Abstract
Chelerythrine (CHE), one of the main active components of the medicinal plant, Chelidonium majus, (Figure 1) Botanical authority is traditionally used as a natural medicine for its significant antitumor activity. The relevant literature on the antitumor activity of CHE has been reviewed to provide a theoretical basis for further study and utilization. This review aimed to provide new ideas for developing tumor-targeted drugs.
Introduction
Chelidonium majus, a plant in the Papaveraceae family, is a traditional Chinese herbal medicine with an analgesic effect and is used to relieve cough and ulcer pain, for disinfection, and as a diuretic. Chelerythrine (CHE) (Figure 2) is one of the main effective active components of C majus. Major pharmacologically relevant components, most of which were isolated, are isoquinoline alkaloids. CHE has been found to be antibacterial, 1 antiparasitic, 2 anti-inflammatory, 3 antiplatelet, 4 anti-diabetic, 5 and to relieve stomach ulcers induced by ethanol. 6 It also provides protective effects against lipopolysaccharide (LPS)-induced endotoxic shock, 7 among other biological activities. Moreover, Chmura et al 8 reported that CHE is cytotoxic to 9 types of tumor cells (eg, MCF7, MCF7ADR, H329, DaOY, LnCaP, SQ120B, JSQ13, SCC135, and SCC61). However, after years of research, the ability of CHE to induce apoptosis in a variety of cancer cells, including those of the prostate, 9 liver, 10 and kidney, 11 has been favored by numerous researchers. It has also been shown that apoptosis may be associated with reactive oxygen species (ROS)-dependent endoplasmic reticulum (ER) stress.12,13 Additionally, Gao et al 14 showed that CHE can inhibit the growth of cisplatin-resistant non-small cell lung cancer (NSCLC) A549 cells, through enhanced growth inhibition and apoptosis induction, showing a synergistic antitumor effect on NSCLC A549 cells and subcutaneous graft tumors in nude mice. Furthermore, Lin et al 15 found that CHE selectively inhibits the growth of triple-negative breast cancer (TNBC) cell lines and shows a synergistic/additive effect in combination with the chemotherapeutic drug, paclitaxel. The antitumor activity exhibited by such compounds isolated from natural sources has the potential to address major problems in the field of oncology.

A photograph of celandine (Chelidonium majus).
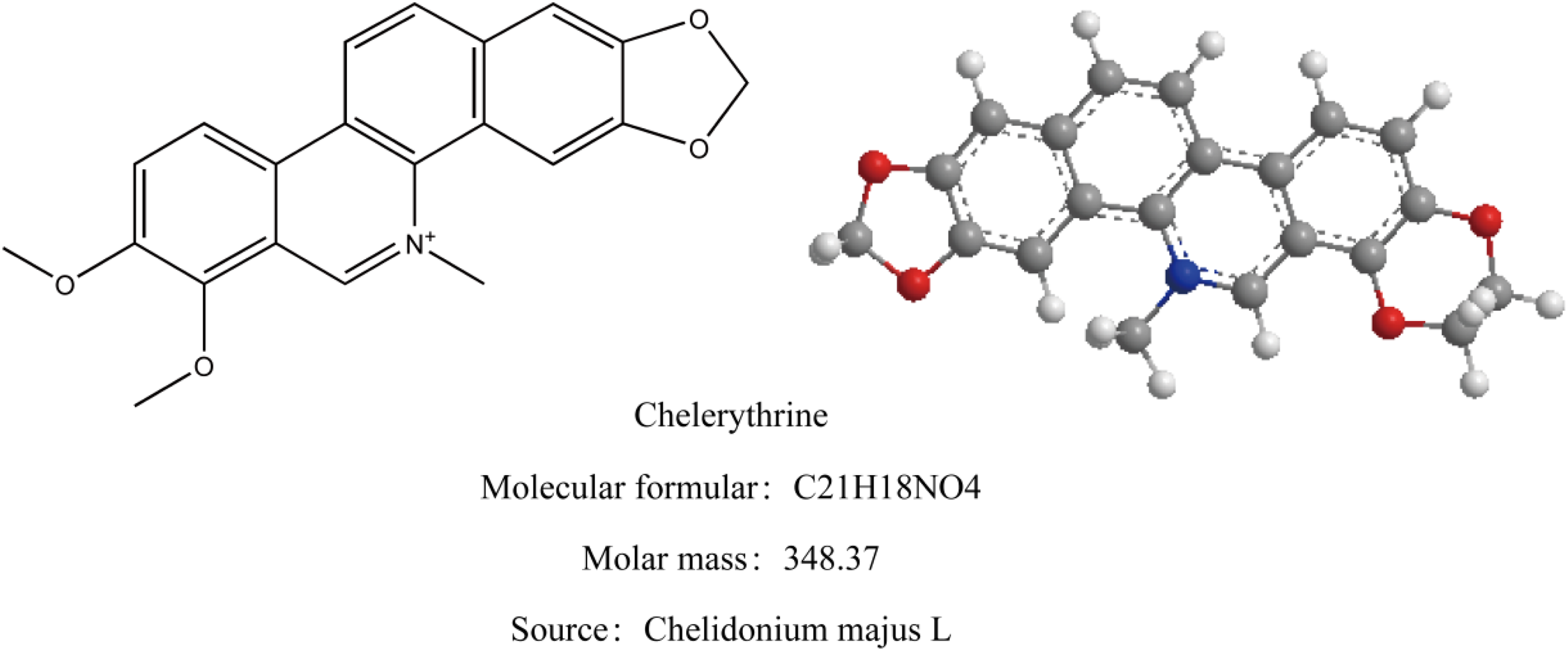
The chemical structure of chelerythrine (CHE).
The Antitumor Activity of CHE
Influence of the Signal Transduction Pathway
Influencing signal transduction pathways is an effective way to control cell proliferation and differentiation.
Yang et al
16
reported that detection of the signaling pathway activated by CHE in osteosarcoma through protein blotting revealed that CHE-induced apoptosis is mediated by rapidly accelerated fibrosarcoma (RAF)/mitogen-activated protein kinase/ERK)
Kumar et al 18 also investigated the effect of CHE on the activation of the p53-dependent/independent pathway in Dalton lymphoma (DL) cell death. DL cells were treated with CHE for 1, 3, and 6 h. It was found that treatment with CHE significantly increased the expression of total p53/phosphate-53 (ser-15) at the protein and mRNA level, which led to the activation of the p53-dependent apoptotic pathway in DL cells. Furthermore, the increased activity of cytochrome c, caspase-9, and caspase-3, as well as the degradation of DNA fragments, confirmed the activation of the p53-independent apoptotic pathway in p53-knockdown RNAi-DL cells. These findings show that apoptotic pathways that mediate signal targeting have the potential to aid in the development of better therapeutic tumor regimens (Table 1).
Induction of Apoptosis of Tumor Cells by Chelerythrine (CHE).

Abbreviations: ERK, extracellular signal-regulated kinase; PAPRP, poly (ADP-ribose) polymerase; RAF, rapidly accelerated fibrosarcoma; MAPK, mitogen-activated protein kinase.
Cell Growth Cycle Arrest
Because of the uncontrolled growth of tumor cells, cancer can be regarded as a periodic cell cycle defect. As such, both, tumor cell cycle arrest and the induction of tumor cell apoptosis can be used as effective means to control the tumor (Table 2).
Chelerythrine (CHE) can Block Tumor Cells at Different Times.

Zong et al 19 found that the rate of apoptosis in BGC823 gastric cancer cells induced by CHE was positively correlated with the No. of cells in the S and G2/M phase. However, in human leukemia HL-60 cells, Vrba et al. 20 found that CHE with a half-maximal inhibitory concentration (IC50) of 2.6 µM stalled the cell cycle at the G1 phase and induced apoptosis (by stimulating caspase-8 activity) and necrosis. Furthermore, Deljanin et al 21 intervened with a whole-plant-dried ethanol extract of white cabbage in human lung cancer (A549, H460), colon cancer (HCT116, SW480), and breast cancer (MCF-7, MDA-MB231) cell lines, as well as peripheral blood monocytes from healthy individuals. The extract was found to be selective and differentially toxic to other cancer cells at a concentration of 16 to 500 µg/mL. Additionally, IC50 at intervals ranging from 44 to 143 µg/mL demonstrated synergistic activity with doxorubicin at lower concentrations (1-2 µg/mL), antagonism at higher doses, induced apoptosis of cancer cells (microscopy), and arrest in the G2/M phase. Moreover, Yang et al 22 established a series of new CHE analogs and evaluated NSCLC cell lines, NCI-H1299, NCI-H292, and NCI-H460. Specifically, they compared the cytotoxicity of these new analogs in NSCLC and non-tumor cell lines (including human umbilical vein endothelial cells and LL24 human lung fibroblasts). The results showed that analogue compound 3f was significantly dose-dependent in A549 and NCI-H1299 cells and stalled the G0/G1 cell cycle. In this study, the molecular arrangement and electronic features of 3f were closely related to the structure of CHE and supported its activity.
Induction of the Apoptosis Pathway
The identification of small-molecule inhibitors of anti-apoptotic Bcl-2 family members plays a pivotal role in the mitochondrial regulation of cellular apoptotic pathways, providing new therapeutic opportunities.
Chan et al
23
screened 107,423 extracts from natural products by high-throughput
Oxidative Stress Induces Autophagy
The production of ROS is closely related to apoptosis. ROS were once considered to be a trivial metabolic byproduct that could be harmful to cells if not cleared or neutralized in time. However, recent studies have shown that ROS not only regulates cell proliferation, 25 but a moderate production of ROS also helps cells maintain normal function, while excess production may cause lethal oxidative stress. Cell-intrinsic antioxidant mechanisms generally balance the disruptive effects caused by excess ROS. 26 Moreover, extensive experimental data show that CHE induces the death of NSCLC, prostate, and renal cancer cells by regulating ROS generation, and is reversible by pretreatment through the ROS scavenger, N-acetyl-1-cysteine.12,13,27
Typically, apoptosis, autophagic cell death, and necrosis are caused by small, moderate, and large amounts of ROS, respectively. 25 Wu et al 12 suggested that CHE could increase ROS accumulation in prostate cancer cells and lead to ROS-dependent ER stress and apoptosis and that pretreatment with N-acetylcysteine completely reversed CHE-induced apoptosis, as well as ER stress activation. CHE induces unique apoptosis of ROS-dependent ER stress in prostate cancer cells. Therefore, the discovery of the activated ROS-mediated ER stress apoptosis pathway by CHE provides the framework for new methods of cancer treatment. However, most studies regarding the anti-cancer effects of CHE have focused on its ability to induce apoptosis, a type I method of programmed cell death. 28 However, whether autophagy, as a type II method of programmed cell death, 29 contributes to the anti-cancer role of CHE has also attracted attention from researchers.
Autophagy is a slowly evolving system that induces the degradation of cytoplasmic inclusions in a lysosome-dependent manner. 30 Tang et al 27 first studied the role and mechanism of autophagy in NSCLC cells, and found that CHE reduced cell viability, inhibited colony formation, and induced apoptosis in a concentration-dependent manner. CHE also triggered the expression of microtubule-associated protein in A549 and NCI-H1299 cells. In combination treatment with chloroquine (an autophagy inhibitor), CHE further increased LC3-II expression, and a large No. of red dots stably expressing mRFP-EGFP-LC3 were observed in CHE-treated A549 cells, indicating that CHE induced autophagic flux. Moreover, the silencing of beclin 1 reversed the CHE-induced expression of LC3-II. Additionally, CHE triggered the production of ROS in both cell lines. The reduction of ROS levels through pretreatment with acetylcysteine reversed the CHE-induced decrease in cell viability, apoptosis, and autophagy. This inconsistency in autophagic effects may be due to the varying cancer cell lines. The antioxidant capacity of A549 cells is higher than that of NCI-H1299 cells, 31 and NCI-H1299 cells are more prone to producing excess ROS, which leads to autophagic cell death. 32 Alternatively, different products of autophagic degradation may contribute to the unique role of autophagy. 33 The role of ROS-induced autophagy in cancer treatment may be largely dependent on the source of ROS production.
Inhibition of PKC Interaction
PKC is closely related to tumor development, and its overexpression has been found in many tumor cells, indicating that CHE as a PKC inhibitor can be reasonably manipulated to be an antitumor drug. PKC, which was first identified in the mouse brain in 1977, 34 belongs to the serine/threonine kinase family and is widely distributed in eukaryotic cells as a key molecule for signal transduction. CHE is considered to be a potent PKC inhibitor (Table 3).
Potential Protein Kinase C (PKC) Targeting Effects of Chelerythrine (CHE).

Abbreviations: PKC, protein kinase C; TNBC, triple-negative breast cancer.
Herbert et al 35 found that the antitumor activity of CHE measured in vitro may be attributed to the inhibition of PKC, half of which occurs at 0.66 µM/L, by interacting with the catalytic domain of PKC. In contrast to tyrosine, cyclic adenosine phosphate-dependent, and calcium/calmodulin-dependent protein kinases, CHE can selectively inhibit PKC. TNBC is a subtype of breast cancer that is currently lacking targeted therapy. Lin et al 15 showed that PKN2, a PKC isoform, was also highly expressed in TNBC cell lines, and knockdown of PKN2 in TNBC cells inhibited colony formation and growth of xenografts. These findings suggest that PKN2 is required for the survival of TNBC cells and is most likely a targeted marker that can be selectively inhibited by CHE. Although CHE is used as a PKC inhibitor, its inhibitory effect is controversial. Saavedra et al 36 stated in their most recent study that possible non-targeted effects could be observed between CHE and cultured rodent cortical neurons and neuronal cell lines. Phosphorylation of PKC and cyclic adenosine phosphate-dependent protein kinase substrates in cultured mouse cortical neurons was reduced by 10 µM CHE commonly used in neuronal cultures, but not in rat primary cortical neurons or striatal cell lines. Notably, they also found that CHE induced a concentration-dependent increase in intracellular calcium (2 + ) levels to mediate calpain, CHE-induced ERK1/2, and calpain-independent caspase-3 activation, which could be terminated by the calcium chelator, BAPTA-AM. Additionally, CHE promoted calpain activation by cleaving spectrin, striatal-rich protein tyrosine phosphatase, and calcineurin A. 36 Similar to normal synaptic mechanisms, increased calcium concentrations can promote metastatic growth of breast cancer cells, according to previous studies that documented the ability of cells to couple to astrocytes through gap junctions, reprogramming them to create a tumor microenvironment that supports their growth. 37 Zeng et al 38 reported a significant interaction between brain metastatic cells and neurons and demonstrated that activates the GluN2B-NMDAR signaling axis in the glutamate-stimulated brain tumor microenvironment, while breast cancer cells functionally produce interactions with neurons and astrocytes. Moreover, Venkataramani et al 39 suggested that neuronal activity can promote the deterioration of glioma cells through non-synaptic paracrine and autocrine mechanisms. It can be argued that CHE, as a specific and widely used PKC inhibitor for signal transduction, should be excluded as a PKC inhibitor in future studies when it is shown not to have an effect on PKC in non-neuronal cells.
Targeted G-Tetrameric Chimerism
The 6 hallmarks of malignancy include cells that are anti-apoptotic, insensitive to anti-growth signals, have self-sufficient growth signals, sustained angiogenesis, unlimited replication potential, and tissue infiltration/metastasis. 40 These 6 markers are closely related to the gene promoters forming the G-tetraplexes, including c-MYC, c-KIT, and KRAS (self-sufficiency); RB1 (insensitive); Bcl-2 (anti-apoptotic); VEGFA (angiogenesis); hTERT (unlimited replication); and PDGFA (transfer).41–46 These oncogene promoters form extremely diverse folded forms of G-tetramers.
Verma et al 47 proposed that G-tetramers can affect expression at the gene promoter and can be directly regulated at the transcription level. 48 Jana et al, 49 employed CHE after the treatment of MCF7 breast cancer cells, and confirmed through various spectroscopic techniques (ultraviolet absorption spectroscopy, fluorescence anisotropy, circular dichroism spectroscopy, and CD melting) and molecular dynamic simulations that effectively monitored the combination of CHE and G-tetramers indexes, that the formation of CHE-tetramer complexes is thermodynamically feasible. To confirm the ability of CHE to bind to DNA, the binding constant of CHE to CT-DNA (dG-dC) · Poly · Poly (dG-dC), Poly (dA-dT), Poly (dA-dT), and 7 other double-stranded oligonucleotides was determined using a pH of 5.3, indicating that CHE showed a highly specific binding ability for DNA containing continuous GC base pairs [5′-TGGGGA-3′/3′-ACCCCT-5′]. 50 Using UV visible spectroscopy, fluorescence spectroscopy, and circular dichroism spectroscopy,. Shu et al 51 found that CHE can bind to human telomeric DNA and induce it to form G-4 strands, thus preventing telomerase activity and inhibiting lung cancer A549, colon cancer HCT-8, and liver cancer Bel-7402 cell proliferation. This indicated that CHE is a telomeric, G-tetrastrand stabilizer and an effective telomerase inhibitor. The G-tetraplex-binding nature of CHE plays a crucial role in studying the antitumor activity of this compound and provides new insights into the future development of targeted cancer therapies (Table 4).
The High Binding of Chelerythrine (CHE) to DNA in Cancer Cells.

Specific Targeted Effects in Drug Combinations
As a traditional Chinese medicine (TCM) monomeric compound, CHE has nonspecific cellular targets and multi-target properties. Additionally, plant-derived chemotherapeutic agents are toxic to some extent. To improve cancer targeting with phytochemical compounds, targeting must be specific yet flexible (Table 5).
Synergistic Effect of Chelerythrine (CHE) in Drug Combination Therapy in Cancer Cells.

Abbreviations: ERK, extracellular signal-regulated kinase; MAPK, mitogen-activated protein kinase; TNBC, triple-negative breast cancer
Concentration-Dependent Improvement of the Targeted Pathways
The cell viability assay by Heng et al 52 confirmed that the use of the correct concentration of CHE hydrochloride could improve the targeting pathway through its effects on cytotoxic and antiproliferative activity in NSCLC cell lines. Immunofluorescence staining of β-catenin showed that the nuclear content of β-catenin as well as the overall cellular content were decreased after treatment of CHE hydrochloride with NCI-H1703. Moreover, the favorable inhibitory activity of CHE hydrochloride on cancer stem cells (CSCs) was observed, including soft agar colony formation, migration, invasion, and spheroid formation. CHE hydrochloride treatment exhibits certain concentration-dependent targeting behaviors with regulated β-catenin expression, cell death, and apoptosis. Moreover, downregulation of catenin implies downregulation of CSC transcription factors (SOX2 and MYC) and usually induces necrosis at high concentrations and apoptosis at lower concentrations. In conclusion, CHE has the potential to mitigate cancer growth due to its inhibitory effect on CSC oncogenic activity in lung cancer and can be flexibly regulated according to concentration to modulate specific targeting in various cell lines.
Combinations Produce Synergistic Effects
A recent study suggests that synergistic effects may be achieved through targeted coverage pathways in a combination treatment regimen consisting of phytochemical compounds and chemical therapies. 54 Phytochemical compounds also have the potential to mitigate chemical therapy-induced toxicity, which further justifies their use with conventional therapies. 55 For example, combination treatment of CHE hydrochloride, erlotinib, and cisplatin had synergistic or additive effects against NSCLC.14,53 Paclitaxel chemotherapy acts by enhancing the polymerization and stability of microtubules, disrupting the progression of the normal cell cycle during mitosis. 56 At present, no targeted therapy exists for patients with TNBC, and chemotherapy is still the standard treatment regimen. Lin et al 15 investigated the effect of CHE combined with paclitaxel on the proliferation of TNBC cells. They evaluated 4 TNBC (MDA-MB-231, BT-549, HCC1937, and MDA-MB-468) and 4 non-TNBC cell lines (MCF7, ZR75-1, SK-BR-3, and MDA-MB-453). All 4 TNBC cell lines experienced reduced proliferation in a time-dependent manner to a greater degree than the non-TNBC cell lines. Therefore, TNBC cell lines were more sensitive and dose- and time-dependent to this treatment than non-TNBC cell lines. These findings showed that CHE exhibits selective, antiproliferative activity against TNBC cells, and the dual treatment of CHE and paclitaxel has additive or synergistic effects. Therefore, combination therapy with CHE and chemotherapeutic agents is a significant treatment plan for tumors.
Conclusion
CHE is an active ingredient extracted from traditional Chinese herbal medicine and has the advantages of low toxicity, high utility, many targets, and strong specificity. From a safety perspective, naturally derived phytochemical compounds are superior to the immune response of patients with cancer. Several TCM monomers and natural compounds with similar properties to CHE have been present in other official treatment regimens used in different countries. However, in recent years, CHE research in China has lacked momentum. Moreover, most experiments related to natural compounds and TCM monomers with chemotherapeutic drugs harbor many limitations, such as the lack of stochastic methods, non-standardized methods of tumor size measurement, subjective evaluation, and the lack of an appropriate sample size and statistical methods. Traditional and modern medicine from various ethnic backgrounds should be brought together to tap into the full potential of the field of antitumor medicine.
The following aspects should be further investigated: First, pharmacological techniques should be employed to utilize the targeted specificity of CHE and determine the forms of preparation and individualized dosage. Second, in addition to in vitro cells, in vivo models should be utilized to explore the specific antitumor mechanism of CHE. Moreover, computer models should be developed to optimize the exploration of CHE in tumor cells and reduce the cost of this modality. Third, due to the various ways in which different cancer cells produce ROS, the mechanisms of various pathways are understood only at the level of conjecture and have undergone preliminary verification. Therefore, CHE-induced ROS production in various types of cancer cells warrants further research. Fourth, CHE can effectively inhibit the G-tetrastrand structure, combining human telomeric DNA and RNA G-tetramers. Thus, the complete signaling cascade and fine regulatory sites of the tetrastrand structure formed in the promoter regions of oncogenes such as Bcl-2, VEGFA, and KRAS, as well as their expression, should be explored. Fifth, artificial intelligence is the leading method of accurate detection, characterization, and monitoring, especially with the development of fine dynamic tumor reconstruction. Therefore, future research should be multidisciplinary and include artificial intelligence to deduce tumor genotype and biological process, predict clinical results, and evaluate CHE targets to understand the impact on adjacent organs. Sixth, studies show that the aberrant expression of the PIWI-interacting RNAs (piRNA) signaling pathway regulates the expression of DNA methyltransferase, and the highly promising mechanism of piRNA regulation using CHE will be a breakthrough in the field of oncology. Seventh, currently based on network pharmacology of Chinese medicine monomer, drug, compound, are in the lateral verification of its multiple target effect, such research lack of deep exploration of the circuit link, so makes the research appears two large ends, but small middle, so based on network pharmacology reverse molecular docking and computer modeling, under the same structure relationship, and from the existing natural plant compounds screening drugs, should be a new idea. Finally, epigenetics is the main controlling factor of oncogenes in the development of cancer. Therefore, applying the epigenetic properties of CHE to treat malignant tumors demonstrates great potential in the field of epigenetic research.
The discovery of natural resources promotes research on plant-derived compounds and their TCM monomers. Research and clinical transformations focused on the applications of CHE will provide new strategies for the precise diagnosis and treatment of cancer. Therefore, in this era of rapid technological development, various disciplines and TCM can complement each other, and together a multidisciplinary approach toward an effective treatment for cancer can be made.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Youth Traditional Chinese Medicine Science and Technology Innovation Plan of Heilongjiang Provincial Traditional Chinese Medicine Association (grant number ZHY19081).
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
