Abstract
Biocatalytic asymmetric reductions of acyclic and cyclic α,β-unsaturated carbonyl compounds are favorable protocols for introduction of chiral centers to α- and/or β-positions of the carbonyl groups. Representative biocatalytic reductions of electron deficient olefins are compiled from a synthetic point of view according to compound types from the papers in 2012 to early 2022. Applications to syntheses of some enantiomericaly enriched perfumery ingredients are presented to show the feasibility of the biocatalytic reductions.
Keywords
Introduction of chiral centers to organic molecules is an important task, especially for total syntheses of physiologically active compounds. Carbonyl groups have been utilized to introduce chiral centers at the α- or β-position by asymmetric nucleophilic reactions of enolates, 1 asymmetric protonation2,3 or asymmetric conjugate additions to α,β-unsaturated carbonyl compounds (Figure 1).4,5 Catalytic asymmetric hydrogenation of a double bond employing chiral organometallic catalysts has an alternative advantage, especially in the reduction of α,β-unsaturated carbonyl compounds, which play a major role not only on a laboratory scale but also on a process scale to introduce up to two stereogenic centers in a single step. 6 Compared to other asymmetric inductions, the catalytic reductions do not require any extra reagents other than catalysts, which is superior in atom economy, benign and sustainable, with less waste. A simple synthetic operation is an extra advantage.
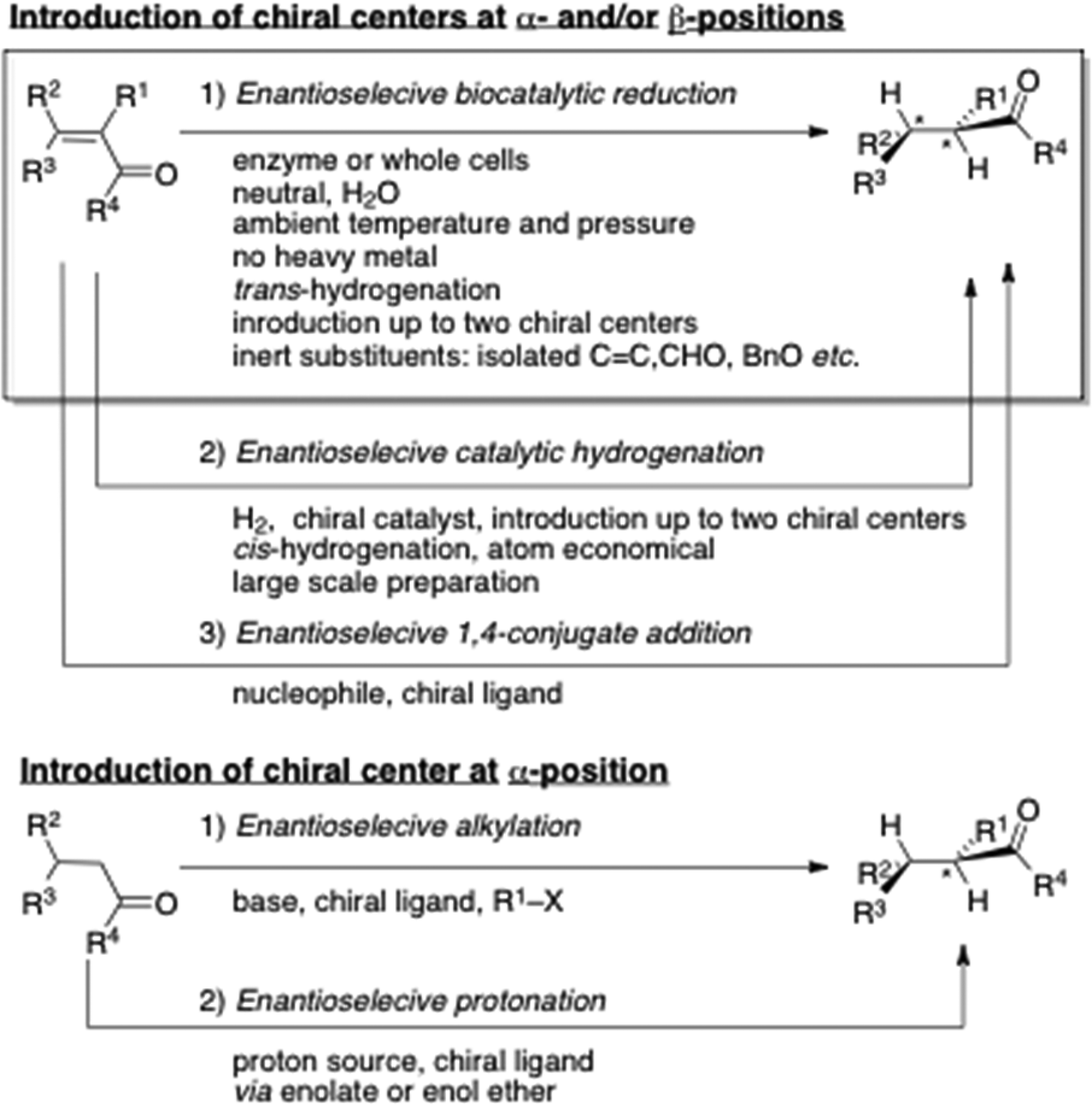
Introduction of chiral centers at α- and/or β-positions of carbonyl groups.
In addition to the catalytic asymmetric hydrogenation to introduce chiral centers at the α- and/or β-positions of carbonyl groups, biocatalytic asymmetric reductions of α,β-unsaturated carbonyl compounds have attracted more and more attention employing enzymes or whole cells, which proceed in aqueous solvents under neutral conditions at room temperature under atmospheric pressure. The reaction condition is benign and environmentally friendly. An additional advantage is that there is no contamination of heavy metals in the products and wastes, which is different from catalytic hydrogenations. Chemoselectivity of the reduction is high, because the reaction proceeds only with electron-deficient olefins. Isolated double bonds, carbonyl, formyl, nitro, and cyano groups, and benzyl ethers are left intact under the reaction conditions, which could be reduced by catalytic hydrogenations. Although selective reduction of a double bond of an α,β-unsaturated carbonyl compound in the presence of an isolated double bond could be carried out chemoselectively by electron transfer reduction with alkaline metals, the reaction is not suitable for chiral induction.
These characteristic features of biocatalytic reductions are favorable for syntheses of physiologically active compounds including not only flavor and fragrance substances, 7 but also pharmaceutical ingredients. 8
The quality of perfumery ingredients highly depends on the absolute stereochemistries, their enantiomeric purities, and on the character of very small amounts of contaminants. The benign nature of biocatalytic reduction might be suitable to apply the flavor substances to human bodies. Some representative flavor and fragrance substances having chiral centers at α- or β-positions of carbonyl groups are highlighted in Figure 2.

Some representative flavor ingredients having chiral centers at α- and/or β-positions of carbonyl groups and their related derivatives.
A great deal of research data has been accumulated on the biocatalytic asymmetric reduction of electron-deficient olefins,9–14 and during preparation of this manuscript, a comprehensive review on the topic by Hollmann, Opperman and Paul has appeared. 13 Synthetic organic chemists are familiar with reagent-based reductions, in spite of the big advantage of biocatalytic reduction. In this regard, the present review deals with developments in enantioselective biocatalytic reduction of electron deficient olefins to emphasize the synthetic utility of biocatalytic reduction, in which more specific examples of bioreductions and applications to the syntheses of some perfumery ingredients and physiologically active small molecules are listed to exemplify the further feasibility of bioreduction in organic synthesis.
Among recent progress in biocatalytic asymmetric reductions, old yellow enzymes (OYE: NADPH dehydrogenase, EC 1.6.99.1) have been studied to reduce electron-deficient olefins, such as α,β-unsaturated carbonyl compounds. A variety of OEYs are found in a variety of plants such as tomatoes and microorganisms, which are relatively stable and available on a large scale. Their structures are well established by X-ray crystallography.15,16 With the development of genetic engineering, 17 variants by genetic modification and homologues by cloning have been created to elucidate the reaction mechanism, to expand the scope of applications, and to improve the reaction conditions.
The mechanism of the reduction by OYE is considered as shown in Figure 3.9,18–20 The substrate taken up in the enzyme is reduced in trans-fashion by 1,4-addition of the hydride by the flavin residue present in the protein, which is in contrast to the cis-type addition in the catalytic reduction in the presence of metal catalyst under a hydrogen atmosphere. The oxidized flavin residue is reduced by a cofactor NADPH, which starts the reduction cycle again. The resulting NADP+ is reduced back to NADPH by a combination of glucose and glucose dehydrogenase to turn the catalytic cycle. A combination of glucose-6-phosphate and glucose-6-phosphate dehydrogenase, formate and formate dehydrogenase, 2-propanol and alcohol dehydrogenase, or phosphite and phosphite dehydrogenase can also be used in this cofactor recycling process.
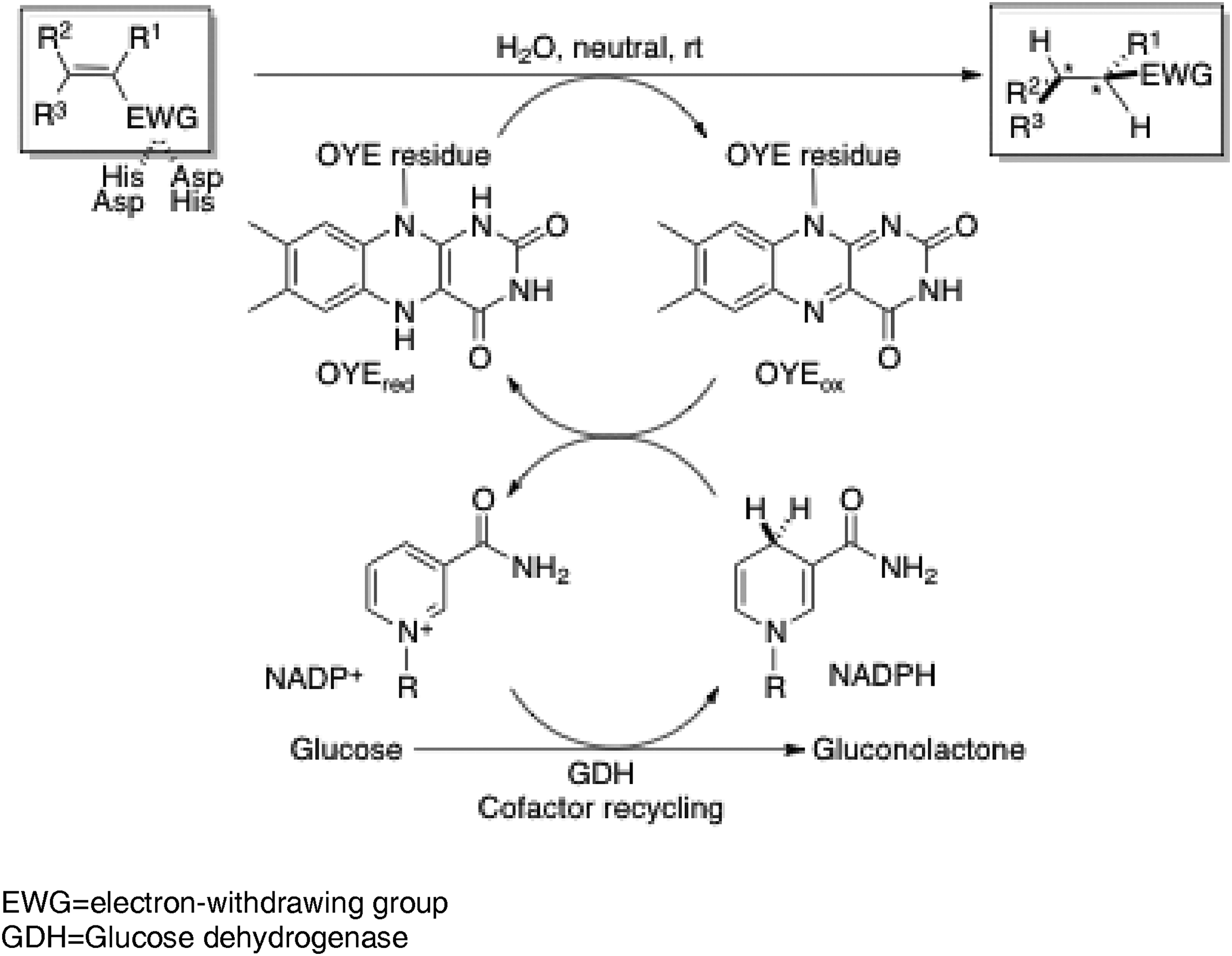
Representative OYE catalyzed reduction of electron deficient olefins by NADPH recycling with glucose. EWG = electron-withdrawing group. GDH = Glucose dehydrogenase.
Biocatalytic Reductions of Cyclic α,b-Unsaturated Ketones
Cyclic ketones having chiral centers at α- and/or β-positions of ketones are important chiral building blocks for total syntheses of natural products. Some representative results to introduce chiral centers by bioreductions at β-positions of β-substituted cyclic enones are shown in entries 1∼8, Table 1. In entry 1, whole cells of E. coli BL21(DE3)(pOYE-pET3b) were employed to reduce 3-methylcyclohexenone in 100% conversion with 96% enantioselectivity (ee) without adding NADP+ and GDH. Cyclohexenones having larger substituents also resulted in high enantioselectivity. but in lower yield. β-Cyclodextrin was added to promote solubility of the substrate in aqueous media. In entry 2, glucose was added to maintain the strain. In entry 3, OYE homologue, YqjM, enabled the reduction of cyclohexenone having a longer substituent. In entries 4 and 5, both enantiomers were obtained by changing ene reductases, and also in 6 and 7 by changing YqjM mutants. (R)-(–)-Muscone (

Synthesis of unnatural (S)-( + )-muscone (
Biocatalytic Reductions of Cyclic α,β-Unsaturated Ketones.

Ampicillin, aminobenzylpenicillin; ClER = OYE homologue from Clavispora lusitaniae; FDH, formate dehydrogenase; FMN, flavin mononucleotide; GDH, glucose dehydrogenase; G6PDH, glucose-6-phosphate dehydrogenase; IPTG, isopropylthio-β-D-galactoside; NCR, nicotinamide-dependent cyclohexenone rductase; OPR, OYE homologue from Lycopersicon esculentum (tomato); OYE 2.6, OYE homologue from Pichia stipitis; TEOA, triethanolamine; TsOYE, OYE homologue from Thermus scotoductus; XenA, Oxidized OYE from Pseudomonas putida; YqjM, OYE homologue from Bacillus subtili.
Some examples of asymmetric bioreductions of α-substituted cyclic enones to introduce chiral centers at α-positions are presented in entries 9∼19. The same reaction condition in entry 1 was applicable to the reduction of 2-methylcyclohexenone in entry 9, with the same efficiency. The same enone was reduced by 90 kDa reductase from Nicotiana tabacum plant culture in entry 10, while the opposite enantiomer was obtained in entry 12 by 44 kDa reductase from the same plant. In entry 11, the bioreduction was accomplished without addition of cofactor, NADH, in which photo-excited rose bengal reduced the oxidized OYE to turn the catalytic cycle. The oxidized rose bengal was driven back to rose bengal by triethanolamine (TEOA). The process is clean, cost effective and favorable for large scale preparation. In entry 13, the same reaction conditions to reduce 3-methylcyclohexeneone in entry 1 was applied to the reduction of 2-methylcyclohexenone. The same (R)-2-methylcyclohexanone was obtained from 2-methylenecyclohexanone in entry 14. Similarly, 2-methylcyclopentenone was reduced by ClER and OPR1 in entries 14 and 15. The Baylis − Hillman substrate was reduced by OYE 2.6 in entry 16. Among various mutants investigated, the wild type mutant exhibited the best performance. In entry 18 and 19, the bioreductions were carried out without employing the GDH recycling system, in which the reduction of entry 18 was scaled up to 400 mg of the substrate.
Bioreduction of ketoisophorone (oxophorone) (
Dihydrocarvone (
Biocatalytic Reduction of Acyclic α,b-Unsaturated Ketones
In entry 1, Table 2, bioreduction by microorganism, S. cerevisiae, was carried out by adsorbing the substrate in filter paper. On the other hand, the opposite enantiomer was obtained by the cascade reaction in entry 1, Table 2, which was applied to the total synthesis of (R)-tropinal® (

Synthesis of (R)-tropional (
Biocatalytic Reductions of Acyclic α,β-Unsaturated Ketones.

Biocatalytic Reductions of α,b-Unsaturated Aldehydes
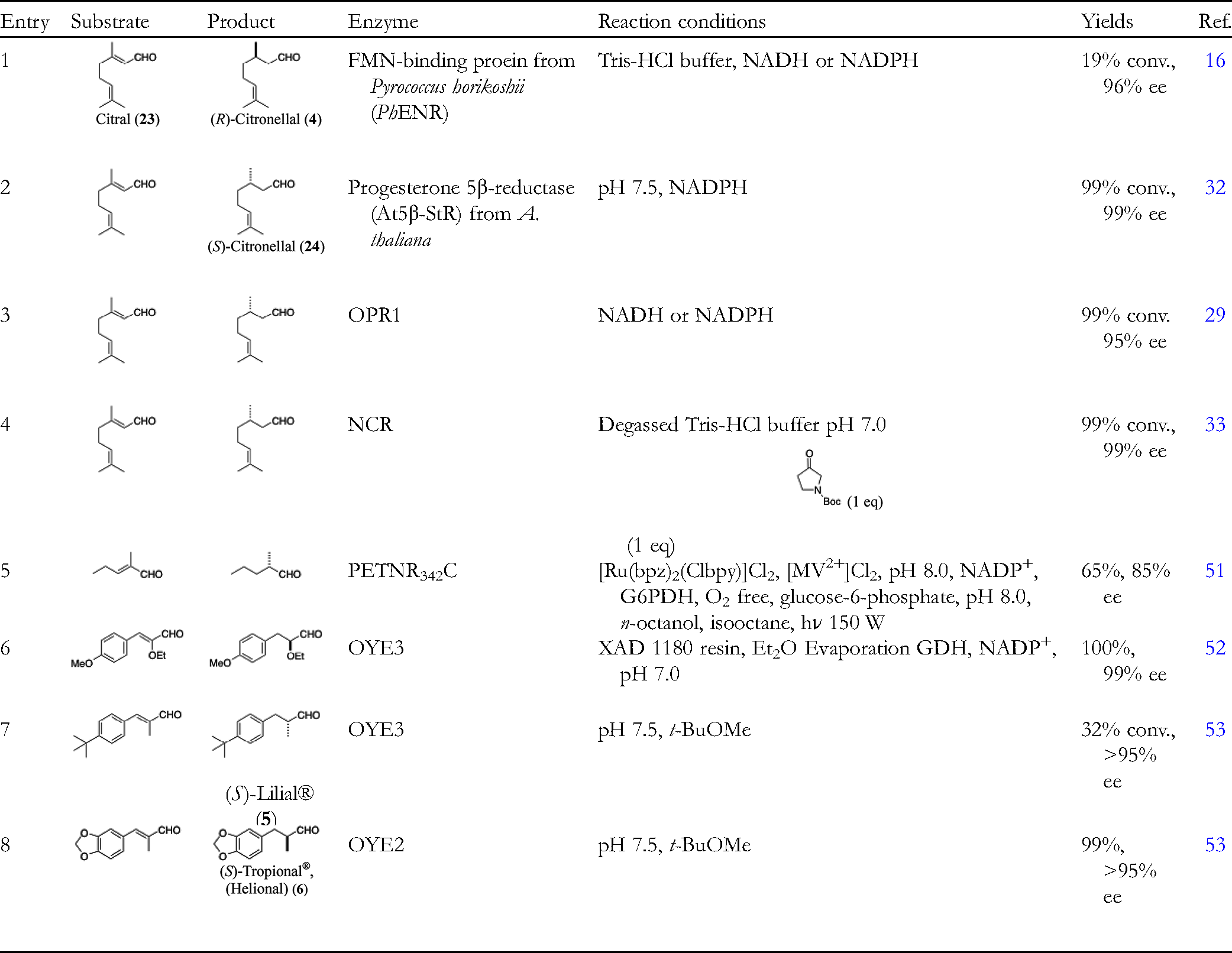
Some aldehydes of low molecular weight are versatile fragrance components due to their volatility. Introduction of chiral centers at α- or β-position of a formyl group is not well exploited because of inherent sensitive nature of a formyl group toward various chemical transformations. An asymmetric catalytic hydrogenation is not feasible due to the possibility of reduction of the formyl group into primary alcohol. Citronellal (
Biocatalytic Reductions of α,β-Unsaturated Aldehydes.
Biocatalytic Reductions of α,b-Unsaturated Acids, Esters and Lactones
Unsaturated acids and esters, especially those having extra electron- withdrawing groups (EWG), are suitable for bioreduction, in which the EWG groups make the reduction easier by both lowering the barrier and stabilizing the product so shifting the equilibrium compared to substrates with electron donating groups (Table 4).
Biocatalytic Reductions of α,β-Unsaturated Acids, Esters and Lactones.

Regio- and asymmetric reduction of (E)-β-cyanoacrylic acid is depicted in entry 1, in which the presence of γ,d-unsaturation was vital for substrate acceptance. The (Z)-double bond isomer was not reduced. Catalytic hydrogenation of the product led to the γ2-aminoester (S)-pregabalin (
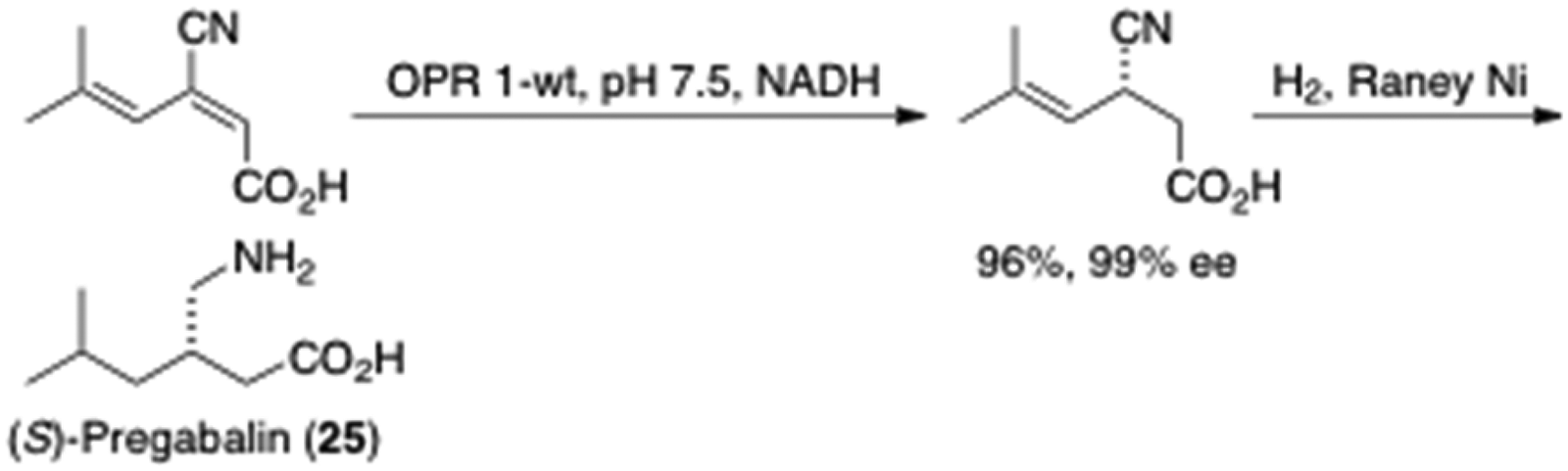
Synthesis of (S)-pregabalin (
In entry 2, by employing an oxidized OYE, XenA, the unsaturated acid, led to (R)-naproxen, an antipode of the anti-inflammatory drug, (S)-naproxen. Development to afford the pharmacologically important (S)-isomer is expected.
Electron deficient (E)-β-cycanoacrylate was reduced by OYE3 in entry 3. Bioreduction of the corresponding (Z)-isomer was less efficient. The present protocol was applied to the synthesis of a GABA analogue (

Synthesis of γ2-aminoester (
When Trp in position 116 in OYE1 was replaced by Leu, the reduction of β-cycanoacrylate provided (R)-3-cyano-3-phenylpropanoate in entry 4; replacement with Ala led to the (S)-enantiomer in entry 5. Similarly, both enantiomers were obtained from Baylis-Hillman product by two alternative YqjM variants in entries 6 and 7. In entries 8 and 9, (Z)- and (E)-2-methylsuccinate furnished by the same enzyme (R)- and (S)-enantiomers, respectively. The isolated double bond was stable under the same reaction conditions in entry 10. It is worthy of note that both (Z)- and (E)-α-bromocrotonate were reduced by OYE3 quite efficiently in entry 11 to provide (S)-2-bromobutanoate, which is difficult to prepare by other means, such as catalytic hydrogenation, owing to the labile nature of the C–Br bond toward hydrogenation, along with easy racemization. α-Chlorocrotonate was also reduced by the same enzyme to afford the (S)-enantiomer highly efficiently. In spite of the absence of an electron-withdrawing group, β-substituted butenolides were reduced in entry 12. Both enantiomers of β-methyl-δ-valerolactone were prepared in entries 13 and 14, which is superior than transition metal catalyzed asymmetric reduction. Scale-up synthesis of the (R)-enantiomer by OYE2 in entry 13 was accomplished in a 1.3 L batch reactor to result in a final concentration of 1.0 g L–1.
Cascade Reductive Biotransformations
Synthesis by multiple biotransformations in a one pot operation is very favorable in terms of pot economy. 62 Several efforts have been reported by either combining two different enzymes, or a chemical reaction and enzyme.63–65
Entry 1 in Table 5 shows that reduction of an acetoxy-enone provided (R)-α-methylketone in 63% and 96% ee in a 5 mmol scale. The reduction was carried out by adsorption of the substrate on filter paper. The product was applied to the synthesis of (R)-tropinal® (
Cascade Reactions Involving Biocatalytic Reduction.

BsGDH, glucose dehydrogenase from Bacillus subtilis; GluER, OYE from Gluconobacter oxydans; MNMR, menthone:( + )-neomenthol reductase from Mentha piperita; MMR, ( − )-menthone:( − )-menthol reductase from Mentha piperita; NtDBR, ene reductase from Nicotiana tabacum; PLP, pyridoxal-5’-phosphate.
In entries 2 and 3, after confirming the reduction of enone by the ene reductase, alcohol dehydrogenase was added sequentially in one pot to reduce the carbonyl group diastereoselectively, in which alcohols having two chiral centers were obtained with high enantioselectivity. OYE3 and alcohol dehydrogenase (ADH) EVO030 were added in one portion at the same time in entry 4 to provide syn-bromohydrin efficiently. However, when the two enzymes were added sequentially, the diastereoselectivity was lowered due to isomerization of the intermediary α-bromoketone. The resulting syn-bromohydrin was transformed to the tetrahydrofuran (

Synthesis of the most pleasant roasted meat flavor (
(4R,6R)-Actinol (

Synthesis of florhydral® (
Transamination was combined with enone reduction in entries 10∼12. Two diastereomers were obtained independently by employing (S)-ω- or (R)-ω-transaminase in entries 10 and 11. Sequential addition of two enzymes was favorable for good conversion in entries 10 and 11, whereas mixing the two enzymes at one time was also effective in entry 12. In entry 13, a one-pot bienzymatic cascade from inexpensive geraniol (
Conclusions
The introduction of chiral centers into the α− and/or β−positions of carbonyl compounds is an important task in organic synthesis, and a variety of methods are known, in which asymmetric alkylation or protonation at the α-position of the carbonyl group, catalytic hydrogenation, and conjugate addition to the β-position of an α,β-unsaturated carbonyl compound has been well investigated. Among them, the reduction of α,β-unsaturated carbonyl compounds using asymmetric organometallic catalysts has been widely studied and achieved in recent years, because the reaction proceeds cleanly and atom economically, does not require extra reagents, and produces less waste, and the operation is simple compared with other methods, which is a promising protocol for process scale asymmetric synthesis. In addition to the introduction of chiral centers by catalytic asymmetric hydrogenation, the biocatalytic asymmetric reduction of α,β-unsaturated carbonyl compounds is an alternative method that should be focused on.
In this review, the asymmetric biocatalytic reduction of α,β-unsaturated carbonyl compounds is summarized according to compound type from the viewpoint of synthetic organic chemistry. The progress in biocatalytic asymmetric reduction has been remarkable in recent years, and a great deal of research data have been accumulated. In particular, much effort has been devoted to the asymmetric reduction of electron-deficient olefins employing OYEs, and many remarkable studies have been reported, including the development of reaction conditions and the preparation of OYE variants by genetic engineering. Compared to the asymmetric catalytic hydrogenation with a chiral organometallic catalyst, only the electron-deficient olefins are reduced chemoselectively, in which isolated double bonds, formyl groups, ketones, benzyl ethers, etc remained intact under the reaction conditions. The reaction proceeds at ambient temperature under atmospheric pressure in aqueous solvent. There is no contamination of heavy metal in either the product or waste. These characteristic benign features are suitable for preparation of chiral building blocks as a starting material for natural products, syntheses of chiral fragrance compounds of low molecular weight, and for transformations of compounds with multifunctional groups, especially in the final stage of syntheses of pharmaceuticals. At the same time, various variants of OYEs have been prepared by genetic engineering and used either as purified enzymes or in whole cells, which led to the expansion of substrate scope and improvement of the reaction conditions. Development in the reduction using OYEs has made improvements in co-factor regeneration in various ways.
In spite of the abundance of research work on the biocatalytic reduction of α,β-unsaturated carbonyl compounds, there still remains some issues to be considered. Owing to the higher substrate specificity of the enzymes, diversity of substrates is limited. Only one enantiomer can be obtained by a specific enzyme, and only one enantiomer can be accepted by an enzyme. Substrates of higher molecular weight having more complex structures are difficult to be reduced. Asymmetric biocatalytic reduction of a tetra-substituted olefin is rare introducing two chiral centers at α− and β−positions of a carbonyl group at the same time. 77 In a process scale reaction, recycling of the catalyst system and improvement of the turnover number are desired. In order to stabilize unstable enzymes and make them reusable, immobilization on solid or liquid support is suggested, which might be useful also for flow synthesis. Even though the reduction by enzymes is useful in organic synthesis, synthetic organic chemists are not used to isolate or bioengineer enzymes on a preparative scale. Utility of these enzymes like stock reagents is expected. How to introduce efficiently chiral centers into the α- and/or β-positions of carbonyl compounds is a long-lasting topic in synthetic organic chemistry. It is a desired challenge to improve these issues. Since biocatalytic reactions are basically sustainable and atom economical, their potential is high. Further progress in this area is expected.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
