Abstract
A new sterol, ganolutol A (
Introduction
Ganoderma luteomarginatum is a rare polypore fungus belonging to the family Ganodermataceae. It is mainly distributed throughout tropical and subtropical areas including China, Laos, Myanmar, Thailand, and Vietnam.1,2 The G luteomarginatum fruiting bodies have been utilized in traditional medicine to treat and prevent various diseases such as hypertension, diabetes, hepatitis, and cancers in China, Japan, and Korea.3,4 The fruiting bodies are known as “Lin zhi” in Myanmar and their extracts have long been used locally for the purposes of liver protection, blood purification, and detoxification, as well as tumor treatment. 5 A few previous phytochemical investigations of ethanol extracts of G luteomarginatum fruiting bodies collected in China led to the isolation of 12 lanostane-type triterpene acids, a pair of alkaloid enantiomers, and 9 lanostane triterpenoids.1,6,7 Among them, lanostane triterpenoids such as (17Z)-3β,7β,15β-trihydroxy-11,23-dioxolanost-8,17(20)-dien-26-oate, (20E)-15β-hydroxy-3,7,11,23-tetraoxolanost-20(22)-en-26-oate, and (5α,24E)-3β-acetoxyl-26-hydroxylanosta-8,24-dien-7-one exhibited strong cytotoxicities against gastric HGC-27, cervical HeLa, and lung A549 human cancer cell lines.1,6 In the course of our search for cytotoxic compounds from traditional folk medicines in Myanmar, phytochemical investigations of the ethyl acetate-soluble fraction of a 70% ethanolic extract of the Myanmar G luteomarginatum fruiting bodies revealed the presence of one new ergostane-type steroid and four known compounds. Herein, we report the isolation and structure elucidation of the compounds and their cytotoxicities.
Results and Discussion
Various chromatographic procedures on the ethyl acetate-soluble fraction of the 70% ethanolic extract of G luteomarginatum led to the isolation of 5 compounds, including 1 new ergostane-type steroid, named ganolutol A (

Structures of isolated compounds
Compound
1H (400 MHz) and 13C (100 MHz) NMR Data of

The above NMR data closely resembled those of the ergostane-type steroid isolated from Panellus serotinus, 5α,9α-epidioxy-(22E)-ergosta-7,22-diene-3β,6β-diol (

Key HMBC (arrows) and 1H-1H COSY (bold lines) correlations of
The relative configuration of

Key NOESY correlations (dashed arrows) of
Primary CD calculation analyses of the (3S,5R,6R,9R,10R,13R,14R,17R)-

Calculated and experimental ECD spectra of
The ethyl acetate-soluble fraction and the isolated compounds
Cytotoxic Activities of Compounds

Note: aμg/mL.
μM.
Ethyl acetate-soluble fraction.
Positive control.
Conclusions
A new sterol, ganolutol A (
Experimental Section
General Experimental Procedures
NMR spectra were recorded on a JEOL ECA400II spectrometer. Pyridine-d5 and CDCl3 were used to solve all isolated compounds. δH 8.74 and δC 150.35 for pyridine-d5 and δH 7.26 and δC 77.0 for CDCl3 of the residual solvent peaks were used to calibrate the signals. HR-ESI-MS data were obtained with a Shimadzu LCMS-IT-TOF spectrometer. Optical rotation and CD measurements were performed on a JASCO P2100 polarimeter and a JASCO J-805 spectropolarimeter, respectively. UV and IR spectra were measured on an Implen NP80 nanospectrometer and a JASCO FT/IR-460 Plus spectrometer (KBr pellets), respectively. Silica gel 60N (spherical, neutral, 40-50 μm, Kanto Chemical) and Cosmosil 75C18-OPN (Nacalai Tesque) were used to perform normal phase and reverse phase open column chromatography, respectively. Thin-layer chromatography (TLC) was performed on silica gel GF254 pre-coated plates (Merck). The compounds were detected under a UV lamp (254 and 365 nm) and by spraying with a p-anisaldehyde stain solution followed by heating at 170 °C for 10 min in a drying cabinet.
Fungal Material
The fruiting bodies of G luteomarginatum were purchased from the local medicinal market in Aungban, Shan State of Myanmar and were identified by Dr Ni Lar Cho, a botanist at the Department of Botany, Meiktila University. A voucher specimen (31185) has been deposited in the Museum for Materia Medica, Analytical Research Center for Ethnomedicines, Institute of Natural Medicine, University of Toyama, Japan.
Extraction and Isolation
The fruiting bodies of G luteomarginatum (1.49 kg) were chipped and extracted with 70% ethanol under sonication (12 L, 3 h, × 5) at 30 °C after drying. The solvent was then evaporated, and the resultant 70% ethanolic extract (45.9 g) was suspended in water and partitioned with ethyl acetate to give an ethyl acetate-soluble fraction (31.5 g). The ethyl acetate-soluble fraction was chromatographed on a silica gel column eluted with n-hexane − ethyl acetate (90:10 to 0:100) and ethyl acetate − methanol (90:10 to 0:100) to give eleven fractions (F1−11). Fraction F4 (2.8 g) was rechromatographed on a silica gel column eluted with n-hexane − dichloromethane − ethyl acetate (40:40:20 to 0:50:50) to afford 4 subfractions, F4−1 (180 mg), F4−2 (166 mg), F4−3 (54 mg), and F4−4 (2.4 g). Subfraction F4−2 (166 mg) was subjected to a silica gel column eluted with n-hexane − ethyl acetate (80:20 to 0:100) to afford
Ganolutol A (
ECD Calculations
The conformational search was conducted as previously described with some modifications. 13 The Avogadro 1.2 program was used to obtain the MMFF94 molecular force field. The optimization of all possible conformers at the B3LYP level of theory was performed by using the 6-31G(d) basis set, and the minimum energy of the structure was ensured by calculating their optimized geometries. The geometries used for the ECD calculations were obtained from Gaussian 16 at the B3LYP/6-31G(d) level. GaussSum using a half bandwidth of 0.2 eV was used to generate the ECD curves. All spectra of the lowest energy conformations were averages.
Cell Culture and Cytotoxicity Assay
The 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay
14
was used to evaluate the cytotoxicities of the isolated compounds in cell lines. The human cancer cell lines (A549, MCF-7, and HeLa) were cultured in α-Minimum essential medium with phenol red and L-glutamine (α-MEM, Wako).15–20 A 1% antibiotic antimycotic solution (Sigma) and 10% fetal bovine serum (FBS; Sigma) were supplemented to all media. For the MCF-7 cells, 1% 1 mM sodium pyruvate (Gibco) and 1% 0.1 mM nonessential amino acids (NEAA, Gibco) were supplemented in the growth medium. Briefly, each cancer cell line was seeded at 2 × 103 cells per well in 96-well plates and incubated for 24 h in the respective medium at 37 °C, under a 5% CO2 and 95% air atmosphere. The cells were then washed with phosphate-buffered saline (PBS), and the tested samples were added to each well at 6 concentrations (6.25, 12.5, 25.0, 50.0, 100, and 200 μM). After a 72 h incubation, the cells were washed with PBS, and to each well, 100 μL of medium containing 10 µL of the MTT solution (5 mg/mL) was added and incubated for 3 h. Subsequently, each well was monitored to calculate cell viability at 570 nm, using the following equation (each set of cell viability at each concentration was the mean value of the data from 3 wells):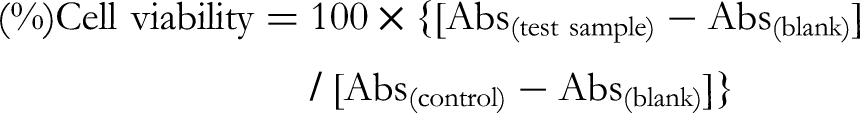
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221098852 - Supplemental material for A New Sterol From the Polypore Fungus Ganoderma luteomarginatum and Its Cytotoxic Activities
Supplemental material, sj-docx-1-npx-10.1177_1934578X221098852 for A New Sterol From the Polypore Fungus Ganoderma luteomarginatum and Its Cytotoxic Activities by Zin Paing Htoo, Takeshi Kodama, Nwet Nwet Win, Naotaka Ikumi, Ken-ichi Shiokawa and Hiroyuki Morita in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
