Abstract
Autonomic nervous system disorders have many causes, and thus, no effective remedy for these disorders has been established. In this study, we conducted animal experiments with essential oil from Pelargonium graveolens (EOPG, geranium essential oil), which may regulate the autonomic nervous system. EOPG was administered to awake mice by inhalation. Heart rate variability (HRV) was analyzed from the pulse wave of the mice on an elevated platform, and the ratio of sympathetic to parasympathetic nerve activity was determined. The results showed that inhalation of EOPG did not affect the heart rate but made activity of the parasympathetic nervous system dominant. This effect was thought to be due to linalool, a component of EOPG that was transferred into the brain and body.
Introduction
Dysautonomia is defined as an imbalance between the sympathetic and parasympathetic autonomic nervous systems, and results in persistent mental and physical discomfort. Anxiolytics and antidepressants may be used for dysautonomia. However, due to the wide variety of causes, 1 including other illnesses and stress, partial remedies exist, but an effective cure does not.
Aromatherapy using plant essential oils is widely used for treatment and mental relaxation through scent. 2 Among aromatherapies, essential oil from Pelargonium graveolens (EOPG, geranium essential oil) may help regulate the autonomic nervous system. In aromatherapy, EOPG has been empirically used to regulate the autonomic nervous system. However, insufficient scientific evidence exists for the effects of EOPG. Scientific research reports on EOPG have shown anti-tumor activity against breast cancer 3 and anti-inflammatory effects. 4 Our group has reported that EOPG lowers blood pressure and heart rate in mice under anesthesia. 5
Various methods can be used to assess the autonomic nervous system. 6 Among them, we focused on heart rate variability (HRV), which can be used to evaluate the autonomic nervous system status relatively objectively in real time. HRV can be used to describe the ratio of sympathetic to parasympathetic nerve activity based on fluctuations in the R-R interval of the heartbeat and has been studied in humans and animals. 7
The current study was conducted to clarify the effects of EOPG on the autonomic nervous system in awake animals (Experiment 1). Because the effects of psychological effects arising from perceptual responses to olfaction are strong in humans, we used mice to examine the effects on the autonomic nervous system.
In addition, this study examined the effects of inhalation administration, which involves both neurological pathways from olfactory stimulation and pharmacological pathways in which compounds are transferred to the brain and other parts of the body. In this study, we focused on the pathway by pharmacological transmission, which has a clear pharmacological effect. 5 To clarify the mechanism of action of EOPG, we investigated the migration of EOPG compounds into the body (Experiment 2).
Experiment 1
To measure the autonomic nervous system of mice, we measured pulse waves from the neck using a pulse oximeter, which is less burdensome for mice. Because pulse wave and heart rate are correlated, we analyzed HRV from pulse waves and examined the balance between activity of sympathetic and parasympathetic nerves, which are indicators of the activity of the autonomic nervous system. To measure pulse waves in awake mice, we wanted the mouse to move as little as possible. Therefore, we applied the elevated platform test, 8 which is used to evaluate freezing behavior, to measure pulse waves of awake mice.
Mice were randomly divided into the Control group (n = 8) and EOPG group (5 μL EOPG/L air, n = 9) for the experiment. Mice from both groups were placed on an elevated platform, and pulse waves were measured for 10 min. From the pulse waves obtained, the heart rate and the ratio of sympathetic to parasympathetic nerve activity were analyzed (the first time). The results showed no difference in heart rate and sympathetic and parasympathetic activity ratios between the 2 groups, as expected (Figure 1A, Figure 2A and B).

Heart rate (BPM, beats per minute) on an elevated platform before and after 90-minute inhalation of 5 μL water/L air or 5 μL EOPG/L air. (A) Heart rate before inhalation (the first time) and (B) Heart rate after inhalation (second time).
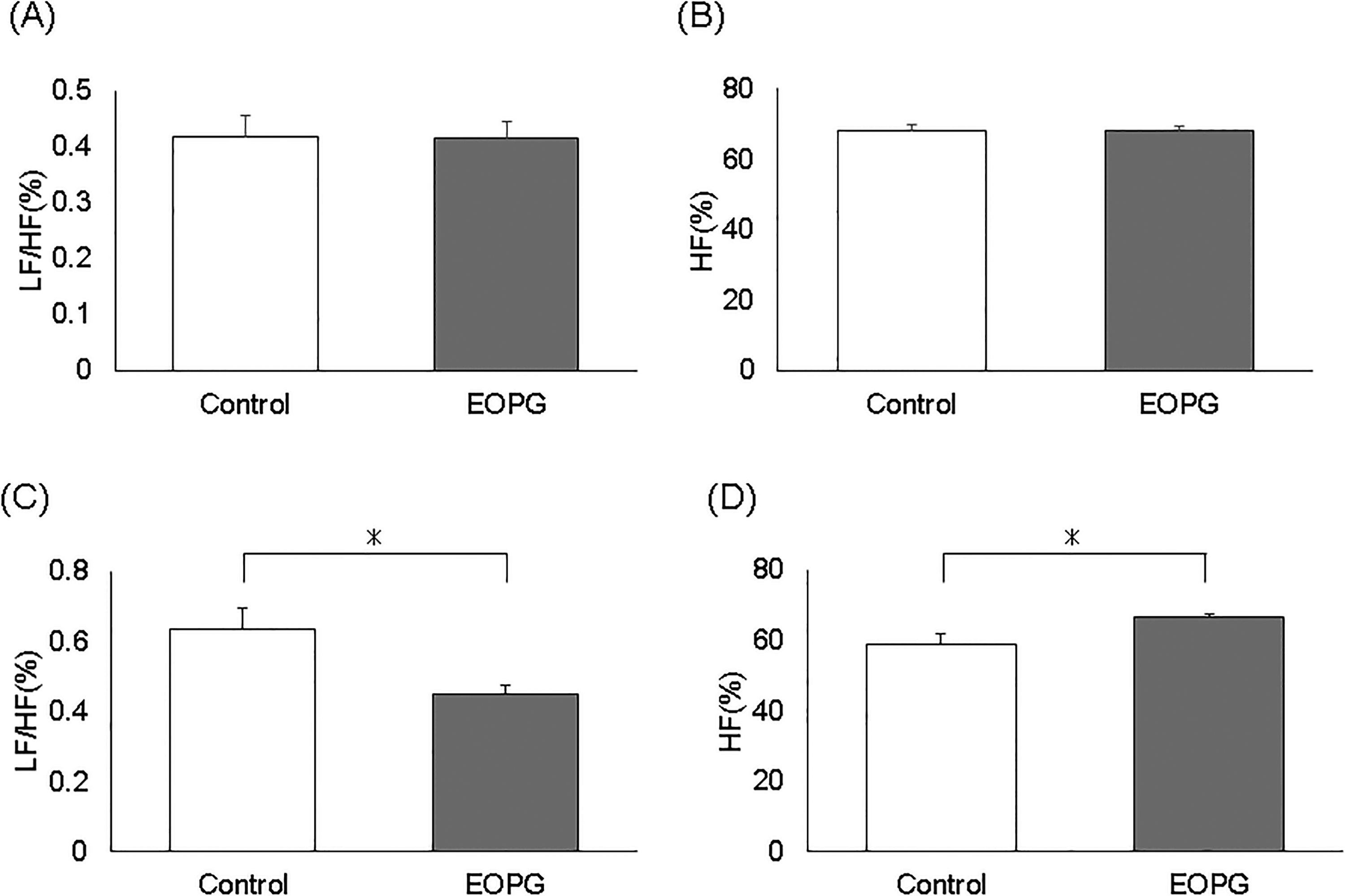
Parasympathetic (high frequency [HF] (%)) and sympathetic (low frequency [LF]/HF (%)) ratio of mice on an elevated platform before and after 90-minute inhalation of 5 μL water/L air or 5 μL EOPG/L air. (A) Percentage of sympathetic nerves stimulated before inhalation (the first time). (B) Percentage of parasympathetic nerves stimulated before inhalation (the first time). (C) Percentage of sympathetic nerves stimulated after inhalation (second time). (D) Percentage of parasympathetic nerves stimulated after inhalation (second time).
Next, each group of mice was administered 5 μL water/L air (control group) or 5 μL EOPG/L air (EOPG group) by inhalation for 90 min. Subsequently, 0.3 mL saline was administered intraperitoneally to mice in both groups. After 30 min, mice from both groups were placed on an elevated platform for the second time, and pulse waves were measured for 10 min. Then, the heart rate and the ratio of parasympathetic to sympathetic nerve activity of mice in both groups were analyzed from these pulse waves as in the first experiment (second time).
The results showed no difference in heart rate between the 2 groups (Figure 1B). On the other hand, sympathetic nerves were significantly inhibited in the EOPG group (0.45 ± 0.02%) compared to the control group (0.63 ± 0.06%) (Figure 2C) (P = .012). The parasympathetic nerve activity was significantly increased in the EOPG group (66.5 ± 1.2%) compared to the control group (59.1 ± 2.8%) (Figure 2D) (P = .022). In other words, inhalation of EOPG suppressed the activity of the sympathetic nervous system and increased the activity of the parasympathetic nervous system, thereby reducing the fear of the mice on the elevated platform.
Experiment 2
EOPG contains 6.3% linalool. To clarify the mechanism of action of the effects of EOPG inhalation on the autonomic nervous system, the transfer of linalool into the brain was examined. In the present conditions of inhalation of EOPG, linalool was not detected in the brain because it was below the detection limit of gas chromatography/mass spectrometry (GC-MS). Intraperitoneal administration of linalool was expected to produce a high detectable concentration, and linalool was detected in the brain at a concentration of 339.9 ± 74.3 pg/g brain as shown in Table 1. In other words, linalool, a component of EOPG, was sufficiently transferred into the brain through the blood–brain barrier if it is transferred into the blood vessels.
Brain Concentration of Linalool 30 min After Intraperitoneal Administration of 2.9 mg Linalool/g Mouse (100 μL/Mouse).

Values are the means ± standard error, n = 4.
Discussion
Inhalation of geranium essential oil suppressed the activity of the sympathetic nervous system and increased the activity of the parasympathetic nervous system in awake mice. We believe that these results support the hypothesis that EOPG balances the autonomic nervous system. In our previous studies, we reported that inhalation of EOPG significantly decreases blood pressure and heart rate in anesthetized mice (the mixture was 0.3 mg/kg medetomidine hydrochloride, 5 mg/kg butorphanol tartrate, and 4 mg/kg midazolam). 5 In the awake mice in the current study, inhalation of EOPG had no effect on heart rate, suggesting that the interaction of EOPG with anesthetics resulted in a decrease in blood pressure and heart rate.
Linalool, a component of EOPG, has antidepressant-like effects in the central nervous system 9 and vasodilatory effects in the periphery. 10 Both lavender essential oil and linalool have been reported sedative and anxiolytic-like effects in the elevated-plus maze test using lavender essential oil and linalool alone. 11 In the present study, linalool was detected in the brain following intraperitoneal administration. This indicates that if linalool is transferred into the blood via inhalation, it will be readily transferred into the brain. Low-molecular weight compounds with alcohol such as linalool (citronellol and geraniol) are difficult to detect in the brain because they are metabolized quickly after entering the body via inhalation administration. On the other hand, these low-molecular weight compounds with alcohol have been reported to easily cross the blood–brain barrier and transfer to the brain once they enter the bloodstream. In addition, migration into organs differs depending on the type of compound, and linalool is less easily transferred into the brain by inhalation compared to limonene and other compounds. 12 Regarding the mechanism of action, inhalation of EOPG is thought to have caused linalool to migrate into the brain via the blood and also to affect the autonomic nervous system.
On the other hand, the current study focuses on the pharmacological transmission pathway, but the experiment does not exclude the sense of smell in mice. In other words, the involvement of olfactory effects in the effect of geranium essential oil in this study cannot be completely ruled out. Reports have shown that scent stimulation with linalool causes a decrease in blood pressure in rats, 13 and that the scent of linalool makes the parasympathetic nervous system dominant in humans. 14 In addition, this study is only an animal experiment. Due to species differences between humans and animals, results in clinical studies may vary. Further research is expected in the future.
Experimental Methods
Samples
EOPG was purchased from GREEN FLASK Co., Ltd (Tokyo, Japan). The EOPG used mainly contains 21.9% citronellol, 13.3% geraniol, 8.6% citronellyl formate, and 6.3% linalool. 5
Animals
Male ICR strain mice were used in the experiments. The cages were placed in a room illuminated by fluorescent lights. The room was set to a 12-hour light/dark schedule (light period from 7:00 to 19:00) and a room temperature of 24 ± 5 °C. Mice had free access to food (Labo MR Stock, Nosan Corporation) and water. Four-week-old mice were purchased from Japan SLC Co., Ltd (Shizuoka, Japan), and 1 week later, 22 5-week-old mice (about 30 g) were used. Animal experiments were conducted with the approval of the International University of Health and Welfare Experimental Animal Committee (Animal Experiment Protocol Approval Number: No. 17004).
Pulse Wave Measurement Procedure
To set up the pulse oximeter, the hair around the neck of the mice was shaved the day before the test. For the elevated platform, we used a modified version of the method of Ito et al. 15 An elevated platform (height: 40 cm, diameter of the observation surface: 5 cm) was used for the test, and the apparatus was surrounded by a 55-cm high wall to prevent escape. MouseOx® PLUS (Starr Life Sciences Corp.) was used to measure the pulse wave of the mice, and PowerLab 2/26 (AD Instruments Japan Inc.) and LabChart 8 (ADInstruments Japan Inc.) were used to analyze the pulse wave. On the day of the test, mice were placed on an elevated platform for 10 min, and their pulse waves in normal conditions were measured from around the neck (the first test). The mice inhaled 5 μL water/L air or 5 μL EOPG/L air in a 10-L sealed container for 90 min. The mice were intraperitoneally injected with 0.3 mL/mouse of saline and left in an open container for 30 min. The mice were then placed on the elevated platform again, and the pulse wave around the neck was measured for 10 min using the same device as the first measurement (second test).
HRV Analysis
LabChart 8 was used to analyze the heartbeat from the pulse wave. Very low frequency (VLF), low frequency (LF), and high frequency (HF), which reflect sympathetic and parasympathetic nerve activity, were set as follows:
VLF: 0 to 0.15 Hz, LF: 0.15 to 1.5 Hz, HF: 1.5 to 5 Hz. Sympathetic activity was expressed as LF/HF and parasympathetic activity as HF. Experimental procedures for the transition of volatile compounds into the brain
Linalool (Tokyo Chemical Industry Co., Ltd) was diluted with olive oil (KENEI Pharmaceutical Co., Ltd) to make the sample. Mice were administered linalool intraperitoneally at a concentration of 2.9 mg/g (100 µL/mouse), and 30 min later, brains were sampled to measure linalool concentration. Each brain collected was treated with 2 mL hexane and processed with an ultrasonic homogenizer THU-80 (AS ONE CORPORATION). The obtained sample was centrifuged at 2000 rpm for 5 min (Kokusan Co., Ltd) and then dehydrated with magnesium sulfate (FUJIFILM Wako Pure Chemical Corporation) to make the sample solution. The sample solutions were analyzed by GC-MS (SHIMADZU CORPORATION). The mobile phase for GC-MS was ultra-pure helium (99.99995%, 1.82 mL/min), and the column was a DB-5 ms capillary column (30 m × 0.25 mm ID, 0.25 μm, nonpolar column; Agilent Technologies Inc.). Inlet line temperature and source temperature of GC-MS were set at 250 °C. The set temperature of the column oven was held at 40 °C for 2 min, increased to 200 °C at 5 °C/min, and held at 200 °C for 2 min. The chemical components were identified by comparing NIST 02 spectral library (National Institute of Standards and Technology), comparing the 1095 linear retention indices presented by Adams,
16
and by direct comparison with standard compounds (linalool, citronellol, and geraniol). Quantitative analysis was performed by the absolute calibration method of standard compounds.
Statistical Analysis
Statistical analysis was performed using Excel statistics (Bell curve in Excel, Social Survey Research Information Co., Ltd). The statistical significance was determined by the two-sided paired t-test, and P < .05 was considered significant.
Footnotes
Acknowledgments
The authors would like to thank Mr Shinichiro Hayashi of GREEN FLASK Co., Ltd for providing the EOPG.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the JSPS KAKENHI (grant number JP20K11562).
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
