Abstract
In the present study, the chemical composition of the essential oil from aerial parts of Parentucellia latifolia (L.) Caruel collected in Central Sicily was analyzed by gas chromatography and gas chromatography–mass spectrometry. The results showed the presence of sesquiterpene hydrocarbons, with germacrene D and germacrene B accounting, respectively, for 59.2% and 24.3% of the total oil. Different colonies of bacteria and fungi frequently affect cellulosic objects such as books stored in libraries and museums. The antibacterial and antifungal activity against some microorganisms infesting historical-artistic craftsmanship was determined, demonstrating that the essential oil was particularly active against Bacillus subtilis, Staphylococcus aureus, and Proteus vulgaris.
Keywords
Introduction
The genus Parentucellia (family Orobanchaceae) comprises annual herbs with generally opposite and sessile upper leaves and flowers spike-like or arranged in racemes. 1 Four species are distributed in Western Europe, the Mediterranean, Central and Southwestern Asia: Parentucellia latifolia (L.) Caruel [syn. Bartsia latifolia (L.) Sibth. & Sm., Euphrasia latifolia L., Eufragia latifolia (L.) Griseb., Lasiopera latifolia (L.) Samp., Trixago latifolia (L.) Rchb.], Parentucellia latifolia subsp. flaviflora (Boiss.) Hand.-Mazz. [syn. Eufragia latifolia var. flaviflora Boiss, Parentucellia flaviflora (Boiss.) Nevski, Bellardia latifolia subsp. flaviflora (Boiss.) Raus], Parentucellia viscosa (L.) Caruel [syn. Bartsia viscosa L., Bellardia viscosa (L.) Fisch. & C. A. Mey., Eufragia viscosa (L.) Benth., Euphrasia viscosa (L.) Benth., Lasiopera viscosa (L.) Hoffmanns. & Link, Trixago viscosa (L.) Rchb.] and Parentucellia floribunda Viv. 2
According to Scheunert et al, 3 together with Bellardia trixago (L.) All., Bartsia mutica (Kunth) Benth., and B. canescens Wedd., these species belong to the clade Bellardia of the tribe Rhinantheae (Orobanchaceae). Previous studies have highlighted the presence of very interesting nonvolatile compounds in some species of this clade. Several iridoid glucosides4,5 and diterpenes 6 were identified in Parentucellia viscosa. These compounds showed many biological activities, for example, antibacterial, 7 plant growth inhibition against Italian ryegrass and lettuce seedlings, and antifeedant. 8 Terpenoids and malonic acid derivatives9,10 and iridoids 11 were isolated from P. latifolia. Besides, Ortiz De Urbina et al 12 purified from P. latifolia aucubin, catalpol, and the antispasmodic peracetylated penstemonoside. Diterpenoids, 13 γ-cyclogeranil geraniol derivatives,14,15 flavonoids, 16 and iridoids 17 were identified in the extracts of Bellardia trixago.
On the other hand, very few investigations have been reported on the volatiles of the species belonging to the Bellardia clade and concern a Sicilian accession of Bellardia trixago, 18 and Turkish populations of Parentucellia latifolia subsp. flaviflora 19 and Parentucellia viscosa. 20
In recent decades, scientific research has employed highly eco-friendly techniques to control the biodeterioration of the artistic heritage, always seeking green biocidal procedures in order to use no-harmful and nontoxic compounds, effective in different atmospheric and temporal conditions, and without negative effects on health. In particular, essential oils (EOs), whose antimicrobial properties have been known since ancient times, are mixtures of secondary metabolites and they are widely used in countering the different bacterial and fungal spread. 21 Historical artifacts, such as paintings, paper, papyri, wood, incunabula, books, and leather, are mainly formed by natural fibrous structures which are excellent colonization environments for various heterotrophic microorganisms, that is, bacteria and fungi.22,23 In recent years, several scientific articles have been used and published that deal with new natural biocides, also obtainable from waste sources, which can prevent, reduce and destroy the various colonies of microorganisms that devastate the world's artistic heritage.24–32 Cellulosic objects such as books stored in libraries, museums, and archives, are often affected by different colonies of bacteria such as Aspergillus, Fusarium, Bacillus, Trichoderma, Chaetomium, Memnoniella, Myrothecium, Stachybotrys, Verticillium, Alternaria, and Penicillium, which cause their deterioration. Additionally, the use of glues of animal and vegetable origin is a means of spreading bacterial growth. Strong microscopic and macroscopic damages have been observed, such as oxidation, depolymerization, and breakdown of the molecular and supramolecular structure, resulting in loss of elongation and strength, aging, alterations in appearance, and the formation of spots. Cellulases, for example, fibrolytic enzymes, synergistically hydrolyze cellulose obtaining cellobiose, oligosaccharides, and glucose, so they are chiefly responsible for the degradation of cellulosic fibers. 33
Consequently, in the framework of our continuous research on essential oils from Sicilian plants34–36 and the study of their biological properties,37–40 we investigated the phytochemical composition of the Sicilian essential oil of Parentucellia latifolia (L.) Caruel, obtained from the hydrodistillation of its aerial parts, as well as its antimicrobial potential by testing it on several representative Gram-negative and Gram-positive bacteria that typically affect historical art crafts.
Results and Discussion
Hydrodistillation of Parentucellia latifolia aerial parts gave a yellow oil in 0.45% yield. Overall, we identified 5 compounds, representing 99.5% of the total oil that are listed in Table 1 according to their retention indices (KIs) on an HP-5MS column. The essential oil was particularly rich in sesquiterpene hydrocarbons. Germacrene D was the main component, accounting for more than half of the total oil (59.2%), followed by germacrene B (23.4%), (E)-β-caryophyllene (9.4%), and α-humulene (6.6%). The monoterpene hydrocarbon (Z)-β-ocimene was present in only a trace amount.
Chemical Composition of the Essential Oil From Parentucellia latifolia Aerial Parts.

HP-5 MS column.
HP Innowax column.
1, retention index, 2: mass spectrum, 3: co-injection with authentic compound.
t = trace, less than 0.05%.
A comparison with the essential oils of the other taxa included in the Bellardia clade showed completely different profiles. In fact, the oil of the other subspecies, Parentucellia latifolia subsp. flaviflora, collected in Turkey, 19 was characterized by β-pinene (5.6%-27.1%), caryophyllene oxide (9.8%-32.5%), limonene oxide (13.1%-12.9%), β-ocimene (23.7%-2.7%), and (E)-anethol (15.1%-4.1%). On the other hand, the oil of Bellardia trixago aerial parts, collected in exactly the same location as Parentucellia latifolia, 18 was rich in (E,E)-farnesyl acetone (42.1%), trixagol (8.0%), and 4-vinyl guaiacol (5.4%).
Overall, the very high level of germacrene D renders P. latifolia essential oil peculiar. To the best of our knowledge, the amount of this compound is one of the highest found in essential oils. Previously, germacrene D was found to be abundant in Torilis japonica (57.9%-71.8%) 41 and Kundmannia sicula (81.2%), 42 2 members of the Apiaceae family. Therefore, P. latifolia may represent a new source of this metabolite. Germacrene D is a common plant volatile considered to be the biogenetic precursor of different sesquiterpenes such as muurolane, cadinane, and amorphane derivatives. 43
The antimicrobial activity of the essential oil is reported in Table 2. The oil was particularly active against Bacillus subtilis, Proteus vulgaris, and Staphylococcus aureus. This is in perfect agreement with data reported in the literature; in fact, the biological properties of the main metabolites (germacrene D, germacrene B, and (E)-β-caryophyllene), accounting for 92.2% in the oil, have been widely demonstrated. In fact, several studies clearly showed that (E)-β-caryophyllene and germacrene D have significant antibacterial and antifungal activities.44–50
MIC (µg/mL) and MMC
a
(µg/mL) of Essential Oils of Parentucellia latifolia (
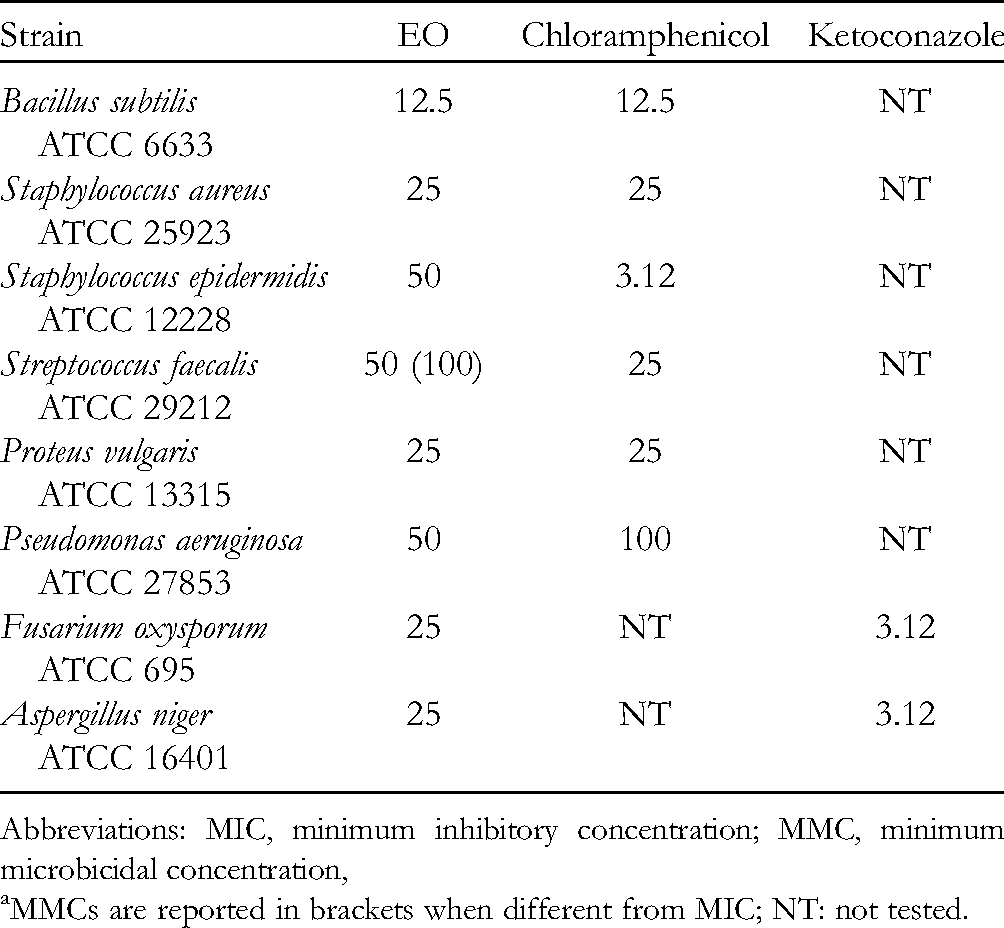
Abbreviations: MIC, minimum inhibitory concentration; MMC, minimum microbicidal concentration,
MMCs are reported in brackets when different from MIC; NT: not tested.
EOs, a complex mixture of bioactive metabolites, are now widely used as fragrances, as food preservatives, as pharmaceutical agents, and in the protection of historical-artistic artifacts. Numerous papers deal with the properties of different EOs against colonizing bacteria from museums, archives, and libraries, 51 very often showing better antimicrobial activity than synthetic fungicides.52,53 For example, the essential oils of marjoram, lemon, and rosemary, tested against colonies of yeast growing in the royal tombs of Tanis 54 showed antibacterial activity over a long time. The EOs of the Lamiaceae Lavandula angustifolia, Origanum vulgare, and Rosmarinus officinalis, used in pharmaceutical and cosmetic industries and in agriculture, 55 were active against fungal strains such as Epicoccum nigrum, Bipolaris spicifera, Aspergillus ochraceus, A. niger, Trichoderma viride, and Penicillium sp., isolated from wooden and stone objects. 25 The essential oil of Thymus vulgaris, another Lamiaceae, already tested against Fusarium sp. and Scopulariopsis sp. growing on paper documents, 56 showed really good activity against Fusarium oxysporum, Aspergillus niger (infesting libraries, archives, and historical objects), Bacillus subtilis, and Staphylococcus epidermidis (comparable to ketoconazole and chloramphenicol controls). 31
The relevant antimicrobial activity showed by the essential oil from aerial parts of Parentucellia latifolia against Bacillus subtilis, Staphylococcus aureus, and Proteus vulgaris, microorganisms typically infesting libraries, archives, and historical textiles objects, renders this plant interesting for possible applications in the disinfestation and protection of museum objects and historical art crafts.
Materials and Methods
Plant Material
Aerial parts of Parentucellia latifolia (L.) Caruel were collected at Alimena, Palermo, Sicily (37°40′50″ N; 14°05′01″ E; 645 m s/l), Italy, at the beginning of May 2019. A typical specimen (PAL 2019/68), identified by Mr Emanuele Schimmenti, is deposited in the University of Palermo, Department STEBICEF.
Isolation of the Essential Oil
The fresh samples, after being pulverized in a Waring blender, were subjected to hydrodistillation for 3 h according to a procedure previously described.57,58 The oil was dried over anhydrous sodium sulfate. Before analysis by gas chromatography (GC) and gas chromatography–mass spectrometry (GC-MS), the oil was stored in sealed vials at – 20 °C. The sample yielded (w/w) 0.45% of yellow oil, with a pleasant smell.
Gas Chromatography–Mass Spectrometry
A method reported by D’Agostino et al 59 was used for the GC–MS analysis. These were performed using a Hewlett-Packard 5890 A, equipped with a fused silica capillary column (HP-5 MS, film thickness 0.25 μm 30 m × 0.25 mm i.d.), linked online with an HP Mass Selective Detector, MSD 5970 HP; multiplier energy, 2000 V and ionization voltage, 70 eV. The oven temperature was programmed at 40 °C for 5 min, rising at 2 °C/min to 260 °C, then isothermal for 20 min; injector and detector temperatures, 250 °C and 290 °C, respectively; carrier gas, helium. The other column used to confirm the exact identification of EO compounds was HP Innowax. Peak identification was accomplished by comparison of the KIs and mass spectra with those reported in the NIST 17, WILEY275, FFNSC2, and ADAMS libraries, as well as by the comparison with authentic substances, whenever possible.
Antimicrobial Assay
Microbial Strains
The antifungal and antibacterial activities of the essential oils were tested against 8 bacterial species, representative of Gram-positive and Gram-negative classes, that is, Bacillus subtilis (ATCC 6633), Staphylococcus aureus (ATCC 25923), Staphylococcus epidermidis (ATCC 12228), Streptococcus faecalis (ATTC 29212), Proteus vulgaris (ATCC 13315), and Pseudomonas aeruginosa (ATCC 27853), and 2 fungi, Fusarium oxysporum (ATCC 695) and Aspergillus niger (ATCC 16401). The strains were grown on Sabouraud Dextrose Agar (SDA) with chloramphenicol for yeasts, SDA for molds, and Tryptone Soya Agar (Oxoid, Milan, Italy) for bacteria. For the antimicrobial tests, Sabouraud dextrose broth (SDB) for yeasts and fungal strains and Tryptone Soya broth (Oxoid, Milan, Italy) for bacteria were used.
Antimicrobial Screening
To evaluate the antimicrobial activity, the minimum inhibitory concentration (MIC) and the minimum microbicidal concentration (MMC) were determined, including minimum fungicidal (MFC), and minimum bactericidal concentrations (MBC), as described before,60,61 using the broth dilution method. 62 Oil samples were tested in triplicate.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Statement of Human and Animal Rights
Not applicable, because this article does not contain any studies with human or animal subjects.
Statement of Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
