Abstract
A new cembrane-type diterpenoid (
Keywords
Introduction
Cembrane-type diterpenoids (cembranoid), comprising 4 isoprene units, are a kind of 14-member macrocyclic diterpene that have been isolated and identified mainly from marine organisms (eg soft corals and gorgonians) and terrestrial plants (eg tobacco, Pinus, myrrh, and frankincense).1,5 As reported previously, 105 cembranoids have been obtained from Nicotiana tabacum, which possess a wide range of biological functions,
1
including antitumor, anti-inflammatory, antibacterial, anti-metastatic, anti-pathogenic, neuroprotective, anti-fouling, and anti-osteoporotic properties. However, studies of bioactivities of those metabolites to date have mainly focused on α-CBT-diol and β-CBT-diol.
6
In our current study, we report the isolation, structure identification, and neuroprotective effects of a new cembranoid (

Cembranoids
Results and Discussion
Compound
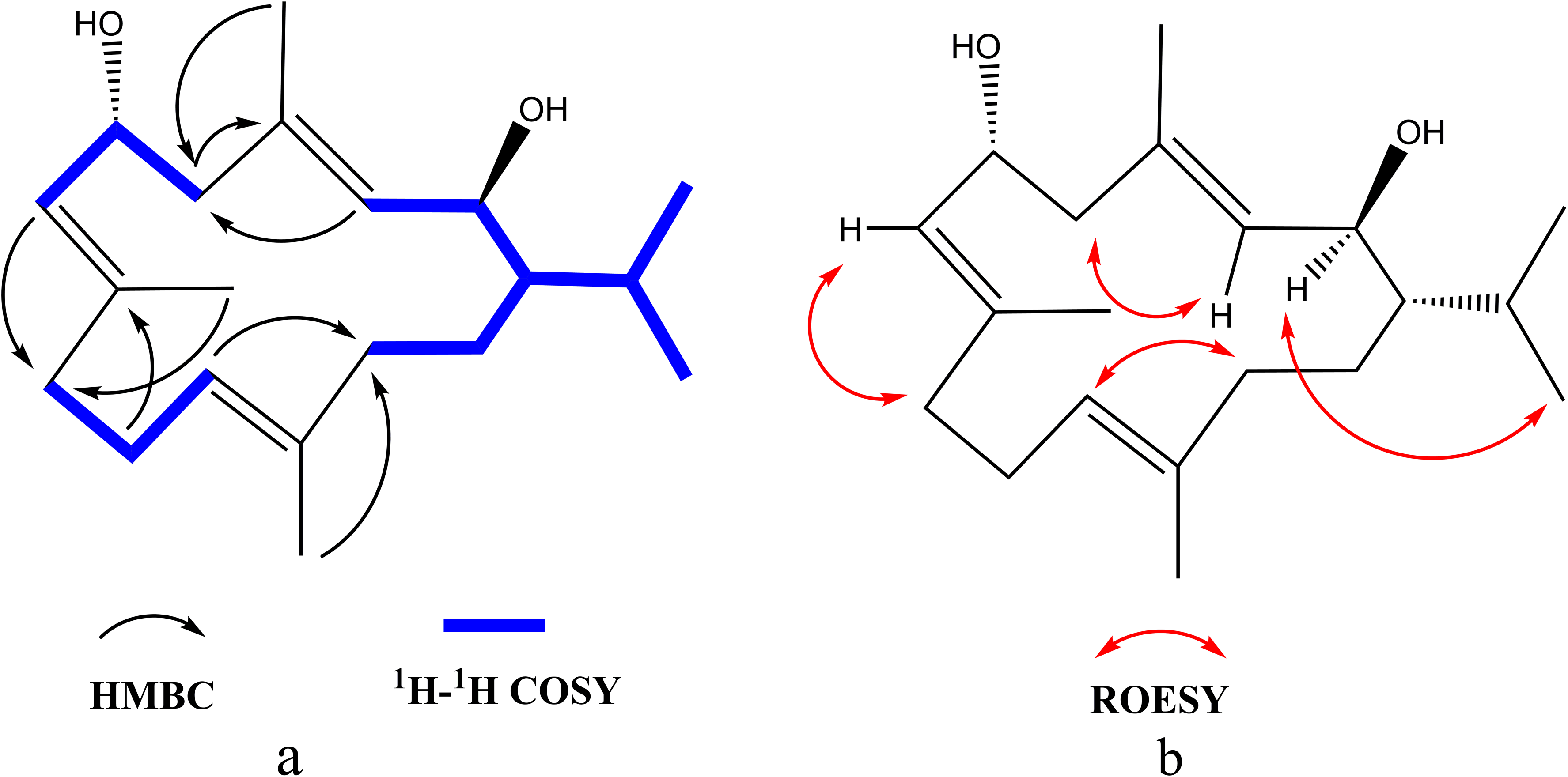
(a) 1H − 1H COSY and key HMBC correlations of compound
1H and 13C-NMR Spectroscopic Data of Compound

Abbreviation: NMR, nuclear magnetic resonance.
By comparing their observed and reported spectroscopic data, 9 known compounds were determined as (1S,2E,4S,6R,7E,11E)-2,7,11-cembratriene-4,6-diol (
To the best of our knowledge, studies of the neuroprotective activities of cembranoids from tobacco to date have mainly focused on α-CBT-diol and β-CBT-diol. Thus, all of our isolates were evaluated for their neuroprotective effects against OGD-induced cell injury in SH-SY5Y cells. Compounds

Neuroprotective effect of compounds 1, 2, 5, and 7 against oxygen-glucose deprivation (OGD)-induced toxicity in SH-SY5Y cells.
Experimental
General Experimental Procedures
IR spectra were measured on a Thermo Scientific Nicolet 5700 spectrometer, NMR spectra using a Bruker ascend 600 MHz spectrometer, and HR-ESI-MS on an AB SCIEX Triple TOF 6600 mass spectrometer. Preparative high-performance liquid chromatography (HPLC) was carried out on a Waters Prep 150 LC system using an YMC-Pack ODS column (250 × 10 mm, 5 μM). Column chromatography was performed on silica gel (200-300 mesh, Qingdao Marine Chemical Co., Ltd ) and Sephadex LH-20 (Pharmacia Fine Chemicals) columns. Neuroprotective activity was performed in a super clean workbench (Suzhou Antai Airtech Co., Ltd). Cells were cultured in a Thermo Scientific Steri-Cycle CO2 incubator. Cell counting was performed under an inverted microscope (Sunny Optical Technology Co., Ltd). The absorbance was detected with a microplate reader (Bio-Rad Laboratories, Inc) at 490 nm wavelength. All of the other chemicals and solvents were of analytical grade.
Plant Material
The leaves of N tabacum L. were collected from Sanmenxia city (Henan province, China) in August 2018 and authenticated by associate professor, Shen Huang, from Zhengzhou University of Light Industry. A voucher specimen (20180024) was stored in the laboratory of the Duobin Mao research group.
Extraction and Isolation
The fresh green leaves of N tabacum (960 kg) were rinsed 4 times with CH2Cl2. After the CH2Cl2 had been evaporated under reduced pressure, dark yellow residues (516 g) were obtained. The obtained extracts were subjected to chromatography on a silica gel column (2000 × 50 cm i.d.) and eluted successively with light petroleum/ethyl acetate (100:0, 50:1, 30:1, 10:1, 5:1, 3:1, 1:1, and 0:1, v/v) as the mobile phase, and finally washed with MeOH to yield 9 fractions (fractions A-I). Fraction D (34 g) was separated on a Sephadex LH-20 column (120 × 10 cm i.d.) and eluted with a MeOH/CH2Cl2 (1:1, v/v) mixture using liquid chromatography with a photodiode array detector to give fractions D1-D9. Fraction D5 was further chromatographed on a silica gel column and separated by light petroleum/ethyl acetate (10:1, 7:1, 5:1, 3:1, and 1:1) to generate fractions D5-1 to D5-12, Fraction D5-9 was purified by reverse preparative HPLC using MeCN/H2O (55:45, v/v) as the mobile phase at 5 mL/min to yield compounds
Neuroprotective Activity Assays
Human neuroblastoma SH-SY5Y cells were maintained in dulbecco's modified eagle medium (DMEM) supplemented with 10% fetal bovine serum (FBS), 100 U/mL penicillin, and 100 μg/mL streptomycin in a humidified incubator at 37 °C under a water-saturated atmosphere of 5% CO2 and 95% air. Cells, in the log growth phase, were seeded in 96-well culture plates (1 × 105 cells/well in 100 µL) until 80% to 90% confluent for the next experiments. After pretreatment with compounds at 10−6 M for 2 h, SH-SY5Y cells were exposed to glucose-free DMEM in a hypoxia incubation chamber with 95% N2% and 5% CO2 for 1 h and then cultured for another 24 h under normal conditions. These cells were included in the OGD/R group compared with SH-SY5Y cells cultured with complete medium under normal conditions as the control group. Cell viability was evaluated by incubating with 1 mg/mL 3-[4,5- dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide (MTT) for 4 h at 37 °C in an incubator. Media were then replaced with 100 μL DMSO and absorbance was read at 490 nm. Data were analyzed by one-way analysis of variance (ANOVA) and expressed as means ± SD with P <.05 regarded as significant.
Conclusion
A new cembranoid, (1S,2S,3E,6R,7E,11E)-3,7,11-cembratriene-2,6-diol (
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221094107 - Supplemental material for Cembranoids From the Leaves of Nicotiana tabacum and Their Neuroprotective Activities
Supplemental material, sj-docx-1-npx-10.1177_1934578X221094107 for Cembranoids From the Leaves of Nicotiana tabacum and Their Neuroprotective Activities by Peng-fei Yang, Mei-yu Chen, Jian-chun Yu, Jing Yang and Qiong-bo Wang, Duo-bin Mao in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant number 81903507).
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
