl-β-cyclodextrin (H107), and protease inhibitor cocktail (P-8340) from Sigma Aldrich (St. Louis, MO, USA); glucose from Tianjin Kemiou Chemical Reagent Co. (Tianjing, China); acetonic acid from Fengchuan Chemical Reagent Technologies Co. (Tianjing, China); Trizol reagents from Invitrogen (New York, CA, USA); RevertAid First strand cDNA synthesis kits from Thermal Scientific (Waltham, MA, USA); cytoplasmic and nuclear extract kits from Active Motif (Shanghai, China); Primary COX-2 (sc-376861), GR (sc-1004), GAPDH (sc-293335), and horseradish peroxidase-conjugated secondary antibodies from Santa Cruz Biotechnology (Dallas, TX, USA); and primers (Table 1) from Synbio Technologies (Suzhou, China). All chemicals and reagents were of analytical grade.
Primers Used in Real-Time PCR.

YLS-1A apparatus was from Yiyan Technology (Jinan, China), Rotarod apparatus (ZB-200) from Chengdou Technology & Market (Chengdou, China), microplate readers from BioTek Instruments (Winooski, VT, USA), and 2720 thermal cycler from Life Technologies (Foster City, CA, USA). Real-time polymerase chain reaction (PCR) was performed using ViiA 7 Real-Time PCR System from Thermal Fisher Scientific (Waltham, MA, USA).
Network Pharmacology Studies
Modified network pharmacology approaches were adopted from our previous work 25 to predict the mechanisms of analgesic and metabolic side effects of P. vulgaris. Briefly, the target molecules and related diseases were obtained from https://tcmsp-e.com. Genes associating with analgesic or glucose intolerance were acquired from the database in Genecards. A list of common genes that are simultaneously involved in analgesic and glucose intolerance was attained by a comparison of the 2 sets of genes. A list of validated target genes relevant to the effect of P. vulgaris on both analgesic and glucose intolerance was extracted by a comparison of P. vulgaris target genes with the list of analgesic and glucose tolerance common genes. Genes with high relevant scores but rare evidence to be relevant to PE treatment, like BDNF and GC signaling molecules, were picked up for this investigation. Together with experimental evidence, a visualized construct showing the connection of PE gradients and target genes associating with the therapeutic and side effects was drawn using Cytoscape. An updated protein–protein interaction (PPI) network exhibiting the connections of the genes was produced using https://string-db.org/ database. The obtained PPI data were applied for GO analysis to demonstrate the frequency of the molecules in the network. KEGG analysis was applied to show the significantly enriched signaling pathways for the gene content in the genome provided by the KEGG PATHWAY database.
All data cleaning and the comparisons were performed using R associating packages. Values of P < 0.05 and q < 0.05 were considered to be significant.
Experimental Animals
Healthy C57 black mice (8-week old, bodyweight 23 ± 2 g) and food were obtained from the Charles River Laboratories (Beijing). Mice were kept in a 12:12 hr light-dark cycle (6:00 AM to 6:00 PM) and fed with standard commercial chow and distilled water ad libitum. The room temperature was maintained at 22 ± 0.5 °C and about 40% relative humidity. The experimental protocols (NITAEE17-0014) were approved by the Animal Experimentation Ethics Committee at Nanyang Institute of Technology under the guidelines of the National Medical and Health Research Council of China. All protocols were designed to minimize animal discomfort. Only naive mice were used for behavioral tests, unless pointed out.
Preparation of P. vulgaris Water Extract and Animal Treatment
PE preparation was made according to the description by Mohaddese et al 2 P. vulgaris was cultured on the campus of Nanyang Institute of Technology. Spikes were obtained and dried when mature during 2018-2020. Three-gram dried spike of P. vulgaris was ground, soaked in double-distilled water (200 mL) for 1 h, and then incubated in a 95 °C water bath for 2 h, followed by filtering through Whatman No.1 filter paper. To avoid interference from intragastrical administration to behavioral tests, mice were kept in groups in a home cage, and fed with PE at 2 different concentrations ad libitum. According to the amount drunk daily (5 g/day), the daily intake of spike was about 70 mg per mouse in the PE70 group, and 35 mg for each animal in the PE35 group.
After acclimatizing to the environment for 3-5 days, mice were pre-selected using the hot plate assay to eliminate animals with abnormal nociception before being randomly divided into 5 groups: control (Con. fed with water), PE35 (fed with 35 mg/day), PE70 (fed with 70 mg/day), PE70 + RU-486 (PE + M) (2.5 mg/kg/day, diluted in PE), and dexamethasone treatment (Dex) (IP, 1 mg/kg/day) groups (n = 8). PE35, PE70, and PE + M groups were pretreated with either PE35 or PE70, respectively, for 2 weeks before the application of mifepristone (RU-486) for 1 week. Dex was applied by IP injection 2 days before the start of the experiment and the last one was usually 1 h before tests. The non-toxic solubilizer, 2-hydroxypropyl-β-cyclodextrin was dissolved in water to serve as a negative control. A schematic figure (Figure 1) is given to illustrate the above experimental process.

Schematic representation of PE treatment protocol. Experimental groups were pre-treated with PE aqueous extract (35 mg and 70 mg) for 2 weeks; then, PE + M group was further treated with mifepristone (M) (2.5 mg/kg/day, diluted in PE). Control group was treated with 2-hydroxypropyl-β-cyclodextrin in water all the time. Positive control group was treated with control water until IP injection of dexamethasone (D) (1 mg/kg/day) on 19th day for 2 consecutive days. The last injection was 1 h before tests. Only naïve mice were used for behavioral tests. Mice were then anesthetized using ketamine/xylazine for blood sampling and then euthanized by cervical dislocation. Tissues were harvested for real-time PCR or Western blot. Mice were individually housed in regular cages for food intake for 4 weeks, which is not illustrated in the schematic diagram.
Acetic Acid-Induced Writhing Test
Peripheral analgesic effects of the aqueous extract of P. vulgaris were evaluated using the acetic acid-induced writhing method. Acetic acid (0.6%) was made from 37% acetic acid with saline. Mice were intraperitoneally injected with 0.1 mL/10 g of 0.6% acetic acid solution 21 days after PE treatments, and 1 h after the administration of either Dex (1 mg/kg) or saline. Thereafter, the mice were observed for concave abdomen, extension of trunks and hind limbs, and lifting of buttocks, indicating abdominal writhing. The number of writhes was counted for 30 min after 5 min of writhing induction. The percentage inhibition of writhing was calculated relative to the control group. At the end of the experiment, the mice were anesthetized, and euthanized by cervical dislocation.
Hot Plate Test for Thermal Nociception
Central analgesic activity of P. vulgaris was evaluated using the hot plate test. The test was performed between 8-10 using YLS-1A apparatus. Mice were habituated to the testing room overnight. They were then placed in a Plexiglas cylinder on a hot plate at 52 °C. Signs of nociceptive behavior were defined as shaking or licking of one of the hind paws or jumping. The latency to lick the hind paw with a 60 s cutoff latency was recorded.
Tail-Flick Measurements
The tail immersion-withdrawal test was conducted mainly according to the previous description, with certain modifications. 26 Mice were pretreated as described above, and allowed to acclimize to the laboratory environment for 2 days before testing. To restrain the mice for the test, 50 mL conical tubes were cut at the bottom to generate a 0.5 cm opening to allow the mouse to breathe freely, and an opening was cut into the cap for each tube to allow the tail access to the water bath. A digital water bath was set at 53 °C prior to test. Control mice were injected IP with either dexamethasone or control solution 1 h before the test. Approximately 3 min before the test, mice were allowed to crawl into the precut 50 mL tubes. The tail was marked with ink at 4 cm from the distal tip. The mouse was held to allow the distal 4 cm of tail to be submerged in the water bath. The submersion time was limited to 30 s to prevent tissue damage. The movement of the tail out of the water was recorded as the tail-flick time.
Rotarod Test
The rotarod test was applied to assess whether or not PE affects motor coordination and balance. Mice were trained twice for up to 5 min before data were collected (spaced 1 h apart to minimize fatigue, 15 and 30 rpm for training, and 30 rpm for testing). The time the animal spent on the rotating rod during each trial was recorded. The rotarod test was carried out 21 days after PE treatment. The chambers were cleaned with water between trials.
Food Intake and Body Weight Study During Treatment
Food intake and body weight were followed as in our previous description.27,28 Briefly, mice were individually housed in regular cages. Body weight and food intake were determined every 3 days through the experiments between 8-10 am. Pre-weighed food was placed in the food hoppers and measured on a per-cage basis. Food intake was expressed as g consumed per day. The body weight gain during the experiment was determined by subtracting the body weight right before the experiment.
For the appetite test, after a 21-day treatment, mice were fasted overnight with either control solution or PE. Body weight and food intake were recorded at 0.5, 1, 4, and 24 h after food was returned. Average food intake and body weight gain of each group were expressed. The ratio of body weight to food intake was calculated for the indicator of calorie disposal as body weight.
Intraperitoneal Glucose Tolerance Test
The intraperitoneal glucose tolerance test was performed 21 days after PE feeding. After overnight fasting, plasma glucose from the tail vein was measured using a One Touch Ultraeasy Glucose Test Strip (Shanghai, China) (time 0). Then, animals were injected with an intraperitoneal injection (2 g/kg) of glucose. Tail vein glucose was measured at 15, 30, 45, and 60 min after injection. Plasma glucose levels were normalized to those at time 0, and reported as relative change (%). Areas under the curves were calculated using Graphpad Prism 8.0.
Blood Sampling and ELISA Tests for GC, BDNF, and IL1β, IL6, IL10
After a 3-week treatment, periorbital blood samples were taken and placed in ice-cold EDTA containing tubes, followed by a 3000 rpm centrifuge for 30 min. Supernatants were transferred into well-labeled clean tubes, and saved in a −80 °C freezer until ready for tests. Plasma levels of IL1β, IL6, IL10, BDNF, and GC were measured using mouse ELISA kits, according to the manufacturer's instructions. OD values were detected at 450 nm.
Mice were then euthanized by cervical dislocation. Tissues were harvested and prepared for either real-time PCR or Western blot.
Western Blot Analysis on Cyclooxygenase 2 Expression in the Lumber Spine and GR Translocation in Hepatic Cells upon PE Treatment
Sample collection, preparation, and the Western blot process were followed as in previous work. 27 Briefly, the liver and lumber spine (L1-L5) were dissected, and sonicated in cell lysis buffer with protease inhibitor cocktail. After a brief centrifugation (3000×, 5 min), the soluble part was measured for protein concentration using the Nanodrop. Total protein (15 ng) was uploaded from each well for analysis. After SDS-PAGE, proteins were transferred to a polyvinylidene difluoride membrane for incubation with antibodies against COX-2, GR, and GAPDH following the product protocols, respectively. Horseradish peroxidase-conjugated secondary antibodies (Santa Cruz Biotechnology) bound to the primary antibodies were detected by an enhanced chemiluminescence reaction.
Preparation of the cytoplasmic and nuclear extracts from the liver was performed following the protocol with the kit (Active Motif, China). Briefly, the fresh liver was dissected, properly diced, and soaked in 3 mL complete hypotonic buffer with phosphatase and protease inhibitors, followed by homogenization (∼15 strokes) until single a cell slurry was obtained. The cell suspension was incubated for 15 min on ice, and then centrifuged at 850 × g at 4 °C for 10 min. Pellets were collected, and re-suspended in 300 μL hypotonic buffer by pipetting up and down several times. After incubation for 15 min, 15 μL of detergent was added to each tube. Samples were vortexed for 5-10 s, followed by 14 000 × g centrifugation for 30 s at 4 °C. The supernatant, which is the cytosolic portion, was collected. The pellet contained nuclei and debris.
The nuclear pellet was re-suspended in 30 µL complete lysis buffer through pipetting followed by 30 min incubation on ice with 150 rpm rocking. Then the sample was vortexed for 30 s, and centrifuged at 14 000 × g at 4 °C for 10 min to yield the nuclear fraction.
The yield fractions from each step were inspected under a microscope for optimizing the process. Some of the samples were isolated multiple times and combined to obtain enough protein for Western analysis. The intensity of the bands was measured using NIH image, normalized to GAPDH, and expressed as the relative level to control samples.
RNA Isolation, Reverse Transcription, and Real-Time PCR
After behavioral tests, mice were anesthetized using ketamine/xylazine (72/13 mg/kg, i.p.). The liver and the hypothalamus were collected. Total RNA was isolated using the rizol method, according to the manufacturer's instructions. The reverse transcription reaction, target cDNA quantification, and data analysis were performed as in our previous work.16,27
Real-time PCR was performed in duplicates using 50 ng cDNA for each well. Changes in mRNA expression level were normalized to β-actin level and calculated using the ΔΔCt method. Results were expressed as relative fold change ± SEM.
Data Analysis
Means and standard errors (SE) of means were calculated for all experimental groups. Data were subjected to 1-way analysis of variance, followed by Tukey's post hoc test to identify significant differences relative to the controls. Data were analyzed and plotted using Graphpad Prism 8.0 and presented as means ± SE.
Results
Active Ingredients in P. vulgaris, Target Molecules, and Enriched Biological Processes Relevant to Analgesic and Glucose Intolerance
To help understand the relationship between analgesia and metabolic dysregulation during PE treatment, common genes relevant to analgesia and glucose intolerance were extracted from the database in Genecards. A total of 2954 in the glucose intolerance-related module and 1069 in the analgesic-related module were yielded, respectively, among which 547 genes were common to both (data not shown). That is to say, 51.2% analgesia-related genes are associated with the development of glucose intolerance. However, only 8 out of 547 genes appeared as P. vulgaris target genes in the TCMSP database, namely GCG, CCK, PYY, RHO, THBD, INS, TYR, and MPO (Figure 2A). The range of their relevance scores for glucose intolerance was 79.3(INS)-3.4(RHO), and 5.1(CCK)-0.44(THBD) for analgesia. These target genes were associated with 6 compounds in P. vulgaris (oleic acid, rutin, quercetin, palmitic acid, p-coumaric acid, and luteolin) (Figure 2A).

Mechanisms of the analgesic effect and concurrence of glucose intolerance of P. vulgaris administration. A. Construction of compound-target molecule-disease network, visualizing current validated ingredient–target interaction (dots in royal blue; edge, solid grey) and predicted connection information (dots in aquamarine for molecules that were able to connect with P. vulgaris ingredients; dots in cyan represents molecules that do not directly connect to ingredients in the current database; edge, dot grey); P. vulgaris ingredients shown in light green; Prunella shown in dark green: anal glucoin represents analgesic and glucose intolerance, shown in red. B. A PPI network showing current validated target molecules (inner circle dots) and predicted molecules (outer circle dots) by current investigation, where nodes and edges represent proteins and their interactions. C. The degrees of hubs (≥5) in network of Figure 2B. D. KEGG analysis showing significantly enriched signaling pathways of the validated and predicted proteins (P < 0.05, q < 0.05). The calibration scale represents the adjusted p value.
Beyond the above 8 validated target molecules, the molecule with highest relevance score for glucose intolerance was G6PC1 (89.1), a rate-limit enzyme for hydrolyzing glucose 6-phosphate resulting in the elevation of plasma glucose level. G6PC1 is primarily expressed in the liver and the kidney and is up-regulated by GCs through the induction of binding with GR (or NR3C1) to its upstream GC responsive element (GRE) (−231 to −129). 29 GR is one of the 547 target genes with a relevance score of 9.4 for glucose intolerance, and 0.6 for analgesia, and may be activated by Δ7-stigmasterol (an analog of stigmasterol) and oleanolic acid in P. vulgaris to initiate the downstream signaling.17,30 GILZ, SGK, and GSK are GC-responsive genes, 31 and they are also in the module of analgesia-glucose intolerance. Their relevant scores are 2.04, 3.4, and 1.3 for analgesia, and 13.4, 37.7 and 13.6 for glucose intolerance, respectively.
In addition, another very important regulatory molecule, BDNF, is also one of the common genes, with a relevance score of 2.6 for analgesic, and 9.3 for glucose intolerance, indicating that it may play an important role in anti-inflammation and glucose disposal. Some important cytokines, like IL1β, IL6, and IL10, but not limited to these, are common genes too. The above genes are also P. vulgaris compound targeting molecules.
To investigate further the relevance of the PE analgesic effect with glucose disposal, construction of a compound–target-disease network was completed with Cytoscape (version 11.0) (Figure 2A). The connections between all the involved proteins were visualized using String (version 11.0) (Figure 2B). GO enrichment analysis revealed that INS, AKT1, IL6, GCG, NPY, BDNF, GCR, AGRP, CCK, and PYY were hub genes within the above network (Figure 2C). These molecules actively evolved into 9 biological processes, including neuroactive ligand-receptor interaction, and insulin resistance signaling pathways through 13 enriched signaling pathways revealed by KEGG (P < 0.05, q < 0.05) (Figure 2D).
Analgesic Effect of PE Treatment and the Underlying Mechanisms
The peripheral analgesic effect of PE was evaluated through the acetic acid-induced writhing test and expressed as inhibition of writhing frequency and percentage relative to the control group. Both aqueous PE35 and 70 significantly reduced the number of writhes compared to the control group (P < 0.05; Figure 3A). Their inhibitory percentages were 28.5% and 55.8%, respectively (Figure 3B). The application of mifepritone diminished the PE-initiated analgesic effect to the control group level. Dexamethasone significantly decreased the analgesic response compared with the control group (P < 0.05). The decrease was about the same amplitude as that of PE70 treatment (Figure 3A, B).

Enhanced analgesic, Cox2 expression and GR translocation by PE treatment. A. Peripheral analgesic effect of PE using acetic acid-induced writhing test showing in counts (A), and inhibitory rates (B). C. Central analgesic effect of PE shown by hot plate test. D. Central analgesic effect of PE demonstrated by tail-flicking test. E. E-G: PE effect on the level of plasma cytokines IL1β (E), IL6 (F), and IL10 (G). H-J. PE regulatory effect on Cox2 level in the lumbar spine. K-M: PE regulatory effect on Cox2 level in the liver. N-O: GR content in cytosol and nucleus of the hepatocytes treated with PE70 and Dex. Data were plotted as mean ± SE. Con, control; M, mifepristone; PE, P. vulgarias water extract. *P < 0.05 versus control group; #P < 0.05 versus PE35 (n = 8).
The central analgesic effect was evaluated using the hot plate test. After treatment for 20 days, PE35, PE70, and Dex treated groups had significantly increased latencies to lick the hind paw from 18.9 ± 1.46 (control group) to 23.2 ± 1.86, 27.3 ± 1.96 and 28.4 ± 1.1 s, respectively (P < 0.05, n = 8). The increase was diminished by the application of mifepristone (p > 0.05 vs control group) (Figure 3C).
The antinociceptive effect of PE was also evaluated using the tail-flicking test. At 53 °C, the tail-flick response in the control group was observed at an average time of 4.00 ± 0.36 s. As a positive control, dexamethasone increased the response latency to 6.06 ± 0.30 s. PE70 increased the latency to 5.89 ± 0.39 s (n = 8, P < 0.05), which was diminished by the administration of mifepristone (p > 0.05) (Figure 3D). Taken together, PE treatment may have central analgesic effects through GR signaling pathways.
The rotarod test revealed that both PE35 and PE70 treatments significantly reduced the period that the animals remained on the rod after a 21-day treatment compared to the control group (P < 0.05). Mifepristone was able to rescue the decrease (p > 0.05 vs control) (Figure 3E), suggesting that PE may have maladaptive effects on neuromuscular coordination, 32 or neurodegeneration 33 or muscle atrophy. 34
Plasma IL1β, IL6, and IL10 were tested 3 weeks after the behavioral test to ascertain the PE dependency of the inhibition of pro-inflammatory cytokines. IL1βwas inhibited by 20.2% and 34.1% by PE35 and PE70, respectively (Figure 3F) (P < 0.05). Consistently, IL6 was 222.3 ± 18.8 pg/mL (9.6%) and 195.4 ± 14.1 pg/mL (20.7%) in the PE35 and PE70 groups, respectively, and they were significantly lower than that of the control group, 246.4 ± 21.6 pg/mL (Figure 3G) (P < 0.05). Similarly, the IL10 level was down-regulated to 81.9 ± 9.1 pg/mL (13.2%) and 68.7 ± 6.5 pg/mL (27.1%) compared to 94.2 ± 12.6 pg/mL in the control group (Figure 3H) (P < 0.05). RU486 significantly relieved the PE inhibitory effect for the above cytokines (p > 0.05 vs control group) (Figure 3F, G, H). The above results are consistent with the previous work on the regulatory effect of GCs on plasma cytokines. 21
To demonstrate further the analgesic mechanism of PE treatment, cyclooxygenase 2 (Cox2) protein levels in the lumber spine (Figure 3I, J, K) and the liver (Figure 3L, M, N) were measured by Western blots. In the lumber spine, PE and Dex significantly decreased Cox2 expression, whereas they increased Cox2 expression in the liver (P < 0.05); this is consistent with previous works.22,35 The GR analysis of the nuclear and cytoplasmic fractions clearly indicates that PE and Dex treatments induced GR translocation from cytosol to nucleus (Figure 3O, P), which is in line with a previous study on GCs. 22
PE Inhibitory Effects on Body Weight Gain, Food Intake, and Appetite
To determine whether PE treatment altered metabolism, mouse body weight was tracked during the treatment. Body weight gain was significant decreased 2 weeks after PE treatment compared to the control mice. Body weight of the PE70 group increased by 3.16 ± 0.13 g, whereas the control group increased by 4.51 ± 0.31 g (n = 8, p < 005). After 2 more weeks, the body weight comparison became 5.0 ± 0.33 vs 6.34 ± 0.39 g (n = 8, p < 005). The relative body weight change was plotted as Figure 4A. Meanwhile, the PE70 treated mice appeared to have less food intake after 2 weeks’ treatment compared to the control mice (n = 8, p < 0.05) (Figure 4B). PE35 treatment had the same trend, but with less amplitude compared to PE70 for food intake and body weight gain. Mifepristone treatment recovered both food intake and body weight gain to the same level as the control group (Figure 4A, B).
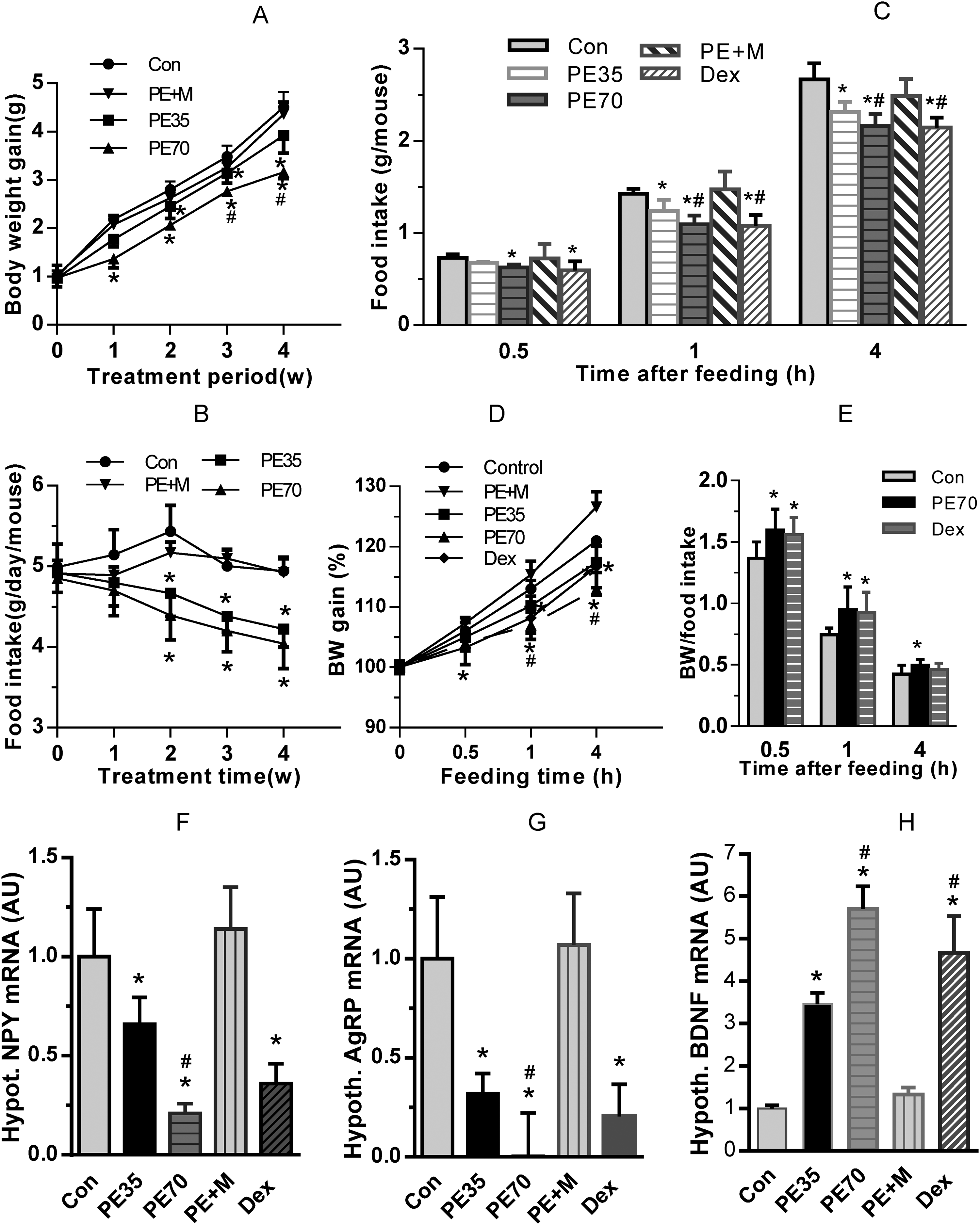
The regulation of PE on food intake, body weight gain, and hypothalamic gene expression. A. B. Food intake using individual host cages showing that PE35 and PE70 significantly decreased body weight gain (A), and food intake (B). C. Fasting food intake at 0.5, 1, and 4 h after food return. D. Relative body weight gain during fasting food intake test. Dex, P70, and P35 significantly decreased body weight gain. E. Comparison of the ratio of weight gain (%) to food intake in control, Dex, and PE70 groups showing Dex and PE70 treatment increase the ratio at 0.5, 1 and 4 h after food return. F. PE and Dex inhibitory effect on hypothalamic NPY expression; G. PE and Dex inhibitory effect on hypothalamic AGRP expression. H. Decreased hypothalamic BDNF expression by PE treatment. Con, control; Dex, dexamethasone; PE, P. vulgaris water extract; data were plotted as mean ± SE. *P < 0.05 versus control group; #P < 0.05 versus PE35 (n = 4-8).
To test further the PE effect on appetite control, mice were fasted overnight after the 21-day PE treatment. Then their food intake was measured 0.5 h, 1 h, and 4 h after food was returned. PE35, PE70, and dexamethasone treatments significantly reduced food intake compared with that of the control mice (Figure 4C), suggesting that PE treatment decreases appetite. Correspondently, dexamethasone, PE70 and PE35 treated-mice gained less weight (P < 0.05 vs control, Figure 4D). However, the ratio of body weight gain to the amount of food intake, which is an indicator of calories deposited as body weight,27,28 was significantly higher in the PE70 group from 0.5 h to 4 h (Figure 4E), suggesting that PE treatment actually increases the tendency of body weight gain relative to food consumption. Dexamethasone treatment exhibited a similar result as PE70 (Figure 4E). This evidence gives an interpretation that short-term GC therapy does not result in increased body weight, which occurs during long-term therapy. 36
To detect the underlying mechanism associated with body weight and appetite, gene expression analysis was performed using real-time PCR. Two hypothalamic orexigenic molecules, NPY (Figure 4F) and AgRP (Figure 4G), were highly decreased to 0.21 ± 0.09 and 0.006 ± 0.285 fold by PE70 compared to control mice (P < 0.05), which is consistent with the decreased appetite. Mifepristone recovered the PE inhibitory effect, suggesting the central effect of PE treatment is through GC signaling. Dexamethasone treatment also gave a similar effect as PE treatment (Figure 4F, G). Two genes related to anorexia, pro-opiomelanocortin and cocaine- and amphetamine-regulated transcript, were also measured, but no significant change was detected (data not shown). Hypothalamic BDNF analysis revealed that PE35 and PE70 treatments significantly increased BDNF level to 3.45 ± 0.27 and 5.70 ± 0.53 folds, respectively, compared to the control group (Figure 4H) (P < 0.05).
Impairment of Glucose Tolerance by PE Treatment
To test the effect of PE treatment on glucose metabolism, after the 21-day PE treatment, mice were fasted overnight, and basal blood glucose level was measured followed by IP injection of glucose (2 g/kg). Plasma glucose was measured every 15 min afterwards. Both PE35 and PE70 treatments led to a lower fasting plasma glucose level (P < 0.05, n = 8) (Figure 5A). Relative levels to the starting point were plotted as in Figure 5B, showing that blood glucose level in the PE35 and PE70 groups surged significantly higher than the control group 15 min after glucose injection, and this was maintained until 30 to 45 min afterwards (P < 0.05, n = 8) (Figure 5B). The AUCs of the PE35 and PE70 groups were increased by 115% and 135%, respectively compared with the control group. The AUC of the mifepristone treated group was about identical to that of the control group (99.5%) (Figure 5C).

PE effect on glucose metabolism and hepatic gene expression. A. Fasting glucose level showing decreased fasting blood glucose level by PE treatment. B. Dynamic change of relative blood glucose level showing increased blood glucose levels 15 min after IP injection by PE35 and PE70 treatments. C. Quantification of the area under curve in B. D. Regulation of hepatic G6PC1 expression by PE35 and PE70 treatments. E. Regulation of hepatic BDNF expression by PE treatment. Data were plotted as mean ± SE. *P < 0.05 versus control group; #P < 0.05 versus PE35 (n = 4-8).
To investigate the underlying mechanism of the over-abrupt plasma glucose level in PE treated mice, hepatic glucose-6-phosphatase catalytic-subunit-encoding gene (G6PC1) expression in the liver was checked using real-time PCR. G6PC1 mRNA level was increased by 1.83 ± 0.27 and 2.78 ± 0.31 folds in the PE35 and PE70 groups, respectively, compared with control mice (P < 0.05, n = 4) (Figure 5D). Mifepristone inhibited the PE-induced G6PC1 mRNA level (p > 0.05 vs control). Hepatic BDNF was down-regulated by PE35 and PE70 to 0.70 ± 0.11 and 0.42 ± 0.07 folds, respectively (P < 0.05) (Figure 5E).
Elevated Plasma GC and BDNF Levels and the Expression of Hepatic GC-Target Molecules by PE Treatment
Measurement of plasma GCs revealed that the GC level in the control group was 2.94 ± 0.47 ng/mL. PE35 and PE70 treatments significantly increased GCs to 9.1 ± 1.8 and 15.7 ± 2.3 ng/mL (Figure 6A) (P < 0.05). Meanwhile, plasma BDNF levels were significantly elevated to 143.7 ± 13.1 and 176.6 ± 18.4 pg/mL by PE35 and PE70, respectively compared to 118.4 ± 13.7 pg/mL in the control group (n = 8) (P < 0.05, n = 8). The PE70 increase was significantly higher than that of the PE35 treatment (P < 0.05) (Figure 6B). Mifepristone administration dramatically diminished the PE70 effect to control group level (Figure 6B, P < 0.05). The elevated level of plasma BDNF was consistent with its expression in the hypothalamus (Figure 4H), and other organs (data not shown), but not the liver (Figure 5E).

Influence of PE treatment on plasma GC (A) and plasma BDNF (B) level (n = 8), hepatic GILZ (C), hepatic SGK1 (D), GSK3β (E), and PKB (F) expression (n = 4). Data were plotted as mean ± SE. *P < 0.05 versus Con.; #P < 0.05 versus PE35.
To confirm further the GC-like effects of PE treatment, the transcriptional expression of GC-target genes, including GILZ, serum- and GC-induced protein kinase 1(SGK1), glycogen synthase kinase 3β (GSK3 β), and serine/threonine-specific protein kinase (Akt2/PKB), was quantified by real-time PCR. Hepatic GILZ, which serves as a mediator of the anti-inflammatory effects of GC in a variety of cells, 31 was increased by 2.98 ± 0.41 folds in the PE70 group (P < 0.05) (Figure 6C). Hepatic SGK1, which accelerates the development of metabolic syndrome, including glucose intolerance, 7 was highly increased by 15.55 ± 1.43 folds (P < 0.05) in the PE70 group (P < 0.05) (Figure 6D). Hepatic GSK3 β was mildly, but significantly, increased by 1.4 ± 0.11 (P = 0.0484) and 1.78 ± 0.26 (P = 0.007) folds by PE35 and PE70, respectively (Figure 6E). GSK3β was originally found to inactivate glycogen synthase and mediate the development of insulin resistance, then identified as a potential target of the therapy for diseases associated with inflammation. 9 In contrast, Akt2/PKB, another GC-target molecule inhibiting glucose release from hepatocytes and increasing insulin sensitivity, 37 was also highly up-regulated by 21.41 ± 2.48 folds in the PE70 group (Figure 6F).
Discussion
Network pharmacology studies in the current investigation found that 8 genes for both analgesic and glucose intolerance are targets of 6 ingredients of P. vulgaris. Experimental studies confirmed that the central and peripheral analgesic effects of P. vulgaris are through increasing plasma GC level, translocating of GR, elevating the expression of the anti-inflammatory marker gene GILZ, and down-regulating some inflammatory cytokines, as shown by previous works on GCs.21,22,31,35,38 Consequently, PE treatment increased the expression of G6PC1 and some GC-responsive kinases like SGK1, GSK3β and PKB, which, in turn, modulate glucose homeostasis and the anti-inflammatory process. These findings are consistent with the previous investigations on GCs,21,23,24,29,39 and confirmed the network pharmacology studies. Therefore, the analgesic effect and impairment of glucose disposal of PE treatment are actually from 2 different downstream signaling pathways of GCR/NR3C1. For the first time, the current investigation found that plasma BDNF is significantly elevated, and hypothalamic NPY and AgRP are down-regulated by PE treatment. The above results at least partially elucidate the mechanisms of analgesic activity of P. vulgaris and its side effect of glucose intolerance.
The elevated GC content and potentiated downstream signaling may be directly induced by the ingredients of P. vulgaris. For example, oleic acid can modulate the activity of GR and the feedback regulation.40,41 In addition, stigmasterol in P. vulgaris, which has similar pharmacokinetic properties as dexamethasone, 42 may exhibit anti-inflammatory action through repression of COX-2 expression. Nevertheless, the above suggestions do not exclude the possibility that sterols in P. vulgaris are precursors in the synthesis of steroid hormones, for example, GCs in mice. 43 Given the fact that GCs are widely applied in numerous diseases such as allergies, inflammation, and rheumatoid arthritis, the current investigation provides an interpretation for the application of P. vulgaris to a variety of symptoms.1-4,6,11 Hence, P. vulgaris may provide a natural replacement of synthetic GCs for the treatment of numerous diseases, with lesser side effects.
GCs are commonly used at high doses and for prolonged periods from weeks to months in the treatment of numerous symptoms with some well-known side effects, such as glucose intolerance, and diarrhea, which were also observed with PE treatment in this research and previous studies. The causative components of glucose impairment in PE may be different from GCs. For instance, oleic acid may stimulate glucagon secretion, 14 and GC-analogues in P. vulgaris may elevate the expression of G6PC1 resulting in the escalated gluconeogenesis.23,44 However, both treatments exhibit a similar pattern regarding the upregulation of GC-responsive genes, including hepatic SGK1 and PKB/Akt, which are highly involved in the pathophysiology of glucose intolerance, obesity, diabetes, and inflammation.7,45 In addition, both treatments also raise similar symptoms. The current investigations found that the P. vulgaris treated mice have a higher ratio of bodyweight gain vs food intake, which is consistent with the previous conclusion on GCs for the increase of weight gain with central obesity. 23 Regarding the side effect of diarrhea, GCs are causative factors of chronic watery diarrhea through the epithelial sodium channel that contributes to absorption of the cation in the human colon, 46 which may also occur in the clinical use of P. vulgaris. In addition, the diminished expression of NPY caused by PE treatment may also lead to diarrhea.46,47
Like GC treatment, some evidence of P. vulgaris application in the current investigation appears counter-intuitive. For example, as the effector molecules of the stress response, GCs are known to increase the consumption of foods enriched in fat and sugar, but acute stress usually results in decreased eating, whereas chronic stress results in increased eating. 48 GCs are known as catabolic molecules, but GC excess manifests as visceral adiposity. 49 Similarly, P. vulgaris treatment shows a decreased appetite, but an increased ratio of body weight gain to food intake. Likewise, an elevated plasma BDNF level and hypothalamic BDNF expression, coupled with a down-regulated hepatic BDNF expression occurred in the current research. Moreover, P. vulgaris treated mice had a relatively lower fasting plasma glucose level compared with control mice, but had a higher surge after glucose injection, which is found in nondiabetic and type I diabetic humans with impaired glucose disposal following mild hypoglycemia. 50 The above scenarios may be caused by a cluster of GC-responsive genes with opposite functions. For instance, PE treatment simultaneously increases both SGK1 and Akt2/PKB mRNA levels. Herein, Akt2/PKB inhibits glucose release from hepatocytes, enhances glucose utilization, and increases insulin sensitivity, 37 whereas the elevated SGK1 in the liver potentiates glucose generation, exacerbates diet-induced obesity and metabolic syndrome. 7 Opposing PKA regulation but cooperating with SGK, the elevated GSK3β, followed by phosphorylation and inactivation of glycogen synthase, acts as a negative regulator in the hormone control of glucose homeostasis. 51 Yet, PE treatment may organ-specifically regulate gene expression to harmonize body functions. For example, PE treatment increased hypothalamic BDNF expression to reduce appetite, but decreased hepatic BDNF expression to facilitate G6Pase expression to fulfil the elevation of blood glucose. 52 To muddy the waters further, the elevated PKB expression by PE treatment, in turn, may mediate the repression of GC-stimulated gluconeogenesis to counter balance GC function, and produce GC resistance for feedback inhibition of the anti-inflammatory effect of GC therapy. 44 Confusingly, but importantly, the dynamic change may lead to a variety of coping mechanisms to maintain the small oscillating amounts of GCs and normalize behavioral and physiological functions. Therefore, like GCs, PE increases animal metabolic flexibility to adapt better to environmental stress through increasing both counters at the same time.
The current investigation showed a differential BDNF expression pattern in the hypothalamus and the liver, and plasma BDNF is significantly increased under the condition of highly elevated plasma GCs. The increased plasma BDNF may be a combinatorial consequence of tissue specific regulation from GCs, as shown in current research and linolenic acid-induced increase of serum BDNF shown in healthy adult humans. 53 Given the fact that BDNF serves important roles in regulating metabolic homeostasis and nociception, the elevated plasma BDNF and organ-specific regulation of BDNF fine tunes the GC effects for the subject to be better assimilated into the environment.54-56 Current research demonstrated a reduced appetite, which is in line with the expression of anorexic genes in the hypothalamus, and the phenotypes in a neurogenesis-deficient mouse model. 57 However, GCs have long been recognized as a food craving stimulus, indicating a similar but different regulation pattern of P. vulgaris treatment from GC application.
In summary, at least 11 compounds of P. vulgaris, including ursolic acid, rosmarinic acid, and oleanolic acid, contribute to the anti-inflammation and plasma glucose regulation through 12 hub genes and 13 relevant biological processes. PE-induced GC is a curative mediator for the anti-inflammatory effects of P. vulgaris through Cox2, GILZ, and downstream signaling. Consequently, PE treatment impairs glucose disposal, impedes mouse body weight gain and decreases mouse appetite, but may raise the risk of obesity. PE treatment also increases the plasma BDNF level allowing the subject to adapt better to internal and external condition changes. The investigation delineates the mechanisms of the analgesic effect of P. vulgaris and the concurrence of the side effect of glucose intolerance, which may help to develop optimal therapeutic strategies for a spectrum of symptoms when P. vulgaris is applied.
Footnotes
Acknowledgements
We thank Professor Shuke Cui for discussing P. vulgaris side effects, and Stephanie Zhang for assistance with network pharmacology studies.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from Henan Natural Science Foundations (212300410376, 162300410203) (to ZZ); Science and Technology Project of Henan Province (172102310218 to CZ), (202102310512 to LG), and the National Natural Science Foundation of China (81803661) (to CL).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
