Abstract
Phytochemicals from the genus, Fagonia, have been attracting increasing attention due to their potential beneficial effects on human health. Fagonia species contain various types of phytochemicals such as flavonoids, alkaloids, saponins, terpenoids, coumarins and tannins. In this study, we investigated the phytochemical composition of unhydrolyzed and acid-hydrolyzed extracts of Fagonia indica and their bioactivity toward breast cancer MCF-7 cells in vitro. The results revealed that F. indica contains phytochemicals consistent with the reported phytochemical composition of this Fagonia species, with greater amounts of aglycones detected in the hydrolyzed extract. The crude extract of F. indica without acid hydrolysis was found to be ineffective in inhibiting the growth of MCF-7 cells at doses below 1000 µg/mL. However, after acid hydrolysis (to mimic gastro-intestinal hydrolysis), the F. indica extract became growth-inhibitory to MCF-7 cells as low as 10 µg/mL and the cytotoxicity increased with increasing dose and time of treatment. The results suggest that F. indica extracts contain phytochemicals in glycosidic forms whose aglycones are active as anti-proliferative agents toward breast cancer cells in vitro.
Introduction
Breast cancer is one of the most prevalent types of cancer in women worldwide, accounting for an estimated 30% of all new cancers in women in 2019. Female breast cancer surpasses lung cancer as the leading cause of global cancer incidence, with an estimated 2.3 million new cases of breast cancer and 685,000 deaths in 2020. 1 It is difficult to eradicate or treat this type of cancer because it has various phenotypes and is linked to genetic, epigenetic, and non-hereditary factors. 2 The common therapeutic approaches for cancer treatment are chemotherapy, immunotherapy, radiotherapy, surgery, and a combination of modalities 3 but these therapies are not always successful especially when the cancer cells develop resistance to anti-cancer drugs. The use of anti-cancer drugs is frequently accompanied by harmful side effects. Moreover, the primary tumor could be successfully treated but it may have already metastasized. 4
Therefore, a strategy for dealing with breast cancer in women is to inhibit or reverse carcinogenesis at the pre-malignant phase by chemoprevention, which involves the use of pharmacological agents and phytochemicals. 5 Certain plants such as those belonging to the Zygophyllaceae family have been used as herbal medicines and their secondary metabolites such as polyphenols have been investigated for their cancer chemopreventive properties. 6 Extracts of Zygophyllum album Desf. (synonym Tetraena alba) have been reported to exhibit anti-proliferative activity against human lung carcinoma and colon adenocarcinoma cells. 7 One medicinal plant of the Zygophyllaceae family that is gaining interest is Zygophyllum indicum (Burm.f.) Christenh. & Byng) 8 now known as Fagonia indica Burm, one of 34 species of the genus, Fagonia. F. indica is a thorny herb distributed largely in Pakistan, Afghanistan, Egypt, and Western India 9 and thrives in arid and rocky areas. F. indica contains flavonoids, alkaloids, saponins, glycosides, pectin, and tannins.10‐13 It is used by practitioners of traditional medicine for various ailments in certain parts of South Asian countries because of its purported anti-diabetic, laxative, hepatoprotective, antipyretic, anti-microbial, analgesic, and anti-inflammatory properties.11,14 This plant species may also act as a potential anti-cancer agent.15,16 An aqueous extract of Fagonia cretica was found to induce growth inhibition and apoptosis in breast cancer cells. 17 The authors of that publication later identified their plant material as Fagonia indica based on DNA sequencing analysis. 18 A saponin glycoside 19 and quinovic acid 20 isolated from F. indica have been shown to induce apoptosis or necrosis in MCF-7 cancer cells in vitro. F. indica contains flavonoids as glycosides or as aglycones such as quercetin, isorhamnetin, kaempferol, and apigenin which have been shown to induce growth-inhibition or apoptosis in MCF-7 cells and other breast cancer cell lines.21‐23 Other constituents of F. indica such as ursolic acid,24,25 ferulic acid, 26 and arbutin 27 also induced apoptosis in breast cancer cells in vitro.
The present study was conducted to determine the phytochemical components of unhydrolyzed and acid-hydrolyzed extracts of F. indica stems and leaves by liquid-chromatography-mass spectrometry (LC-MS/MS). A previous phytochemical screening of various parts of the F. indica plant showed that the stems have high amounts of saponins whereas the leaves have high amounts of tannins and cardiac glycosides. 11 However, most studies on phytochemical composition of F. indica are qualitative in nature and at best semi-quantitative11‐13,28,29 There were no previous reports of actual quantitative evaluation of phytochemicals present in unhydrolyzed and acid-hydrolyzed F. indica extracts. Thus, it was of interest to examine the relative abundance of phytochemicals existing as glycosides or as aglycones in our F. indica extracts with and without acid hydrolysis. Phytochemicals in plant extracts such as anthocyanins, saponins, alkaloids, phenolic acids, and flavonoids occur predominantly as glycosides. 30 Acid hydrolysis using HCl has been used to release the aglycones from saponin-rich extracts of fenugreek 31 and quinoa 32 and from flavonoid glycosides in oil palm leaf extracts. 33 The present study would reveal what bioactive glycosides and aglycones are most abundant in the unhydrolyzed F. indica extract and what glycosides and aglycones are most abundant in the extract after acid hydrolysis. This information would provide an insight into the potential use of F. indica extracts as cancer chemopreventive agents and whether acid hydrolysis would improve the bioactivity of the crude extract of this plant species. The release of aglycones from glycosides by acid hydrolysis mimics the hydrolysis of ingested glycosides in edible plants, fruits and herbal products by the acidic environment of the stomach and by hydrolytic enzymes in the small intestines 34 and in the gut microbiota. 31
The second objective of this study was to evaluate the bioactivity, measured as growth-inhibition of MCF-7 cells in culture, of both unhydrolyzed and acid-hydrolyzed F. indica extracts. Some glycosides such as isoflavone glucosides 35 and saponins 32 are less bioactive than their corresponding aglycones. However, there are glycosides such as the steroidal saponin glycoside from F. indica, 19 hederagenin saponin from Clematis ganpiniana 36 and the anthocyanin, cyanidin-3-O-glucoside, 37 that are bioactive as shown by their ability to inhibit the growth of cancer cells. Therefore, it is possible that there might be glycosides in the F. indica extract that are bioactive by themselves. On the other hand, hydrolysis of inactive or less active glycosides in F. indica extract may release aglycones with greater bioactivity. Our results suggest that acid hydrolysis made the F. indica extract more bioactive in inhibiting the growth of MCF-7 breast cancer cells, suggesting that aglycones likely contributed to the anti-proliferative activity of the extract.
Results and Discussion
Analysis of F. indica Extract by Mass Spectrometry
Several phytochemicals such as flavonoids, alkaloids, terpenoids, saponins, tannins, cyanogenic glycosides, sterols, anthraquinones, iridoids, and coumarins have been reported to be present in F. indica.11‐13 In the present study, the phytochemicals in unhydrolyzed and acid-hydrolyzed extracts of F. indica (Table 1, Figures 1 and 2) were detected using PeakView software (XIC manager). A list of 143 Fagonia phytochemicals was created based on comparison with in-house library spectra, the KNApSAcK database and with literature reports.38‐42 The peak picking parameters were: intensity ≥ 100 count, S/N ≥ 10 and mass error ≤ 5.0 ppm. In the unhydrolyzed extract, the major glycosides detected were two quinovic acid glycosides, cauloside A, and a glycoside of kaempferol (Table 1, Figure 1). After acid hydrolysis, the triterpenoids, ursolic acid and quinovic acid, and the flavonoid, kaempferol, were the most abundant aglycones in the F. indica extract (Table 1, Figure 2). These results demonstrated that the saponins and flavonoid glycosides in F. indica extract were hydrolyzed to yield their respective aglycones. Quinovic acid itself was detected as the major aglycone in the unhydrolyzed extract (Table 1). The finding that ursolic acid is the major aglycone in the hydrolyzed extract was surprising because no known glycosides of ursolic acid such as indicasaponin A 40 were detected in the unhydrolyzed extract. Indicasaponin A 40 and its aglycone, ursolic acid have been isolated previously from F. indica. Other compounds such as nahagenin, hederagenin, indicacin and fagonicin reported earlier to be present in F. indica 11‐13,25,40 were not detected in our F. indica extract. The failure to detect certain compounds known to be present in F. indica could be due to the lack of spectral data for these compounds in our in-house library, differences in extraction method, or the compounds were below the limits of detection by our ESI-MS analysis. Furthermore, plant composition is known to vary with regards to growth stage, location, and year. Nevertheless, acid hydrolysis was found to be effective in cleaving the glycosides to give rise to aglycones but some glycosides like cauloside A and quinovic acid-3β-O-β-D-glucopyranoside may be resistant to acid hydrolysis since the amounts of these glycosides were not reduced after acid hydrolysis of the F. indica extract (Table 1).


Phytochemicals detected in an extract of the aerial parts of Fagonia indica before (unhydrolyzed) and after acid hydrolysis (sorted by accurate mass).

When compounds were detected in both the positive and negative ion mode, the ions with the greatest peak area are reported.
Discrepancies between retention times of positive ion peaks reported in Table 1 and negative ion peaks in the figures are because samples were recorded at different times.
Missing numbers indicate that the ion was not detected.
NMR Analysis of Triterpenoid Saponins from F. indica
The structures of the compounds present in the chromatographic fractions of the F. indica extract were determined using spectroscopic methods and confirmed by comparison against literature data. On the basis of the results of combined spectral analyses including 1H COSY, HMQC, and HMBC NMR experiments, the structures of the aglycones were determined to be quinovic acid (1-3) or hydroxyoleanolic acid (4). The sugar portions were proven to be assemblies of one, two or three units of glucose (1-3), and one unit each of glucose and glucuronic acid (4). Four compounds were identified (Figure 3) as follows: (

Chemical structures of triterpenoid glycosides isolated from Fagonia indica. (1) quinovic acid-3β-O-β-
Effect of F. indica Extract on MCF-7 Cancer Cell Culture
In previous studies, aqueous extracts of Fagonia species demonstrated antiproliferative and cytotoxic effects in human cancer cell lines,17,19 with a markedly reduced effect on normal human mammary epithelial cells 17 or normal endothelial cells. 19 These studies highlighted phytochemicals in F. indica extracts as promising new bioactive compounds that could be beneficial to human health. Some anti-cancer drugs such as paclitaxel, camptothecin, and etoposide in clinical use are of plant origin. 20 In this study, F. indica extracts were evaluated for their growth-inhibitory effects in breast cancer cells (MCF-7) as compared to the reference xanthohumol (XN), a hop flavonoid known to be toxic to MCF-7 cells but not to normal hepatocytes at low micromolar doses. 46 There are 92 known breast cancer cell models 47 and the MCF-7 cell line (estrogen-positive) is one of the most common cell lines used to screen natural products for their potential anti-breast cancer activities.
The effects of different concentrations of the unhydrolyzed extract on MCF-7 cell viability after 24 (a), 48 (b) and 72 (c) h of incubation are shown in Figure 4. No significant reduction in cell viability was observed after 24 or 48 h of incubation even at a concentration of 1200 μg/mL of the unhydrolyzed F. indica extract. After 72 h of incubation, an inhibitory effect of the extract on cell viability was observed at a concentration of 1200 μg/mL. These results indicate that unhydrolyzed F. indica extract was rather ineffective in reducing the viability of human breast MCF-7 cancer cells and appeared to be weaker than an aqueous extract of F. cretica which was shown to be effective in reducing cell viability of MCF-7 cells at a dose of 250 µg/mL after a 24-, 48-, or 72-h treatment and in causing an approximate 75% reduction in cell viability after 72 h treatment with 2 mg/mL. 17 Another study showed that an ethanol extract of F. indica caused a dose-dependent inhibition of growth of MCF-7 cells, with more than 70% growth inhibition at a dose of 500 µg/mL. 19 A bioassay-guided fractionation with silica gel chromatography revealed that the most active component of the ethanol extract was a steroidal saponin glycoside. 19 Our extract was prepared by sonicating powdered aerial parts of F. indica in 70% methanol for 30 min whereas the ethanol extract with the bioactive saponin glycoside was prepared by macerating the aerial parts of F. indica in 100% ethanol for 14 days 19 which may have extracted greater amounts of bioactive compounds than our extraction method.

Effect of unhydrolyzed Fagonia indica extract on breast cancer MCF-7 cell viability. MCF-7 cells were treated with the extract from 1 to 1200 µg/mL for 24, 48, and 72 h prior to analysis of cell viability by MTT assay. Data represent means ± SE from four experiments, with *p < 0.05, versus DMSO control cells analyzed by one-way ANOVA with Dunnett's multiple comparison posthoc test.
One explanation for the weak inhibitory effect of unhydrolyzed F. indica extract on the viability of MCF-7 cells is that the glycosides and aglycones as a mixture in our extract may be inactive towards MCF-7 cells, although other studies17,19 have shown that F. indica extracts inhibited the growth of breast cancer cells. Our extract was prepared in a different manner as compared to previous studies17,19 and we may not have the same amounts and type of bioactive glycosides and aglycones in our unhydrolyzed extract as other F. indica extracts17,19 prepared in other laboratories that were active towards breast cancer cells. The phytochemicals in Fagonia species that may exist as glycosides are saponins, flavonoids, alkaloids, tannins, and terpenoids,48‐50 which were detected in our own extract. We hydrolyzed the glycosides in our F. indica with acid to model gastrointestinal digestion and tested the hydrolyzed extract for cytotoxicity to MCF-7 cells as before. Our results (Figure 5) showed that the viability of the MCF-7 cancer cells was inhibited both in a concentration and time-dependent manner by the hydrolyzed extract. We observed that after a 24-h exposure period, the viability of the MCF-7 cells was significantly reduced by the hydrolyzed extract at a dose of 200 µg/mL. When the exposure period with the test compound was extended to 48 h, the hydrolyzed extract was able to significantly reduce cell viability at a lower dose of 50 µg/mL. At 72 h of exposure, a statistically significant modest reduction in cell viability was observed as low as 10 µg/mL of the hydrolyzed extract and substantial reductions were observed at 200 µg/mL and above. These results suggest that acid hydrolysis of the F. indica extract released the bioactive aglycones, notably, quinovic acid, ursolic acid and kaempferol (Table 1) from the inactive or less active glycosides that might account for the enhanced bioactivity of the hydrolyzed extract.
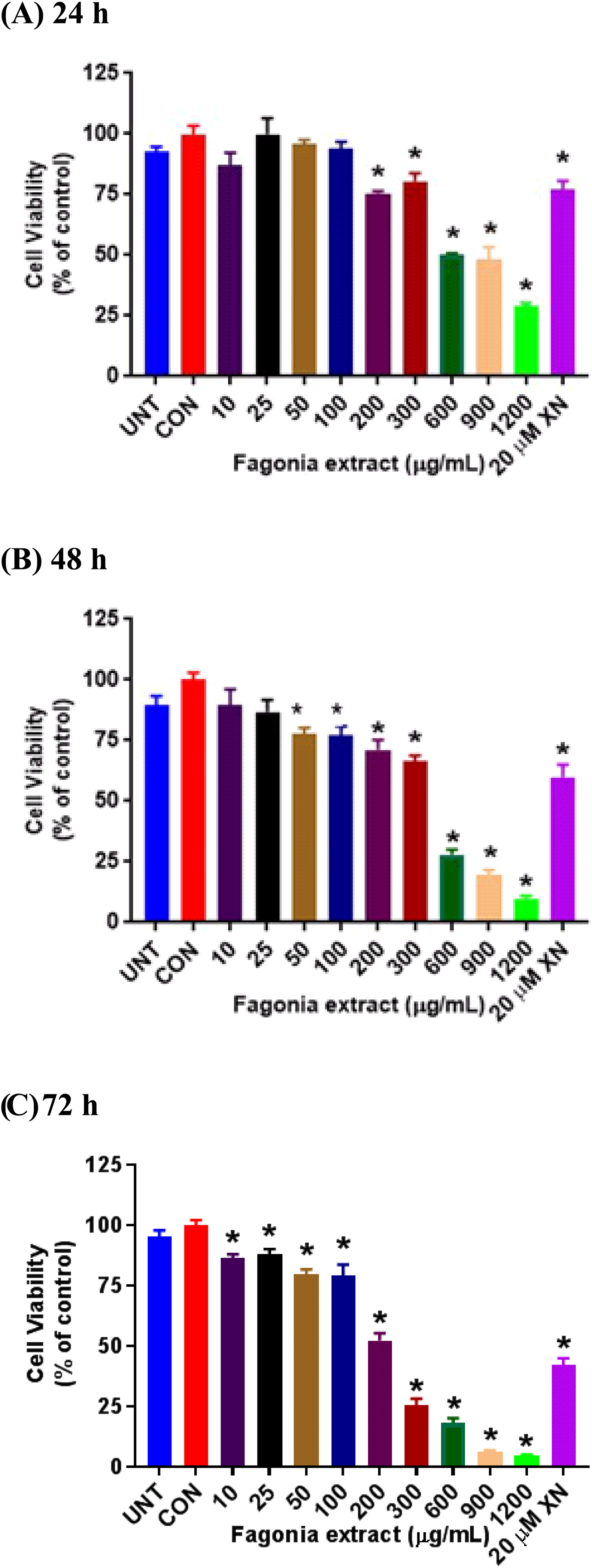
Hydrolyzed Fagonia indica extract inhibits the viability of MCF-7 cells in a concentration- and time-dependent manner. Cells were treated with increasing concentrations of the extract for 24, 48, and 72 h, before testing by MTT assay. Data represent means ± SE from four experiments, with *p < 0.05, versus DMSO control cells analyzed by one-way ANOVA with Dunnett's multiple comparison posthoc test.
The mechanisms of F. indica extract-induced cytotoxicity in breast cancer cells were not examined in this study. However, the cytotoxic activity of an aqueous extract of F. cretica towards human breast cancer cell lines was attributed to cell cycle arrest and apoptosis in wild type p53 MCF-7 and mutant p53 MDA-MB-231 cells.
17
Those authors reported that the cell cycle arrest may be associated with induction of DNA damage via activation of the ATM (mutated in ataxia-telangiectasia)/p53-mediated DNA damage response and on the induction of FOXO3a expression in both cell types.
17
Recently, it was shown that quinovic acid purified from F. indica suppressed the growth of breast cancer cells (MDA-MB-231) by inducing apoptosis through the upregulation of the death receptor 5 pathway.
20
Quinovic acid did not affect the growth and viability of non-tumorigenic MCF-10A cells even up to 40 µM.
20
The flavonoids such as kaempferol, apigenin, quercetin and their derivatives found in F. indica are known to induce apoptosis and growth inhibition in breast cancer cells through various mechanisms such as inhibition of glucose uptake by the cancer cells,
22
down-regulation of polo-like kinase 1
51
and Bax expression,
52
cell cycle arrest in the G2 phase,53,54 and activation of the caspase cascade pathway.51,55 Some polyphenols may induce toxicity in cancer cells by their ability to act as protonophores and prooxidants in mitochondria of cells.
56
Ursolic acid, one of the major triterpenoids found in our hydrolyzed F. indica extract, was also anti-proliferative in MCF-7 cells by modulating the ERK and P13/AKT signaling pathways.
24
Gallic acid, the other major acid in our hydrolyzed F. indica extract, was shown to induce apoptosis in MCF-7 cells by altering P53, P21, and Mcl-1 gene expression.
57
Hederagenin also induced apoptosis in MCF-7 cells via the mitochondrial and caspase cascade pathways.
36
In future studies, we will evaluate the bioactivity and mechanisms of action of saponins such as quinovic glycosides
Conclusions
The results of this study support the potential of F. indica extract after acid hydrolysis as a potential source of anti-proliferative agents for breast cancer. The hydrolyzed F. indica extract was more effective than the unhydrolyzed extract in inhibiting the growth of MCF-7 cells. Further studies are needed to identify novel components in hydrolyzed F. indica extract that are bioactive toward breast cancer cells. Additional studies are also needed to determine whether the bioactive components in F. indica extract are toxic to normal breast cells or to normal liver cells. The cytotoxic effects of F. indica bioactives on other human cancer cell lines are also of interest to establish the selectivity of this plant species as a potential anti-cancer agent in humans.
Materials and Methods
Preparation of F. indica Extract
The aerial part (stems and leaves) of F. indica was collected in Islamabad, Pakistan by and identified by Dr Zabta K. Shinwari, Director, Biotechnology Department, Quaid-i-Azam University (PAK) as F. indica, with a voucher specimen located at Linus Pauling Institute, Oregon State University. It was further authenticated by Dr Shahina Ghazanfar, Royal Botanical Gardens, Kew (UK) and Dr Melanie Schori (USDA-ARS). F. indica extracts were prepared essentially as described by Zheljazkov et al. 58 Air-dried stems and leaves of F. indica plants were ground for 60 s in a Proctor-Silex model E160B coffee grinder to obtain a fine powder. An aliquot of the plant powder (5 g) was mixed with 70% methanol (50 mL) at room temperature. The mixture was sonicated for 30 min by submerging the tube in water inside the sonicator (VWR Ultrasonic Cleaner, Model no. 97041-0) operated at 60 Hz and then centrifuged at 8500 rpm for 20 min at room temperature. An ultrasound method has been used in the extraction of polyphenols from oil palm leaf. 33 The clear supernatant was taken to dryness under vacuum using a rotary evaporator. The residue (F. indica extract) was characterized by liquid chromatography in combination with mass spectrometry (LC-MS/MS) using the facilities of the Mass Spectrometry Center at Oregon State University. A portion of the dried F. indica extract was dissolved in dimethyl sulfoxide to make stock solutions for the cell culture study using MCF-7 cells.
Acid Hydrolysis of F. indica Extract
One hundred milligrams of F. indica extract residue was dissolved in 2 mL methanol, and the solution was added to 16 mL of 50% aqueous methanol (containing 2 M hydrochloric acid) in a 50 mL round bottom flask. The mixture was refluxed for 1 h, and then extracted with 50 mL ethyl acetate three times. The organic solvent layers were combined and dried under nitrogen gas flow. The acid-hydrolyzed extract was likewise analyzed by LC-MS/MS and tested for bioactivity in MCF-7 cells.
Isolation of and NMR Analysis of Triterpenoid Saponins from F. indica
The leaves and stems of F. indica (120 g) were extracted for 24 h each with dichloromethane (300 mL) and methanol (300 mL). The solvent was removed using a rotary evaporator, to yield crude extracts (6.25 g) which were partitioned between dichloromethane and water. The dichloromethane layer was concentrated, and the residue was repartitioned between n-hexane and 15% aqueous methanol (4.71 g). The aqueous methanol layer was dried and subjected to C18 reversed-phase vacuum flash chromatography using sequential mixtures of methanol and water as eluents (elution order: 40%, 30%, 20%, 10% water in methanol, and ethyl acetate). The fraction eluted with 20% water in methanol (1.04 g) was dried and separated by reversed-phase HPLC (YMC ODS-A column, 25% water in methanol) to yield triterpenoid glycoside
NMR spectra were recorded in CD3OD solutions on a Varian Unity 500 spectrometer. Proton and carbon NMR spectra were measured at 500 and 125 MHz, respectively.
LC-MS Detection of Other Phytochemicals in F. indica Stems and Leaves
Chromatographic separation of the F. indica extract was conducted using a Shimadzu Nexera UPLC system equipped with an Inertsil Phenyl-3 column (150 × 4.6 mm, 5 µm). The injection volume was 10 µL. Mobile phase A was water with 0.1% formic acid, and mobile phase B was methanol with 0.1% formic acid. The gradient started with 5% B and was held for 1 min at 5% B, followed by a 10-min linear gradient from 5% to 30% B. The gradient was then increased linearly from 30% to 100% B, reached at 35 min, held for 15 min at 100% B and, finally, stepped back to 5% B to equilibrate the column. The flow rate was 0.4 mL min−1, with the auto-sampler set at 10 °C and the column temperature maintained at 50 °C. The column effluent was introduced into a QTOF mass spectrometer (AB Sciex Triple TOF 5600) equipped with a TurboSpray electrospray ionization source operated in the negative and positive ionization modes. The instrument was operated in the information-dependent acquisition (IDA) mode using a collision energy setting of 35 V. Compound identification was based on accurate mass, isotopic similarity, retention time and MS/MS spectral comparison with authentic standards in our in-house library (IROA library consisting of 619 compounds; Enzo Life Science library of 500 natural products) and with spectral databases (METLIN and the Global Natural Products Social Network database).
MCF-7 Cell Culture
Human breast adenocarcinoma MCF-7 cells were purchased from ATCC (Manassas, VA). Upon arrival, the frozen cells were rapidly thawed by gentle agitation of the vial in a 37°C water bath. The cell suspension was then transferred to a T-75 (75 cm2) tissue culture flask (Corning Inc., USA) containing a growth medium consisting of DMEM supplemented with 10% heat-inactivated fetal bovine serum (Atlanta Biologicals, Flowery Branch, GA), penicillin (100 units/mL), streptomycin (100 µg/mL), and insulin (10 µg/mL) from bovine pancreas (Sigma-Aldrich, St. Louis, MO). The cells were incubated at 37˚C in a humidified atmosphere of 95% air / 5% CO2, until 80% confluency.
Cell Viability Screening Using MTT Assay
For treatment, the cells were harvested from the flask using trypsin and seeded into 96-well culture plates at a density of 10,000 cells in 0.2 mL of culture medium per well. After 2 days of incubation at 37˚C in a humidified atmosphere of 95% air / 5% CO2, the cells were treated with F. indica extracts at various concentrations (1-1200 μg/mL). A stock solution (1200 mg/mL) of unhydrolyzed or hydrolyzed F. indica extract was first prepared by sonicating the dried extract in DMSO for 30 min and then this solution was diluted to make other stock solutions at lower concentrations (1 to 900 mg/mL). To treat the cells, 1 µL of the stock solutions was added to 1 mL of culture medium to achieve final concentrations of the extract ranging from 1 µg/mL to 1200 µg/mL, with DMSO attaining a final concentration of 0.1%. Vehicle control cells were incubated with growth medium containing DMSO (0.1%). Positive control cells were treated with 10 µM xanthohumol (Hopsteiner, NY) whereas untreated cells were incubated with complete growth medium alone. The number of viable cells was determined by the MTT assay after 24, 48, and 72 h of exposure to the F. indica extract or DMSO.
The MTT assay was performed according to the method of Mosmann.
59
Briefly, the growth medium with the F. indica extract, XN or DMSO was aspirated from each well of the 96-well plates and a fresh complete growth medium containing MTT (0.5 mg/mL) was added to all wells. The plates were incubated at 37°C for 3 h and then the MTT-containing medium from each well was removed by aspiration to a waste vessel. The blue formazan dye was extracted from the viable cells attached to the bottom of the 96-well plates by adding 180 µL of isopropanol (with 0.04 N HCl) and shaking the plates for 5 min at room temperature. The optical density (OD) or absorbance of solubilized dye in the plates was measured using a plate reader (SpectraMax 190 spectrophotometer) at 570 nm. Each concentration of F. indica extract was assayed in quadruplicate wells and the viability of treated cells was expressed as a percentage of DMSO controls (100%) using the following equation: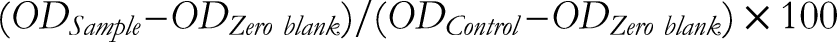
Statistical Analysis
Mean values and standard errors (SE) were calculated from each experiment and one-way analysis of variance (ANOVA) with Dunnett's test was used to compare statistical differences between treated and control (DMSO-treated), with p < 0.05 as the level of significance using GraphPad Prism 7.03.
Footnotes
Acknowledgements
This research was supported by the OSU Foundation Buhler-Wang Research Fund, the OSU BioResource Research Program, and the OSU Mass Spectrometry Center (National Institutes of Health Grant no. S10 RR027878). We extend our gratitude to Dr Zabta K. Shinwari, Director, Biotechnology Department, Quaid-i-Azam University (Pakistan), for collecting the plant material and initial identification; to Dr Shahina Ghazanfar, Royal Botanical Gardens, Kew (UK), and Dr Melanie Schori (USDA-ARS) for authentication of the plant material; and to Gail R. Campbell for providing us with the plant material and for sharing her time and ideas with us during this research.
Author Contributions
Concept of the study and supervision by Jan F. Stevens; design and methodology by Jan F. Stevens, Wenbin Wu, Hyi-Seung Lee, and Cristobal L. Miranda; investigation and analysis by Cristobal L. Miranda, Yadano Kumbi, Wenbin Wu and Hyi-Seung Lee; manuscript writing by Cristobal L. Miranda, Yadano Kumbi, Wenbin Wu, and Hyi-Seung Lee; and manuscript review and editing by Ralph L. Reed and Jan F. Stevens. All authors have read and approved the final version of this manuscript.
Declaration of Conflicting Interests
None of the authors has potential conflict of interest with regard to the research, authorship, or publication of this article.
Ethical Approval
Not applicable as no animal or human subjects were used in this study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the OSU Foundation Buhler-Wang Research Fund, OSU Mass Spectrometry Center, National Institutes of Health, OSU BioResource Research Program, (grant number S10 RR027878).
Informed Consent
Not applicable as no human subjects were used in this study.
Trial Registration
Not applicable as no clinical trials were used in this study.
