Abstract
Polymethoxyflavones (PMFs) are flavonoid compounds present in citrus plants that are proposed to be advantageous to human health. However, the advantageous effects of PMFs in the context of renal dysfunction are unclear. In this study, we made a PMF-rich fraction (PRF) from the leaves of Citrus sunki Hort ex. Tanaka and identified its components using liquid chromatography and mass spectrometry. We then investigated the effect of PRF—comprising 9 types of PMF—on renal dysfunction induced by unilateral ureteral obstruction (UUO) in mice. Animals were divided into four experimental groups (n = 7 per group): I) sham-operated group (Sham); II) UUO group (UUO); III) UUO + Enalapril 0.1 mg/1 mL (UUO + Enap); IV) UUO + PRF 100 mg/kg/day (UUO + PRF). All mice were orally administered with the drugs once a day from 7 days before UUO to 1 week after UUO. After the experiments were over, serum and tissues were taken for biochemical and histological analysis. PRF promoted the recovery of body weight in the background of UUO. Biochemical and histological analysis revealed that PRF ameliorated UUO-induced renal dysfunction and moderately reversed inflammation and tubulointerstitial fibrosis. Further, PRF inhibited the expression of endothelial nitric oxide synthase (eNOS), inducible nitric oxide synthase (iNOS), cyclooxygenase-2 (COX-2), transforming growth factor-β (TGF-β), collagen I (Col-I), and collagen IV (Col-IV). These results suggest that PRF improves UUO-induced renal dysfunction by regulating the expression of inflammatory and fibrotic response-related genes.
Introduction
Chronic kidney disease (CKD) is a serious, worldwide health problem with a prevalence reaching 13.4% annually. 1 Patients with CKD suffer from frequent complications and comorbidities, resulting in poor quality of life. 2 CKD is related to the development of secondary disorders such as diabetes, hypertension, and an elevated risk of autoimmune diseases. Moreover, these diseases are hardly diagnosed at an early stage, and once onset, it is difficult to reverse the progress of these conditions. It is known that the major pathological changes of CKD are progressive nephrotic inflammation and fibrosis, in which macrophages play a fundamental role. 3 In addition, free radicals formed by inflammatory cells cause fibrosis development by stimulating epithelial-mesenchymal transition (EMT). 4
Inflammation and fibrosis are self-protective host cell responses against infection and injury, which recruit the pro-inflammatory factors, pro-fibrotic factors, and immune cells to the injury.5,6 These factors cause inflammatory and fibrotic cascades to augment and interact with other signaling molecules. Intermediation between inflammatory and fibrotic processes is important for facilitating the repair of tissue structure and function.7–9 Immune cells, including M1 subtype macrophages, penetrate the kidney, and initiate and maintain inflammatory processes in the early stage of unilateral ureteral obstruction (UUO). M1 macrophages shift to the M2 subtype, which contribute to the repair of the damaged tissue. 9 The shift in M1 to M2 macrophage subtype encourages fibrosis by inducing cytokines, chemokines, and growth factors such as platelet-derived growth factor (PDGF) and transforming growth factor-β (TGF-β), resulting in the emergence of a pro-fibrotic phenotype in macrophages, which functions in chronic inflammation and fibrosis.10–13 Thus, preventing inflammation and fibrosis may ameliorate chronic kidney disease.
Polymethoxyflavones (PMFs) are a group of flavonoid compounds containing more than two methoxyl groups which have been nearly exclusively found in citrus plants, although they are also found in many other plants. PMFs exhibit a wide range of beneficial effects, including anti-obesity, anti-atherosclerosis, anti-inflammation, neuroprotection, anti-cancer, anti-microbial, and anti-oxidation.14–21 In particular, PMFs possess the ability to alleviate inflammatory injury in the kidney, triggered by cisplatin in rats by decreasing the tumor necrosis factor-α (TNF-α) level and augmenting interleukin 1α (IL-1α) cytokines. 22 However, it remains unclear whether PMFs can protect UUO mice from inflammation and fibrosis. Although PMFs have been identified in various tissues of citrus plants, most studies have been conducted using citrus peels; however, PMFs from citrus leaves have not been investigated. In our previous study, we reported the anti-obesity effects of the PMF-rich fraction (PRF) prepared from the leaves of Citrus sunki Hort ex. Tanaka. 23 In the present study, we characterized the PMF-rich fraction (PRF) and investigated the effect of this on renal dysfunction induced by unilateral ureteral obstruction (UUO) in mice.
Results and Discussion
Chemical Characteristics of PRF
PMFs have a structure in which several methoxy groups are bonded to a 15-carbon skeleton containing two phenyl rings (A and B), and one heterocyclic ring (C, a ring containing internal oxygen). To identify the PMFs contained in PRF, we analyzed the retention time pattern on the HPLC chromatogram and the LC/MS. Each PMF separated by HPLC exhibited unique MS fragment characteristics dependent on the number and position of the methoxyl or hydroxyl substituents. 24 In particular, the MS of PMFs showed a general tendency to lose nCH3 (15 or 30), CH4 (16), H2O (18), and CO (28). Further, ions at m/z 211.0602 (1,3A+) and 211.0604 (1,3A+) show that there are three methoxy substituents on ring A regarded as the ion of flavanone (Figure 1). 24 As shown in Figures 1 and 2, we identified 9 PMF in the PRF; peaks 1, 2, 3, 4, 5, 6, 7, 8, and 9 represented sinensetin, 5,6,7,4′-tetramethoxyflanavone, 5,7,8,3′,4′-pentamethoxyflavanone, 35,7-trimethoxyflavone, nobilentin (5,6,7,8,3′,4′-hexamethoxyflavone), 3,4,6,78,3′,4′-heptamethoxyflavones, tangeretin (5,6,7,8,9′-pentamethoxyflavone), 5-demethylnobiletin, and 5-demethyltangeretin, respectively.24,25
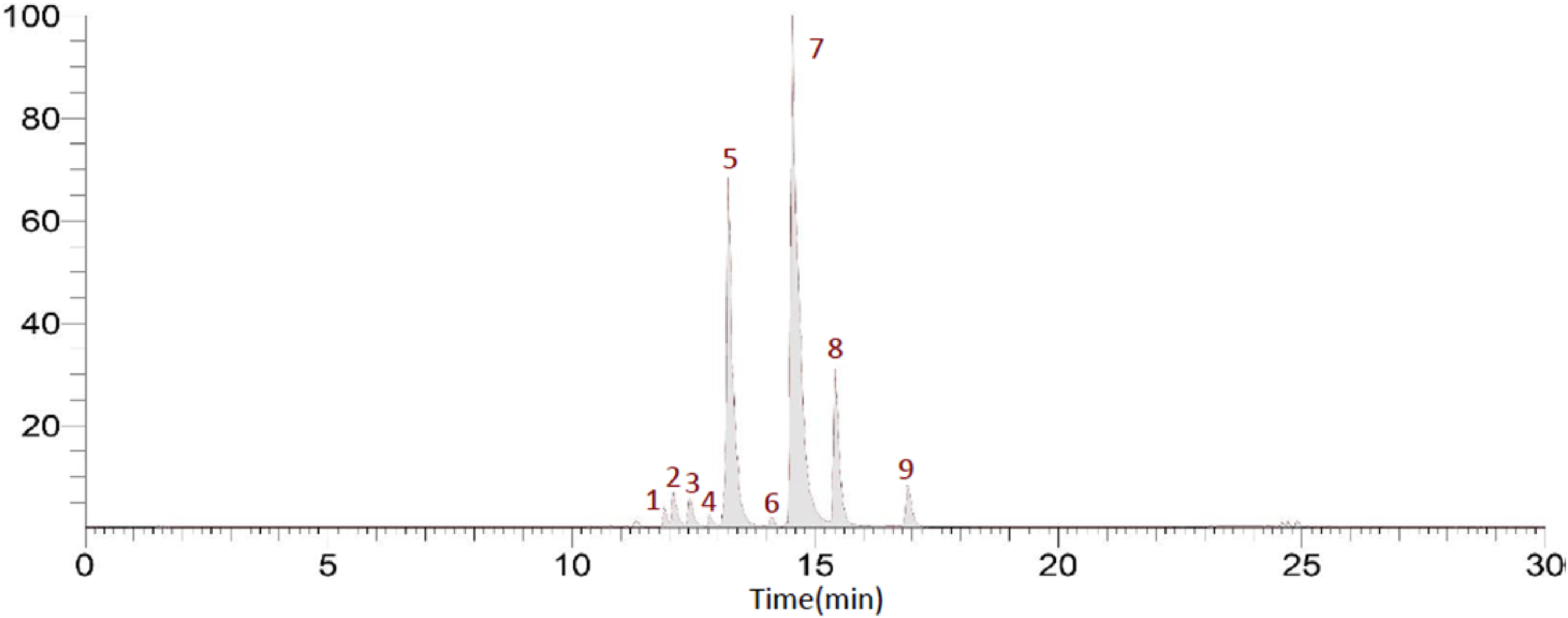
Polymethoxyflavone (PMF) compounds identified in the PMF-rich fraction (PRF) using HPLC and an LTQ XL linear ion trap mass spectrometer.

Mass spectra and fragmentation patterns of different polymethoxyflavone (PMF) compounds contained in the PMF-rich fraction (PRF). The peak numbers in this figure correspond to those used in Figure 1.
PRF Relieves the Symptoms of UUO-Induced Chronic Kidney Disease (CKD)
The protective effect of PRF against UUO-induced renal dysfunction was assessed by measuring bodyweight changes among the experimental groups. Figure 3A shows the body weight changes measured during 14 days. The bodyweights of the UUO, UUO + Enap [enalapril 0.1 mg/mL, an angiotensin converting enzyme (ACE) inhibitor], and UUO + PRF (100 mg/kg/day) groups decreased significantly until 3 days after UUO operation comparing with the Sham group. However, the Sham, Enap (positive control), and PRF groups promptly recovered the bodyweight lost by 7 days after UUO operation in comparison with that of the UUO group. The representative renal function evaluation markers were then compared among the groups. The levels of serum creatinine, blood urea nitrogen, urine creatinine, and urine urea nitrogen were increased in the UUO group, but their levels were significantly decreased in the Enap and PRF groups (Figure 3B to E). PRF decreased the albuminuria level and glomerular filtration rate (GFR), and in mice with UUO-induced renal function to levels similar to those with the ACE inhibitor, enalapril (Figure 3F and G). These results are consistent with the report that the interstitial fibrosis causes significant protein loss, increases the proportion of excreted protein, and increases urine creatinine levels above serum, and affects the glomerular filtration rate (GFR)26,27

PRF improved gradual recovery and renal function in unilateral ureteral obstruction (UUO)-treated mice. (A) Body weight changes, (B) serum creatinine, (C) urine creatinine, (D) blood urea nitrogen, (E) urine urea nitrogen, (F) albuminuria excretion levels and (G) glomerular filtration rate (GFR). Data are presented as means ± SD (n = 7). * p < .05, ** p < .01, **** p < .0001 compared with the Sham group. # p < .05, ## p < .01, ### p < .001, #### p < .0001 compared with the UUO group.
UUO represents a typical and common clinical problem of CKD, wherein urine flow is physically blocked by kidney stones or various causes, leading to parenchymal damage and hydronephrosis.28,29 Urea nitrogen and creatinine are widely known as significant renal function evaluation indicators. 30 This result demonstrated that PRF improves renal excretion and reabsorption. Renin-angiotensin-aldosterone system (RAAS) inhibitors effectively limit initial hyperfiltration and reduce systemic blood pressure, which is widely known to aggravate renal disease progression in rats treated with angiotensin-converting enzyme (ACE) inhibitors.31-34 Moreover, detection of proteinuria related to high ACE levels suggested increased angiotensin II production in the proximal tubule. PRF was confirmed to effectively reduce persistent proteinuria, which can promote tissue RAAS activation, suggesting that PRF might have potential as a therapeutic adjuvant in conjunction with RAAS treatment.
PRF Ameliorates UUO-Induced Renal Fibrosis
Histopathological staining was performed to evaluate the effect of PRF on UUO-induced renal fibrosis (Figure 4). The UUO group showed decreased glomerular size and renal cortical damage because of chronic renal failure induction compared with the Sham group. However, the sizes and shapes of glomeruli in the Enap and PRF groups were considerably different compared with the UUO group (Figure 4A and D). This indicated that PRF effectively helped repair the renal interstitial damage induced in response to UUO. Trichrome staining revealed that UUO-induced collagen deposition was suppressed by PRF, as in the Enap group, which served as the positive control (Figure 4B and E). Consistent with the collagen deposition data, the expression of α-smooth muscle actin (α-SMA) was reduced upon the administration of PRF and Enap in UUO-treated mice (Figure 4C and F), indicating that PRF, like Enap, reduced the irreversible glomerular damage as well as collagen and α-SMA accumulation.

PRF relieves glomerular damage and decreases collagen deposition in UUO-induced renal fibrosis. (A) Histopathology of renal fibrosis, (B) collagen deposition, (C) α-smooth muscle actin (α-SMA), (D) glomerulus size, (E) collagen deposition positive area, and (F) α-SMA expression positive area. Data are presented as means ± SD (n = 3). * p < .05, ** p < .01, *** p < .001, **** p < .0001 compared with the Sham group. # p < .05, ## p < .01, ### p < .001, #### p < .0001 compared with the UUO group.
PRF Delays UUO-Induced Fibrosis by Inhibiting the Expression of Inflammatory Factors
To search the underlying mechanism of the relieving effect of PRF on UUO-induced fibrosis, the level of inflammation-related proteins among the groups was compared by Western blot analysis. The expression of inflammatory mediators such as iNOS, eNOS, TGF-β, and COX-2 was considerably diminished in the Enap and PRF groups comparing with the UUO group (Figure 5A to E). Further, the levels of E-cadherin, collagen I, and collagen IV in UUO-treated mice was significantly decreased by Enap and PRF administration (Figure 5A, and F to H). This result indicated that PRF, like Enap, delayed the progression of UUO-induced fibrosis by restoring blood flow control and suppressing the UUO-induced inflammation.

PRF suppressed the expression of inflammatory mediators in UUO-induced fibrosis. (A) Western blot analysis of inflammatory mediators, (B) iNOS, (C) eNOS, (D) TGF-β, (E) COX-2, (F) E-cadherin, (G) collagen I, and (H) collagen IV expression levels. Data are presented as means ± SD (n = 3). * p < .05, ** p < .01, *** p < .001, **** p < .0001 compared with the Sham group. # p < .05, ## p < .01, ### p < .001, #### p < .0001 compared with the UUO group.
Nitric oxide (NO) has a primary role in regulating blood flow in both normal and diseased kidneys. 35 In UUO, renal vasculature also responds to NO-mediated vasodilation, and UUO-associated blood flow changes involve damage of the renal NO synthetic pathways. Expression of eNOS and iNOS increases gradually during the renal obstructed period, resulting in neovascularization, including interstitial fibrosis. 36 Adjusting excessive inflammation is expected to have a potential therapeutic effect of inhibiting progressive renal fibrosis. 33 In the angiotensin II (ANGII) system, the expression and synthesis of ECM proteins such as collagen and α-SMA were achieved by a TGF-β-mediated mechanism. 37 These profibrotic cytokines and growth factors, sequentially, recruit and activate myofibroblasts, leading to progressive glomerular and interstitial fibrosis and late stage renal disease. 38 Further, infiltration of the glomeruli and interstitium by mononuclear cells usually occurs in predominantly progressive renal failure and plays an essential role in inducing irreversible structural changes.39-42
In conclusion, nine PMF compounds were identified in the PRF prepared from C. sunki leaves. The PRF alleviated UUO-induced renal dysfunction through an anti-inflammatory effect. Therefore, administration of PRF might provide a promising therapeutic approach for alleviating the symptoms of renal fibrosis and decelerating disease progression.
Materials and Methods
Preparation of PMF-Rich Fraction (PRF)
The leaves of Citrus sunki Hort ex. Tanaka were gathered in Jeju-do, Korea in February 2021. The leaves were cleaned, dried overnight at 60 °C, and pulverized. The PMF-rich fraction (PRF) was made as described by Jin et al 23 Concisely, the granulated powder was extracted with water at 100 °C for 5 h. Then the hot water extract was mixed with the same amount of n-hexane. The hexane layer was recovered, concentrated, freeze-dried, and stored at −20 °C.
Liquid Chromatography and Mass Spectrometry
The chemical composition of PRF was analyzed using an Accela HPLC system with a binary pump, PDA detector, and autosampler (Thermo Scientific, Bremen, Germany). The PRF was separated on an Accquity UPLC@BEH C18 column (50 mm × 2.1 mm i.d., 1.7 μm). The column was retained at 30 °C with a flow rate of 0.4 μL/min. Polymethoxyflavones were monitored at 200 to 500 nm. The mobile phase comprised 0.1% (v/v) formic acid in water (A) and formic acid in acetonitrile (B) with the elution gradient set as follows: 0–10 min, 5–60% B; 10–20 min, 60–80% B; 22–25 min, 100% B; 25–30 min, 100–5% B. The flow rate was set at 0.4 mL/min. The ionized components of PRF were examined using an LTQ XL linear ion trap mass spectrometer (Thermo Scientific, Bremen, Germany) connected to the LC system via an electrospray ionization (ESI) interface. Full scanning was carried out from m/z 150 to 1500 in the positive ionization mode ([M + H]+). The main parameters were set as follows: spray voltage of 3.5 kV; capillary voltage of 20 V; capillary temperature of 350 °C. Data processing for mass spectrometry was accomplished using Xcalibur data 2.1 software (Thermo Scientific, Bremen, Germany). Deviations from the calculated mass were less than 3 ppm in all measurements (data not shown).
Animals and Experimental Design
Eight-week-old male C57BL/6J mice were purchased from Cronex Inc. (Gyeonggi-do, Korea), and acclimated for one week with standard rodent food (Oriental Bio, Gyeonggi-do, Korea) and with free access to drinking water, under a 12-h/12-h light/dark cycle at 23 ± 2 °C and 60 ± 5% humidity. The experiments were approved by the Institutional Animal Care and Use Committee of Jeju National University (No. 2021-0010). The animals were randomly distributed into four experimental groups (n = 7): I) sham-operated group (Sham), II) unilateral ureteral obstruction (UUO)-operated group (UUO), III) UUO + Enalapril group (positive control), and IV) UUO + PRF group. In the sham and UUO operated groups, only 0.5% carboxy methyl cellulose (CMC, Sigma Aldrich Korea) was administered, whereas in the PRF group, 0.5% CMC mixed with PRF (100 mg/kg/day) was orally administered. The Enalapril group (positive control) received enalapril at 0.1 mg/mL/day via drinking water (100 mg of enalapril/L of water).43,44 All mice were orally administered once a day from 7 days before to 1 week after UUO surgery. For UUO, mice were anaesthetized with isoflurane (Vspharm, Gyeonggi, Korea). After removing the hair around the ribs, the right kidney was shown through the site of the right flank incision. The exposed right ureter was obstructed fully near the renal pelvis using a Black 6-0 silk tie. Sham-operated mice undertook the same surgical procedure excluding ureter ligation. 45 All mice were sacrificed with aerosol anaesthetic isoflurane; the kidneys were then perfusion-fixed in 10% neutral buffered formalin (10% NBF, Biosesang, Gyeonggi, Korea) for histological analyses or frozen in liquid nitrogen for biochemical analyses. To separate the serum, the coagulated blood was centrifuged at 3000 × g for 20 min. The separated serum was deposited at −70 °C until analysis.
Biochemical Parameter Assays
The serum and urine creatinine levels were determined using commercial kits from Abnova (CA, USA). The levels of blood urea nitrogen (BUN) and urine urea nitrogen (UUN) were measured using commercial kits from Asan Pharm (Gyeonggi, Korea), following the manufacturer's protocol. The albuminuria levels were assessed using electrophoresis on sodium dodecyl sulfate (SDS)–polyacrylamide gels and routine staining with Coomassie brilliant blue R 250 (Sigma-Aldrich, Missouri, USA). The glomerular filtration rate (GFR) values were measured using a reported protocol. 27
Histological Analysis
The kidney tissues were dissected and fixed overnight in 10% neutral buffered formalin, dehydrated through an ethanol series, embedded in paraffin at room temperature, and cut into 7 μm-thick slices using a microtome (RM2125RTS, Leica, Germany). Sections of kidney tissue were then stained using hematoxylin and eosin (H&E), Masson's Trichrome, and immunohistochemistry (IHC) staining methods. Photographs were taken using a light microscope (Ni-U, Nikon, Japan).
Western Blot Analysis
The kidney tissues were homogenized in cold lysis buffer (1 × RIPA buffer; 1 mM Na3VO4, 1 mM NaF, 1 mM phenylmethylsulfonyl fluoride, 1 μg/mL aprotinin, 1 μg/mL pepstatin, and 1 μg/mL leupeptin); the lysates were then obtained by centrifugation. Protein concentrations were evaluated using a protein assay kit (Bio-Rad, Hercules, CA, USA). Proteins were separated by electrophoresis on SDS-polyacrylamide gels and transferred to polyvinylidene fluoride (PVDF) membranes. The membranes were blocked with 5% (w/v) skimmed milk and 0.1% (v/v) Tween 20 in Tris-buffered saline (TBST), then incubated overnight at 4 °C with the following antibodies; transforming growth factor-β1 (TGF-β; Abcam, Cambridge, UK), endothelial NOS (eNOS; Abcam), inducible nitric oxide synthase (iNOS; Abcam), E-cadherin (E-cad; Abcam), Collagen-I (Col-I; BD, New Jersey, USA), Collagen-IV (Col-IV; BD), cyclooxygenase-2 (COX-2; Cell signaling, Beverly, MA, USA), and glyceraldehyde 3-phosphate dehydrogenase (GAPDH; Santacruz, CA, USA). The membranes were rinsed with 0.01% TBST and reacted at room temperature for 1 h with a peroxidase-conjugated secondary antibody (Vector Laboratories, Burlingame, CA, USA). The membranes were washed with 0.01% TBST; the proteins were then detected using Westar ETA C 2.0 substrate (Cyanagen, Bologna, Italy).
Statistical Analysis
The experimental results are presented as the means ± standard deviation. Variable variances were analyzed using the t-test with GraphPad Software (version 9.3.1, GraphPad Software, San Diego, CA, USA). Differences between groups were judged statistically significant at p < .05, using Duncan's multi-test method.
Footnotes
Author Contribution (Roles)
Conceptualization, investigation, and writing—original draft preparation, S.B and M.G.J.; methodology and investigation, H.C.K. and J.-W.K. and M.H.N and S.A.P and S.-P.H; writing—review and editing, supervision, project administration, and funding acquisition, S.-J.K. All authors have read and agreed to the published version of the manuscript.
Ethical Approval
All animal experiments were conducted in accordance with protocols approved by the Institutional Animal Care and Use Committee of Jeju National University (2021-0010).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Korea, (grant number Basic Science Research Program/2020R1I1A3A04037169);This study was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) by the Ministry of Education (NRF-2020R1I1A3A04037169 and NRF-2021R1I1A1A01055732).
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
