Abstract
Ultrafiltration membrane separation is a technology used to separate large and small molecules in solution by using the principle of membrane screening. This technology can retain the effective components of traditional Chinese medicine extract, remove macromolecular impurities and endotoxin, and meet the requirements of high quality, high efficiency, and low pollution in the modern production of traditional Chinese medicine. The study investigated the impact of 100 KDa molecular weight prepared from different ultrafiltration membrane on the ginsenosides ingredient composition of ginsenosides Rg1, ginsenoside Re, ginsenoside Rf, ginsenoside Rb1, ginsenoside Rg2, ginsenoside Rc, ginsenoside Rb2, ginsenoside Rb3, ginsenoside Rd and ginsenoside Rg3. Changes in retention rates were compared by the component changes in high-performance liquid chromatography in the volume concentrates, par volume of water constant volume diafiltration and 50% ethanol constant volume diafiltration ultrafiltration process. The results demonstrate that the 100 K cellulose ultrafiltration membrane was used for ginsenosides ultrafiltration. The yield and retention were superior to those of the polyethersulfone ultrafiltration membrane with the same molecular weight cutoffs.
Introduction
The Panax ginseng C.A.Mey. is a precious Chinese herbal medicine with a long medicinal history in China. Modern studies suggest that ginsenosides are the main active ingredients of ginseng. Due to its efficacy in immunomodulatory, anti-inflammatory, and body resistance improving capacity, ginseng has become the critical prescription ingredient in modern traditional Chinese medicine for a long time.1,2 Depending on the sapogenin, the ginsenosides are divided into protopanoxadiol, protopanaxatriol, and oleanolic acid saponin, where protopanoxadiol includes ginsenosides Rb1, Rb2, Rb3, Rc, Rd, Rg3, Rh2, Rs1 and Rs2, and protopanaxatriol includes ginsenosides Re, Rf, Rg1, Rg2, Rh1, Rh3 and Rf1, manifesting various pharmacological activities in different ginsenosides .3–10 Ultrafiltration is a separation technology based on size exclusion. It can be processed without phase change or additional chemicals, and the membrane can be reused. It has been widely used in biopharmaceutical processes to remove macromolecular impurities (such as proteins, polysaccharides, endotoxin and aggregates, etc). So it has great development potential to be applied in the Chinese medicine industry. The integrated ultrafiltration liquid chromatography with mass spectrometry method was an effective and powerful tool for the discovery of active ingredients in Shenqi Jiangtang Granule and PPD-type ginsenoside enriched fraction.10,11 We found that 100 KDa ultrafiltration membrane had selective retention effects on the ingredients of ginsenosides. We studied the yields of ginsenosides by using an ultrafiltration membrane made of different materials with the same molecular weight cutoff (MWCO), analyzed the concentration changes of 10 kinds of ginsenosides in the ultrafiltration process by high-performance liquid chromatography (HPLC), and investigated the yields of the 10 kinds of ginsenosides with 100 KDa ultrafiltration membranes made of different materials and the retention rate curve of the 10 kinds of ginsenosides in the ultrafiltration process, try to find out the reason for the selective retention of ingredients of ginsenosides by the ultrafiltration membrane.
Experimental
Materials and Methods
Chemicals and Reagents
Total ginsenosides; ginsenosides Rg1, Re, Rb1, Rb2, Rb3, Rc, Rd, Rf, Rg2, Rg3 standard control (From National Institutes for Food and Drug Control, batch No.:111562-200605); methanol and acetonitrile (Merck KGaA, CAS-No:75-05-8); phosphoric acid and sodium hydroxide (analytical reagents, CAS:13472-35-0).
Instrumentations
LabscaleTM TFF system (Merck Millipore Corporation), Mini Pellicon TFF system (Merck Millipore Corporation) Biomax 50, 100 kDa NMWL, 50 cm2 (Merck Millipore Corporation), Ultracel 100 kDa NMWL 0.1 m2 (Merck Millipore Corporation); AL204 electronic balance, XS105 Mettler precision electronic analytical balance (on Mettler-Toledo Instruments Co., Ltd); HPLC Waters2695; Waters2489 detector; Waters Symmetry® C18 (250 × 4.6 mm, 5 μm) column; water bath.
Method Development
HPLC Conditions
Column: Waters Symmetry® C18 (250 × 4.6 mm, 5 μm); acetonitrile as mobile phase A, 0.05% aqueous phosphoric acid as mobile phase B, gradient elution was carried out as listed in Table I. Detection wavelength is 203 nm, column temperature is 30 °C, flow rate is 1 mL/min, injection volume is10 µL.
Gradient Elution Procedure.
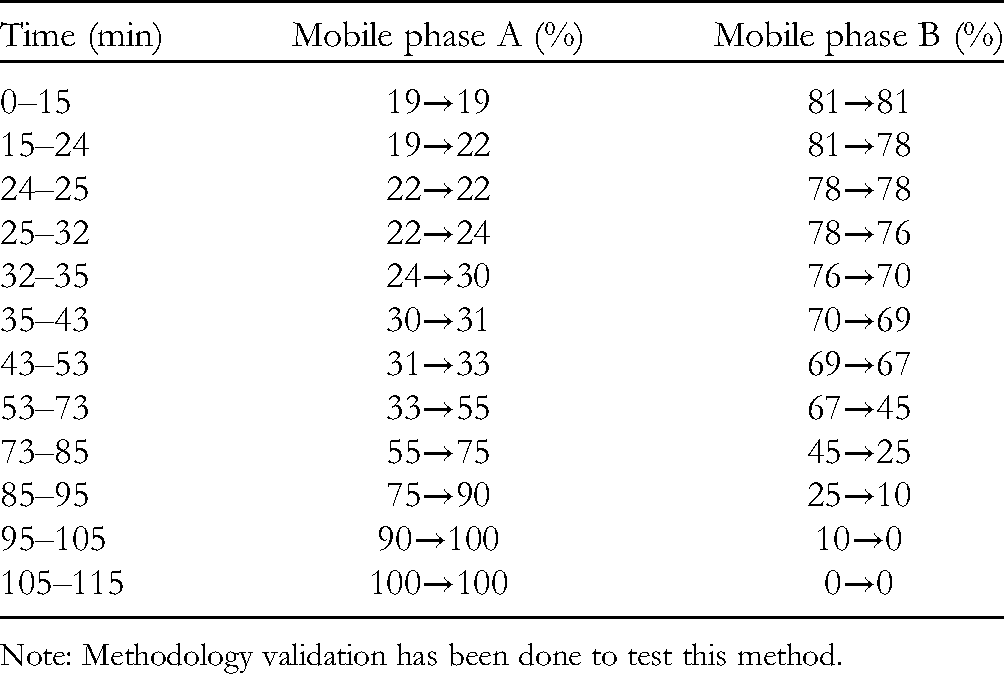
Note: Methodology validation has been done to test this method.
Preparation of Reference Sample of Ginsenosides
Ginsenosides Rg1, Re, Rf, Rb1, Rg2, Rc, Rb2, Rb3, Rd, Rg3 controls were prepared with methanol. The concentrations of ginsenosides Rg1, Re, Rf, Rb1, Rg2, Rc, Rb2, Rb3, Rd, and Rg3 were 0.2096, 0.09801, 0.0925, 0.2989, 0.1061, 0.1049, 0.1977, 0.0689, 0.1001, and 0.0465 mg mL−1, respectively.
Concentration Determination
In total, 10 μl of ginsenoside control, ultrafiltration permeate, and ultrafiltration Retentate were taken for the monomer ginsenosides HPLC test.
Preparation of Ultrafiltration Stock Solution
In total, 5 mg mL−1 ginsenosides raw materials were prepared using purified water and filtrated by a 0.45 μm hydrophilic membrane (Merck Millipore).
Ultrafiltration
In total, 20 L m−2 of stock solution was ultrafiltrated by Biomax 100 KDa and Ultracel 100 KDa under a feed flow rate of 5 L/min m2, the inlet pressure is 30 psi, and the retentate pressure is 10 psi.
Five samples were taken both from permeate and retentate during concentration for ginsenoside concentration determination.
Another five samples were taken from permeate and retentate during constant volume diafiltration against purified water when the diafiltration factor was1, 2, 3, 4, and 5 after 10 times concentration.
After 5 times diafiltration of purified water, 50% ethanol solution was used for constant volume diafiltration of ginsenoside. When the diafiltration number reached 0.5, 1, 1.5, 2, and 3, samples from permeate and retentate were taken to determine the ginsenoside concentration.
The ultrafiltration membrane should be cleaned immediately after each use, according to the ultrafiltration membrane cleaning standard operating procedures. The cleaned ultrafiltration membrane can be directly used in the next test when the membrane integrity test shows an intact result. The membrane cleaning in this paper followed the above method, and measured membrane integrity was always within the specification.
Comparison of Ginsenosides Concentration Before and After Ultrafiltration
According to the ginsenoside concentration in permeate and retentate, the yields of each monomer ginsenoside and the ultrafiltration retention rates of each monomer ginsenoside of each sampling point can be calculated.
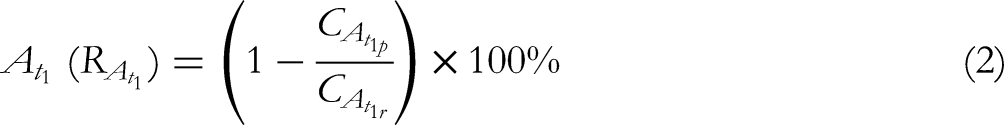



Results
The ultrafiltration and determination of ginsenoside concentration are based on the ultrafiltration conditions in the above ultrafiltration method, and the results are as follows (Figure 1).

High-performance liquid chromatography (HPLC) spectrum of 10 kinds of monomer ginsenosides.
The Selective Adsorption Ability of Biomax 100 K
For Biomax 100 K, during the concentration step, the retention rate of each active ingredient increased with the volume concentration factor (VCF) (Figure 3IA). The retention rate of each monomer ginsenoside is Rg3>Rb2>Rd>Rb3>Rc>Rb1>Rg2>Rf>Re>Rg1. The situation was similar in the process of constant volume diafiltration against par volume of water (PW) (Figure 3IB). The yields of ginsenosides Rg1 and Re reached 100% after PW diafiltration, The yields of ginsenoside Rf and Rg2 were 93.09% and 87.04%; But the yields of ginsenoside Rb1, Rc, Rb2, Rb3, Rd, and Rg3 were 55.91%, 52.74%, 30.75%, 44.84%, 36.25%, and 9.57% (Figure 2B). There was significant yield loss found after PW diafiltration for some kinds of ingredients when using Biomax 100 K, which proposed that some ginsenosides, especially protopanoxadiol (Rb1, Rb2, Rb3, Rc, Rd, Rg3, Rh2) were highly retained, so 50% ethanol was used for further diafiltration. Active ingredients retention rates have substantially decreased for 50% ethanol compared to those in aqueous solution retention rates of active ingredients have substantially decreased for 50% ethanol compared to those in aqueous solution. Some retention rates even turn negative, it indicated that the 50% ethanol has greatly improved the permeate rates of these components.
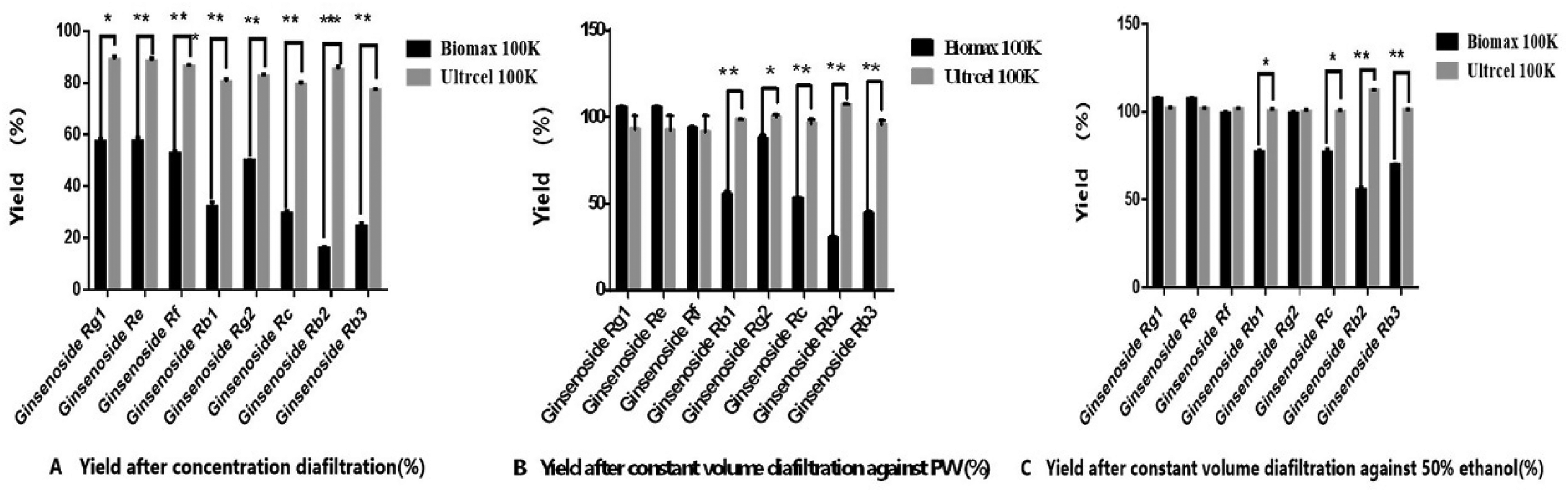
Monomer ginsenosides yield comparison using Biomax/Ultracel 100 KDa at different ultrafiltration stages (***
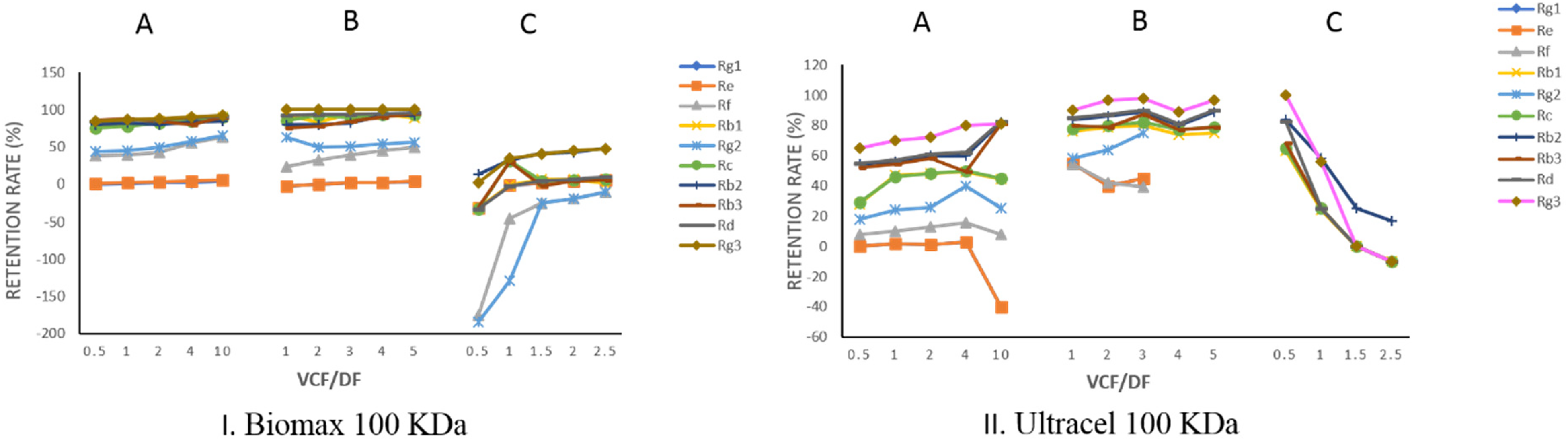
Retention rate change of active ingredients for Biomax 100 KDa and Ultracel 100 KDa in different ultrafiltration stages (A) concentration; (B) constant volume diafiltration against par volume of water (PW); (C) constant volume diafiltration against 50% ethanol.
For Ultracel 100 KDa, during the concentration step, the retention rate of each active ingredient decreased with VCF (Figure 3IIA). The retention rates at each point, in general, were significantly lower than those by using Biomax 100 KDa under the same conditions. Figure 2 showed that after PW constant volume diafiltration, the yields of ginsenosides Rg1, Re, Rf, and Rg2 reached 100%. The yields of ginsenosides Rb2, Rb1, RC, Rb3, Rd, and Rg3 were 100%, 98.76%, 97.74%, 96.45%, 89.82%, and 66.17%, respectively; the concentration of each component in the sample was continuously reduced with an increase of PW diafiltration factors.
The Ultrafiltration Ability of Different Ultrafiltration Membrane
For Ultracel 100KDa, from Figure 2 and Figure 3IIB, when the diafiltration factor reached 3, the concentrations of ginsenosides Rg1, Re, Rf, and Rg2 in the ultrafiltration samples were below the detection limit, the retention rates of ginsenoside Rb2, Rb1, Rc, Rb3, Rd, and Rg3 remained at 80% to 100%. From Figure 3IIC, in the 50% ethanol constant volume diafiltration process, the measured retention rates of Rc, Rb2, Rb3, Rd, and Rg3 were significantly lower than those in aqueous solution, indicating 50% ethanol constant volume diafiltration had improved the permeate rates of each component.
In Figure 2, with the increase of diafiltration volume, the yields of ginsenoside ingredients continued to increase. The yield of Ultracel 100 KDa was significantly better than Biomax 100 K for each monomer ginsenoside after constant volume diafiltration against PW/% and constant volume diafiltration against 50% (*
The Diafiltration on Permeate Rate of Each Membrane
To further study the retention causes, in the following part, the concentration changes of 10 kinds of ginsenosides in ultrafiltration retentate samples at each sampling point were investigated (Table 2).
Concentration of Each Active Ingredient in Feedstock Before Ultrafiltration.

Note: The concentration of 10 kinds of ginsenosides in control at ultrafiltration initial retentate C0 was set to 1, and the other points were compared with it.
Figure 4IA showed that in the volume concentration ultrafiltration process using 100 KDa polyethersulfone ultrafiltration membrane, with the increase of the VCF, the concentration of the active ingredients in retentate increased in different degrees, and the concentrations in descending order were ginsenosides Rg3, Rb2, Rd, Rb3, Rc, Rb1, Rg2, Rf, Rg1, and Re. Figure 4IB showed that the concentration of each component in the retentate had varying degrees of downward trends in PW constant volume diafiltration stage. Figure 4IC showed that in the 50% ethanol constant volume diafiltration, the concentration of other ingredients in retentate tended to be constant at a low concentration level, with the increase of diafiltration factors in addition to a small increase of concentration of ginsenosides Rg3 and Rb2.

Concentration changes of active ingredients in retentate for Biomax 100 KDa and ultracel 100 KDa in different ultrafiltration stages. (A) Volume concentrates stage; (B) par volume of water (PW) constant volume diafiltration stage; (C) 50% ethanol constant volume diafiltration stage. Note: When the volume concentration factor was 1, the concentrations of 10 kinds of ginsenosides in retentate C0 were compared with other points.
The comparison of Figure 4IA and Figure 4IIA showed that in the volume concentration ultrafiltration process using 100 KDa cellulose ultrafiltration membrane, the concentration of each active ingredient in the retentate increased with increasing volumetric concentration factor, however, the high amplitude was less than that of 100 KDa polyethersulfone ultrafiltration membrane under the same experimental conditions. Figure 4IIB shows that in the PW constant volume diafiltration, with the increase in diafiltration factors, the concentration of each component in the retentate tended to decrease with the increasing percolation factor. Figure 4IIC shows that in the 50% ethanol constant volume diafiltration, there is a significant decreasing trend in the concentration of each component in the retentate tended to decrease with increasing percolation factor, apparently different from Figure 4IC, indicating that 50% ethanol constant volume diafiltration has a more pronounced effect on the permeability of each gradient of the 100 KDa cellulose membrane than that of the 100 KDa polyethersulfone membrane.
Discussion
Ginsenosides are Supposed to be the Main Active Ingredients of Ginseng
According to the glycosides, ginsenosides can be divided into protopanaxadiol, protopanaxatriol, and oleanolic acid saponins, where protopanaxadiol includes ginsenosides Rb1, Rb2, Rb3, Rc, Rd, Rg3, Rh2, Rs1, and Rs2, and protopanaxatriol includes ginsenosides Re, Rf, Rg1, Rg2, Rh1, Rh3, and Rf1. The molecular weight of these ginsenosides is about 1000 Da,2,12,13 so they can penetrate through the ultrafiltration membrane with 100 KDa MWCO theoretically. Nevertheless, the experimental results are not satisfactory, indicating that other factors than molecular mass decrease the yield of these components, 14 increasing the retention of ginsenosides by ultrafiltration membranes.
The Hydrophilic Properties of Polyethersulfone and Cellulose Materials
The hydrophilic properties of polyethersulfone and cellulose materials are different, so the impact of the ultrafiltration membrane made of the 2 materials on the yield of ginsenosides is different. In this study, it was found that the polyethersulfone ultrafiltration membrane had a strong selectivity for monomeric ginsenosides in an aqueous solution, and the effect of the ultrafiltration membrane on the aqueous solution of protopanaxadiol was significantly greater than that of protopanaxatriol. A similar trend was observed for the effect of cellulose ultrafiltration membrane on ginsenosides, but the effect was not as pronounced as that of the polyethersulfone material. From the yields of ginsenosides in solution with the ultrafiltration membrane made of the 2 materials, the adsorption of ginsenosides adsorption of ginsenosides by the cellulose ultrafiltration membrane was significantly smaller than that by the polyethersulfone ultrafiltration membrane.
From the aspect of the experimental yields, the applicability of cellulose ultrafiltration membrane in ultrafiltration of ginsenosides was superior to polyethersulfone ultrafiltration membrane at the same MWCO. However, the effects of ultrafiltration membranes made of both materials on the 10 ginsenosides were similar, ie the ultrafiltration membranes had a selective effect on the yield of ginsenosides, which was more pronounced for protopanaxadiol than for protopanaxatriol. The protopanaxadiol saponins are anti-hemolytic, the protopanaxatriols are hemolytic saponins, and the hemolysis or anti-hemolysis are related to their structural characteristics and surface activities. The chemical structure of 10 kinds of ginsenosides in Tables 3 and 4 and Figure 5 showed that the protopanaxatriols have one more hydroxyl at the C-6 position with strong hydrophilic nature, therefore, the differences in physical and chemical properties of ginsenoside and hydrophilicity of the 2 material were likely to be one of the essential reasons that caused the above selectivity. 15

Chemical structure of protopanaxatriol ginsenosides.
Protopanaxatriol Ginsenosides.

The Protopanaxadiol Ginsenosides.

The Ginsenoside can Reduce the Surface Tension of its Aqueous Solution
The surface tension was related to the proportion of hydrophilicity and lipophilicity of its internal molecular structure. If the proportion was appropriate, it was easy to assemble in aqueous solution, which will hinder the ginsenoside from penetrating through the ultrafiltration membrane. To prove this hypothesis, 50% ethanol was added quantitatively to replace ginsenosides in the ultrafiltrate by using constant diafiltration in the final stage of ultrafiltration, which resulted in a further increase in the yield of each ginsenoside and a significant decrease in the retention rate of 10 ginsenosides by ultrafiltration membranes made of both materials. The experiment results showed that the high retention rate of ultrafiltration membrane on some ginsenoside was probably due to, on the one hand, the adsorption of the filtration membrane on the components, and the other hand, retaining of small molecules ginsenoside by large pore ultrafiltration membrane. To verify the above reasoning, high concentrations of ginsenoside in aqueous solution and ethanol solution were passed through SEC, and the presence of volumetric substances in the aqueous solution, but not in the ethanol solution, was found, which was verified as ginsenosides after further separation of such volumetric substances. Therefore, it could be determined that the reason for the retention of ginsenosides was that small-molecule ginsenosides formed bulky aggregates in an aqueous solution, which could not easily pass through the ultrafiltration membrane.
Conclusions
Ultrafiltration membrane can selectively adsorb the monomeric components of ginsenoside in aqueous solution. Compared with the cellulose ultrafiltration membrane, the adsorption capacity of the polyethersulfone ultrafiltration membrane was stronger. For ginsenoside recovery, the cellulose ultrafiltration membrane has a higher recovery rate of ginsenoside, and the cellulose ultrafiltration membrane is more suitable for ginsenoside recovery than the polyethersulfone ultrafiltration membrane.
Footnotes
Acknowledgments
The authors would like to thank Merck Chemicals (Shanghai) Co., Ltd for supporting the membrane for this experiment, and also thank Jing Xue and Kailong An for their help in this experiment.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Science and Technology Major Project on “key new drug creation” (grant number 2010ZX09502-004).
