Abstract
Oxidation of geranyl acetone derivatives with ceric ammonium nitrate (CAN) and Mn(OAc)3 afforded tricyclic and bicyclic compounds as well as hydroxy and nitro compounds as a result of one-electron oxidation followed by carbon–carbon bond formation. This is the first example of radical cyclization (formed by one-electron oxidation) of geranyl acetone derivative
Keywords
One-electron oxidation
1
as well as one-electron reduction is the interesting reaction via a radical intermediate triggered by a radical initiator, such as AIBN, Bu3SnH, SmI2, and so on.
1
Electrolysis has also been studied by many groups,
2
since it is completely benign to the environment; nowadays generally accepted as green chemistry. On the other hand, single-electron oxidation of carbonyl compounds using ceric (IV) ammonium nitrate (CAN) and Mn(OAc)3 as an oxidant has been well studied.3‐5 Recently, Thomson
6
and MacMillan
7
independently used CAN for the radical coupling reaction of the enolate. Quite recently, Yoshida and Shishido reported a coupling reaction between cyclopentanone carboxylates and alkenes using the CAN and Cu(OAc)2 system.
8
And also, Yamashita and Hirama
9
succeeded in tandem cyclization producing tricyclic rings using Mn(OAc)3. Other metals, such as Fe, V, and Cu, were used for the generation of a radical by one-electron oxidation.10-17 We have been investigating one-electron reducing reactions applied to the construction of perhydronaphthalenes, hydrindanones, and perhydroguaianes.18-25 We are also interested in one-electron oxidation induced by CAN or Mn(OAc)3 making bicyclic compounds and we now report the results on the one-electron oxidation of geranyl acetone derivative
A solution of methyl 7,11-dimethyl-3-oxo-6,10-dodecadienoate (
Oxidation of Compound

Isolated yield.
Structures of

Selected HMBC and NOE correlations of compound
The stereochemistry was established by the NOESY spectrum, the selected correlations of which were shown in Figure 1. Fortunately, compound

ORTEP drawing of compound
The spectroscopic data of compound
We have next studied the oxidation of methyl 2-acetyl-5,9-dimethyl-4,8-decadienoate (
Oxidation of Compound

Isolated yield.
The structure of compound

Selected 2D correlations of compounds
Because one-electron oxidation of both compounds
When compound
Oxidation of Compound

Isolated yield.
At 80 ˚C.
The structure of each product was determined by spectroscopic analysis. The molecular formula of compound

Selected 2D correlations of compound
Compound
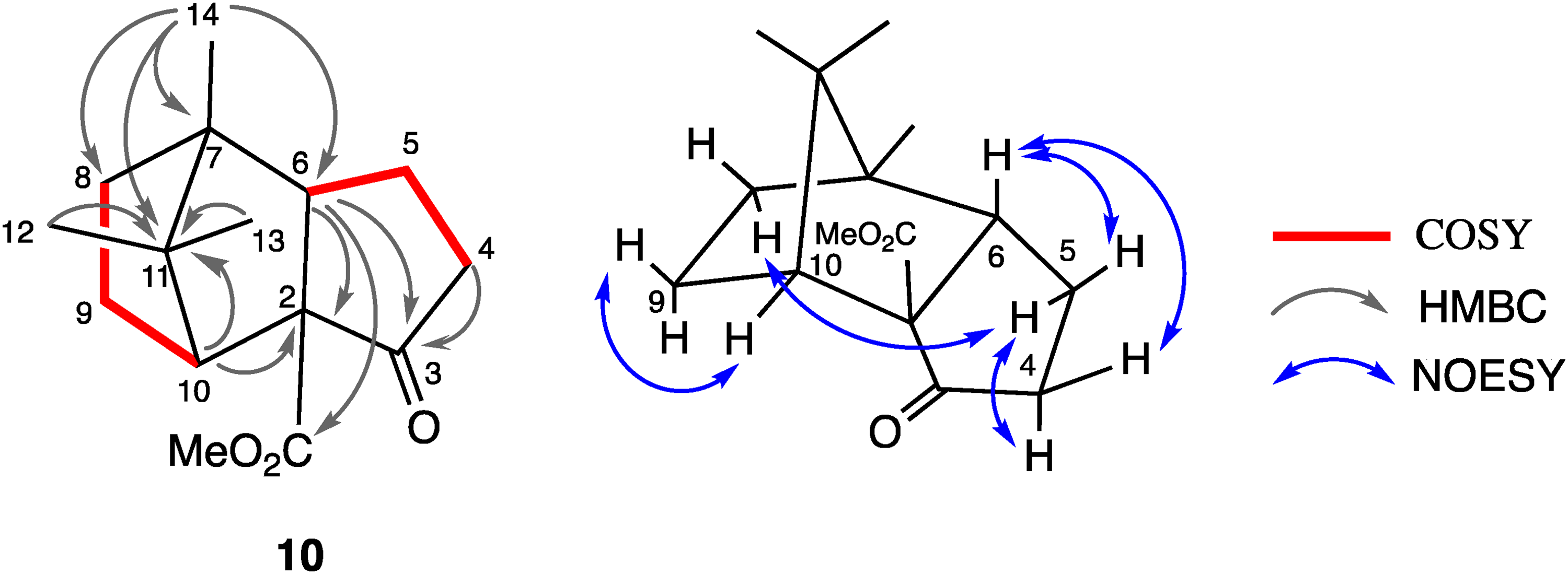
Selected 2D correlations of compound
Keto ester
Oxidation of Compound

Isolated yield.
The molecular formula of compound

Selected 2D correlations of compound
The formation of these products was explained by the mechanism shown in Scheme 1. Treatment of compound
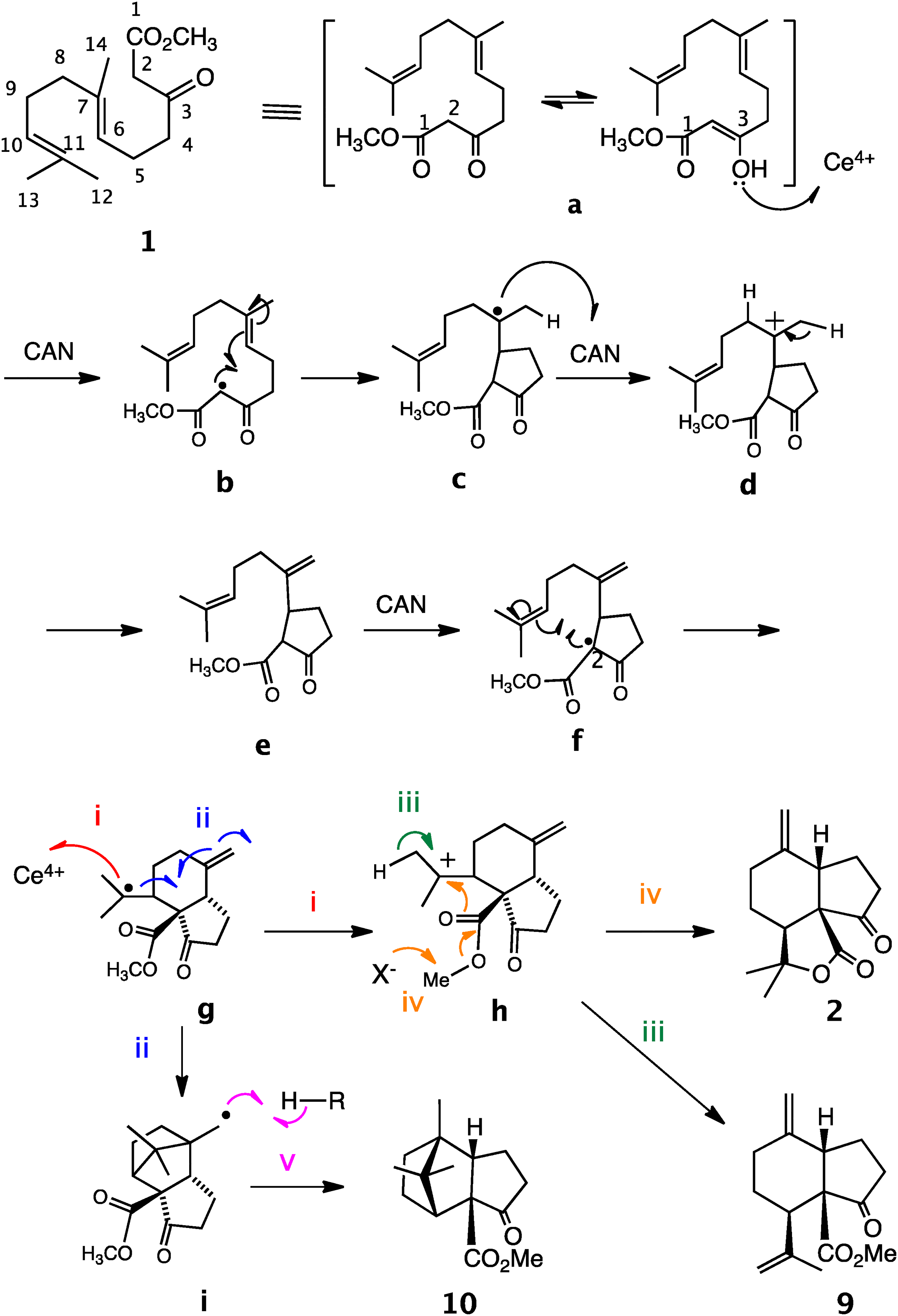
A plausible mechanism of one-electron oxidation of
In the case of compound
In conclusion, we have developed the oxidative cyclization of geranyl acetone derivative
Experimental
Compound 2
Crystals.
mp. 94–95 °C.
FTIR: 1759, 1734 cm−1.
1H NMR (600 MHz, CDCl3) δ 1.48 (3H, s), 1.54 (3H, s), 1.59–1.65 (1H, m), 1.72 (1H, dq, J = 14.6, 5.1 Hz), 2.16 (1H, dt, J = 14.3, 5.5 Hz), 2.23–2.29 (1H, m), 2.30 (1H, s), 2.34 (1H, br q, J = 9.1 Hz), 2.53–2.59 (1H, m), 2.67–2.73 (1H, m), 2.75 (1H, dd, J = 7.1, 4.4 Hz), 3.20 (1H, br d, J = 6.3 Hz), 4.87 (1H, br s), 4.97 (1H, br s).
13C NMR (150 MHz, CDCl3) δ 21.4 (CH2), 23.4 (CH2), 25.2 (CH3), 28.6 (CH3), 30.6 (CH2), 34.0 (CH2), 43.5 (CH), 44.3 (CH), 63.8 (C), 86.3 (C), 110.3 (CH2), 143.9 (C), 173.4 (C), 212.1 (C).
MS (CI) m/z 235 [M + H]+ (base), 217, 191, 189, 179, 161, 147, 129, 89.
HR-MS Found m/z 235.1329 [M + H]+, Calcd for C14H19O3 235.1334.
Compound 3
Oil.
FTIR: 1760, 1730 cm−1.
1H NMR (600 MHz, C6D6) δ 0.83 (3H, s), 1.04 (3H, s), 1.26–1.29 (1H, m), 1.30 (3H, br s), 1.44 (1H, ddt, J = 12.9, 8.8, 3.7 Hz), 1.60–1.66 (1H, m), 1.76 (br dt, J = 18.1, 8.5 Hz), 2.15–2.22 (1H, m), 2.25–2.30 (1H, m), 2.54 (1H, br d, J = 5.8 Hz), 2.86 (1H, d, J = 8.2 Hz), 5.03 (1H, br s).
13C NMR (150 MHz, C6D6) δ 20.8 (CH2), 21.2 (CH3), 23.3 (CH3), 23.5 (CH2), 27.9 (CH3), 35.7 (CH2), 42.1 (CH), 43.7 (CH), 59.8 (C), 85.3 (C), 122.1 (CH), 132.7 (C), 174.3 (C), 212.5 (C).
MS (EI) m/z 234 [M+], 216, 206, 179, 165, 147, 137 (base), 119, 105, 91, 77, 65, 55, 43.
HR-MS Found m/z 234.1222 [M+], Calcd for C14H18O3 234.1256.
Compound 5
Oil.
FTIR: 1780 cm−1.
1H NMR (600 MHz, CDCl3) δ 1.37 (3H, s), 1.47 (1H, dd, J = 5.6, 4.3 Hz), 1.59 (3H, br s), 1.68 (3H, br s), 1.69∼1.72 (1H, m), 1.76∼1.82 (1H, m), 1.96 (1H, dd, J = 8.0, 4.3 Hz), 2.06 (2H, br q, J = 7.2 Hz), 2.57 (3H, s), 2.58 (1H, dd, J = 8.0, 5.6 Hz), 5.05∼5.07 (1H, m).
13C NMR (150 MHz, CDCl3) δ 17.7 (CH3), 21.7 (CH2), 22.5 (CH2), 22.6 (CH3), 25.6 (CH3), 29.3 (CH3), 38.5 (CH), 39.4 (C), 41.9 (CH2), 83.1 (C), 122.4 (CH), 133.0 (C), 172.3 (C = O), 200.8 (C = O).
MS (CI) m/z 237 [M + H]+, 219, 203, 191, 177, 153, 123 (base).
HR-MS (CI) Found m/z 237.1464 [M + H]+, Calcd for C14H21O3 237.1491.
Compound 6
Oil.
FTIR: 3470, 1740 cm−1.
1H NMR (600 MHz, CDCl3) δ 1.59 (3H, br s), 1.64 (3H, br s), 1.68 (3H, br s), 2.00–2.10 (4H, m), 2.27 (3H, s), 2.65–2.87 (2H, m), 3.79 (3H, s), 4.12 (1H, s), 4.95–5.10 (2H, m).
13C NMR (50 MHz, CDCl3) δ 16.4 (CH3), 17.7 (CH3), 24.8 (CH3), 25.7 (CH3), 26.4 (CH2), 34.1 (CH2), 39.8 (CH2), 53.3 (CH3), 84.0 (C), 115.9 (CH), 123.9 (CH), 131.7 (C), 140.4 (C), 171.2 (C = O), 204.6 (C = O).
MS (EI) m/z 268 [M]+, 250, 235, 225, 218, 207, 199, 191, 181 167, 81, 69 (base).
HR-MS (EI) Found m/z 268.1679 [M]+, Calcd for C15H2404 268.1675.
Compound 7
Oil.
FTIR: 1760, 1730 cm−1.
1H NMR (600 MHz, CDCl3) δ 1.59 (3H, br s), 1.63 (3H, br s), 1.67 (3H, br s), 1.98∼2.08 (4H, m), 2.38 (3H, s), 3.05∼3.12 (2H, m), 3.87 (3H, s), 5.01∼5.04 (1H, m), 5.13∼5.16 (1H, m).
13C NMR (150 MHz, CDCl3) δ 16.1 (CH3), 17.7 (CH3), 25.6 (CH3), 26.3 (CH2), 27.1 (CH3), 32.7 (CH2), 39.8 (CH2), 53.9 (CH3), 100.9 (C), 114.7 (CH), 123.6 (CH), 131.8 (C), 142.2 (C), 163.7 (C = O), 194.3 (C = O).
MS (CI) m/z 296 [M + H-2]+, 252, 219, 186, 167, 137, 129, 69 (base).
HR-MS (CI) Found m/z 296.1471 [M + H-2]+, Calcd for C15H22O5N 296.1498.
Compound 8
Oil.
FTIR: 1750, 1560 cm−1.
1H NMR (600 MHz, CDCl3) δ 1.59 (3H, br s), 1.65 (3H, br s), 1.67 (3H, br s), 1.98∼2.01 (2H, m), 2.04∼2.06 (2H, m), 2.84 (1H, dt, J = 14.5, 5.6 Hz), 3.02(1H, dt, J = 14.5, 9.2 Hz), 3.83 (3H, s), 5.02∼5.03 (1H, m), 5.04∼5.05 (1H, m), 5.08 (1H, dd, J = 9.2, 5.6 Hz).
13C NMR (150 MHz, CDCl3) δ 16.1 (CH3), 17.6 (CH3), 25.6 (CH3), 26.3 (CH2), 29.2 (CH2), 39.6 (CH2), 53.5 (CH3), 87.7 (CH), 115.6 (CH), 123.6 (CH), 131.8 (C), 141.7 (C), 164.8 (C = O).
MS (CI) m/z 254 [M + H-2]+, 238, 208, 207, 141, 137, 81, 69 (base).
HR-MS(CI) Found m/z 254.1383 [M + H-2]+, Calcd for C13H20O4N 254.1392.
Compound 9
Oil.
FTIR: 1750, 1720 cm−1.
1H NMR (600 MHz, CDCl3) δ 1.42 (1Η, dddd, J = 13.5, 10.2, 4.9, 4.1 Hz), 1.77 (3H, br s), 1.83 (1H, dddd, J = 13.5, 6.6, 5.5, 4.0 Hz), 2.10–2.16 (2H, m), 2.25–2.37 (4H, m), 2.96 (1H, t, J = 5.2 Hz), 3.50 (1H, br t, J = 5.2 Hz), 3.63 (3H, s), 4.72 (1H, br s), 4.86 (1H, br s), 4.91 (2H, m).
13C NMR (150 MHz, CDCl3) δ 22.8 (CH2), 23.6 (CH3), 27.7 (CH2), 29.8 (CH2), 35.0 (CH2), 43.5 (CH), 45.6 (CH), 52.3 (CH3), 66.8 (C), 110.0 (CH2), 113.5 (CH2), 145.2 (C), 145.4 (C), 169.4 (C), 211.4 (C).
MS (CI) m/z 248 (M)+, 216 (base), 188, 161, 147, 91.
HRMS (CI) Found m/z 248.1411 [M]+, Calcd for C15H20O3 248.1412.
Compound 10
Oil.
FTIR: 1750, 1715 cm−1.
1H NMR (600 MHz, CDCl3) δ 0.81, (3Η, s), 0.87 (3H, s), 0.90 (3H, s), 1.18 (1H, q, J = 7.2 Hz), 1.39 (2H, t, J = 7.2 Hz), 1.68–1.74 (1H, m), 1.81–1.86 (1H, m), 2.16 (1H, dq, J = 14.0, 10.2 Hz), 2.41 (1H, dt, J = 19.5, 10.2 Hz), 2.56 (1H, ddd, J = 19.5, 10.2, 2.4 Hz), 2.57 (1H, d, J = 4.8 Hz), 3.03 (1H, br d, J = 10.2 Hz), 3.70 (3H, s).
13C NMR (150 MHz, CDCl3) δ 14.4 (CH3), 19.2 (CH3), 19.3 (CH3), 19.4 (CH2), 23.2 (CH2), 28.2 (CH2), 41.0 (CH2), 50.5 (C), 51.5 (C), 52.4 (CH), 52.5 (CH3), 53.3 (CH), 68.1 (C), 172.1 (C), 217.8 (C).
MS (CI) m/z 250 [M+], 219 (base), 191, 175, 141.
HRMS (CI) Found m/z 250.1571 [M+], Calcd for C15H22O3 250.1569.
Compound 11
Oil.
FTIR: 3500, 1720 cm−1.
1H NMR (600 MHz, C6D6) δ 0.76 (1Η, qd, J = 12.0, 7.0 Hz), 0.95 (3H, s), 1.04 (3H, s), 1.27 (3H, s), 1.35–1.40 (1H, m), 1.52 (1H, ddd, J = 14.0, 12.0, 2.9), 1.57 (1Η, dd, J = 12.0, 2.1 Hz), 1.74 (1H, ddd, J = 14.0, 9.2, 7.0 Hz), 2.18 (1Η, td, J = 12.0, 7.0 Hz), 3.43 (3H, s), 6.17 (1H, br d, J = 2.1 Hz).
13C NMR (150 MHz, C6D6) δ 19.7 (CH3), 23.4 (CH2), 26.8 (CH3), 28.3 (CH3), 40.7 (CH2), 47.4 (CH), 47.7 (CH), 51.3 (CH3), 76.6 (C), 79.9 (C), 106.5 (CH), 146.0 (C), 163.7 (C).
MS (CI) m/z 240 [M+], 223 (base), 205, 163, 121.
HRMS (CI) Found m/z 240.1391 [M+], Calcd for C13H20O4 240.1362.

A plausible mechanism of one-electron oxidation of
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221109424 - Supplemental material for One-Electron Oxidation of Geranyl Acetone Derivatives Using Ceric(IV) Ammonium Nitrate and Manganese(III) Acetate: Carbon–Carbon Bond Formation
Supplemental material, sj-docx-1-npx-10.1177_1934578X221109424 for One-Electron Oxidation of Geranyl Acetone Derivatives Using Ceric(IV) Ammonium Nitrate and Manganese(III) Acetate: Carbon–Carbon Bond Formation by Masakazu Sono, Yui Yamashita, Mayu Hirai, Yayoi Nishio, Shigeru Takaoka and Motoo Tori in Natural Product Communications
Footnotes
Acknowledgments
We thank Dr Yasuko Okamoto, Tokushima Bunri University, for the measurement of MS spectra.
Author Contributions
Project administration and supervision, MS and MT; Reaction, isolation of compounds, and structure analysis, MS, YY, MH, and YN; X-ray analysis, ST; Writing, MS and MT.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This work was supported by the Tokushima Bunri University,
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
