Abstract
Introduction
The incidence of renal anemia in patients with chronic kidney disease (CKD) gradually increases with attenuation of the glomerular filtration rate. More than half of patients with stage 3 CKD and 90.2% of patients with stage 5 CKD have renal anemia.1 Anemia can reduce the body's oxygen partial pressure, increase cardiac output, and induce left ventricular hypertrophy, thereby reducing cognitive function and concentration. 2 Accumulating evidence indicates that patients with systemic inflammation, such as those with CKD, have inflammatory anemia. 3 Chronic inflammation can cause inflammatory anemia by preventing proliferation of myeloid precursors, activating more macrophages to phagocytose red blood cells, shortening erythrocyte lifespan, and inducing increased synthesis of hepcidin. 4 Studies have confirmed that roxadustat can reduce hepcidin levels in dialysis 5 and non-dialysis patients 6 with CKD, and regulate ferritin metabolism. However, how to improve the proliferation of myeloid precursors inhibited by chronic inflammation or reduce the activation of macrophages remains unknown.
From a holistic perspective, Chinese medicine has the advantage of improving chronic inflammation in patients. Danggui Buxue Decoction (DBD) was first introduced in Li Dongyuan's “Distinguishing Theory of Internal and External Injury.” DBD is composed of Astragalus and Angelica in a five-to-one combination. Studies have confirmed that DBD can treat anemia. Using metabonomics and network pharmacology, Liu et al. showed that DBD could maximize the synergy of Astragalus and Angelica in the treatment of anemia. 7 Moreover, Zhang et al. confirmed that DBD inhibited the activation of the Janus kinase 2-signal transducer and activator of transcription 3 (JAK2/STAT3) pathway and produced active anti-inflammatory factors. 8 Li et al. found that DBD reduced oxidative stress in diabetic nephropathy and improved pathological changes in the kidneys of rats, 9 and DBD reduced the expression of nuclear factor-kappa B and monocyte chemoattractant protein-1 to inhibit renal inflammation and protect kidney tissue. 10 These results suggest that DBD has a protective effect on renal function by improving the chronic inflammatory state. However, whether DBD treats renal anemia by regulating inflammation in CKD patients is still unknown. Therefore, using network pharmacology, this study explored inflammation-related network targets of DBD in the treatment of renal anemia. Moreover, we verified the interaction between active ingredients of DBD and inflammatory proteins by molecular docking.
Materials and Methods
Screening of Active Components of DBD
The Traditional Chinese Medicine System Pharmacology Database and Analysis Platform (TCMSP) is a platform that explores the relationships between drugs, targets, and diseases. 9 “Angelica” and “Astragalus” were searched in the TCMSP database. The criteria for screening the active ingredients of Angelica and Astragalus were oral bioavailability (OB) ≥ 30% and drug-likeness (DL) ≥ 0.18. The potential targets of DBD were screened and acquired using the TCMSP and UniProt databases.
Target Identification for Drug and Disease-Related Effects
Biomolecules related to renal anemia were searched and screened using GeneCards Pharmgkb (https://www.pharmgkb.org) and DrugBank (https://go.drugbank.com/). The targets of DBD were matched with biomolecules related to renal anemia, and the overlapping parts were selected as the targets of DBD in renal anemia therapy. Venn diagrams were drawn using Venny 2.1 online tools (https://bioinfogp.cnb.csic.es/tools/venny/).
Construction of Drug Active Ingredients and Disease Network
The targets of DBD in the treatment of renal anemia were mapped with active ingredients of DBD to establish the corresponding relationship between the effective active ingredients and their targets. Then, files including “effective active ingredients,” “key targets,” “attributes,” and “effective active ingredient-key target relationships” were created and imported into Cytoscape (Version 3.8.0) to construct an “ingredient-target” network. The degree of active ingredients, which suggests the number of interconnected nodes in the network, was calculated using CentiScape.
Construction of Protein-Protein Interaction Network and Filtration of Targets
The targets of DBD were imported into the STRING database (https://string-db.org/), which collects many proteins and protein-protein interactions (PPIs). 11 The “multiple proteins” analysis function was used, “Homo sapiens” was selected as the species, and a high confidence level of 0.7 was set. Then, a PPI network that included key targets was constructed, and the TSV format was downloaded and imported into Cytoscape. Betweenness centrality (BC), closeness centrality (CC), degree centrality (DC), eigenvector centrality (EC), local average connectivity (LAC), and network centrality (NC), which measure the importance of nodes and indicate the nature of nodes in the network, were calculated using App CytoNCA. The higher the BC, CC, DC, EC, LAC, and NC values, the more essential the node in the PPI network. Therefore, the essential targets with high PPI network values were filtered.
Gene Ontology and Kyoto Encyclopedia of Genes and Genomes Enrichment Analyses
The Bioconductor software package “org.Hs.egdb” was installed in the R software (Version 4.0.2) to obtain the entrezID of key targets. The “clusterProfiler” package was also installed to obtain the Gene ontology (GO) function and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment. In the programming language, P < .05 and q < 0.05 were set. The top 30 items with the highest enrichment in the analysis were selected and presented as bar graphs or bubble graphs.
Molecular Docking
The effective active ingredient with the highest DC value in the “ingredient-target” network was subjected to molecular docking with the core targets screened out by the PPI network. In addition, roxadustat, a drug used to treat renal anemia, was chosen for molecular docking with the core targets. The 2D structure of the compound was obtained from the PubChem database (https://www.chemicalbook.com/), the 3D construction of the targets was acquired from the PDB database (https://www.rcsb.org/), and the 2D structure of the compound was converted to a 3D structure using Chem3D. Active pocket analysis and molecular docking were performed using the AutoDock Vina software and optimized using the PyMOL software.
Results
Active Ingredients and Targets of DBD
Two active ingredients of Angelica and 20 active ingredients of Astragalus were obtained. In addition, 865 targets of Angelica and 954 targets of Astragalus corresponding to all components were obtained. One hundred and ninety-three targets corresponding to the 2 active ingredients in Angelica and 20 active ingredients in Astragalus were processed using the Strawberry Perl software.
Construction of the Disease Target set for Drug- and Disease-Related Effects
“Renal anemia” was searched in the GeneCards, OMIM, DrugBank, Pharmgkb, and TTD databases. A total of 5246 targets were obtained from GeneCards, 191 from OMIM, and 4 from DrugBank. No relevant data were retrieved from Pharmgkb and TTD. A total of 5298 biomolecules related to renal anemia were identified (Figure 1a). After mapping with the 193 targets corresponding to the active ingredients, 158 biomolecules for the active ingredients of Angelica and Astragalus in the treatment of renal anemia were selected (Figure 1b).

(a) A total of 5298 genes related to renal anemia. (b) The 158 target genes for the active ingredients of Angelica and Astragalus in the treatment of renal anemia.
Establishment of an “Ingredient-Target” Network
The 158 targets of DBD for the treatment of renal anemia were mapped with 2 Angelica sinensis and 20 Astragalus active ingredients. The connection between the effective components of DBD and their targets was established. Then, 2 Angelica and 16 Astragalus effective active ingredients related to the DBD treatment of renal anemia were acquired (Table 1). Subsequently, an “ingredient-target” network was established (Figure 2). In the network, nodes represent active ingredients and targets. Edges between nodes represent intermolecular interactions. Edges indicate the interaction relationship between active ingredients and targets. 12 Red nodes represent active ingredients and targets for Angelica, and blue nodes represent those for Astragalus. The DC value estimates the number of interconnected nodes in the network. The larger the node DC, the more important the active ingredient in the network. According to the DC value, the top 5 active ingredients are quercetin, kaempferol, 7-O-methylisomucronulatol, formononetin, and isorhamnetin (Table 2).

“Ingredients-target” network of DBD (Danggui Buxue Decoction).
Two Angelica and 16 Astragalus Effective Active Ingredients Related to the Treatment of Renal Anemia With DBD.
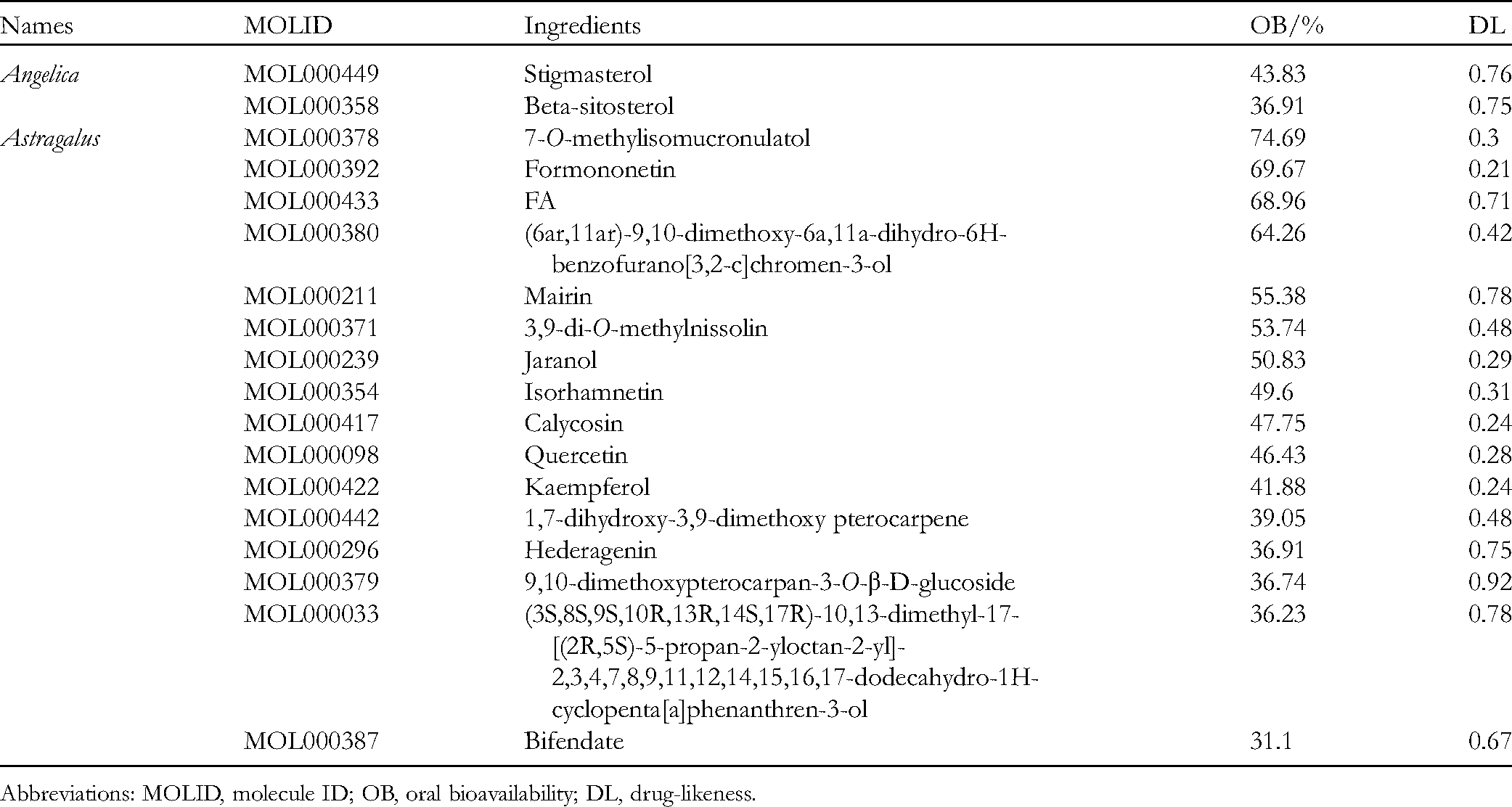
Abbreviations: MOLID, molecule ID; OB, oral bioavailability; DL, drug-likeness.
Degree Centrality of Active Ingredients in “Ingredient-Target” Network.

Abbreviation: MOLID, molecule ID.
PPI Network Analysis
Targets related to DBD in patients with renal anemia were searched on the STRING page using the aforementioned filter conditions. Then, PPI-TSV files were imported into Cytoscape, and DC, BC, CC, EC, NC, and LAC parameters were analyzed using CytoNCA. After the first screening of DC > 10, BC > 33.49117728, CC > 0.2296875, EC > 0.042733967, NC > 6.3680555555, and LAC > 5.325, 47 nodes were gained. The second screening was based on DC > 19, BC > 12.6407074, CC > 0.630136986, EC > 0.134702757, NC > 13.84794495, and LAC > 12.23076923, and we identified 20 nodes by which core target genes were obtained, including MAPK14, VEGFA, ESR1, MYC, EGF, FOS, MMP9, JUN, MAPK1, CCND1, STAT1, AR, IL1B, RELA, AKT1, TP53, C-X-C CXCL8, CASP3, MAPK8, and PTGS2. The PPI network is shown in Figure 3, where the core proteins of the PPI are indicated in yellow.
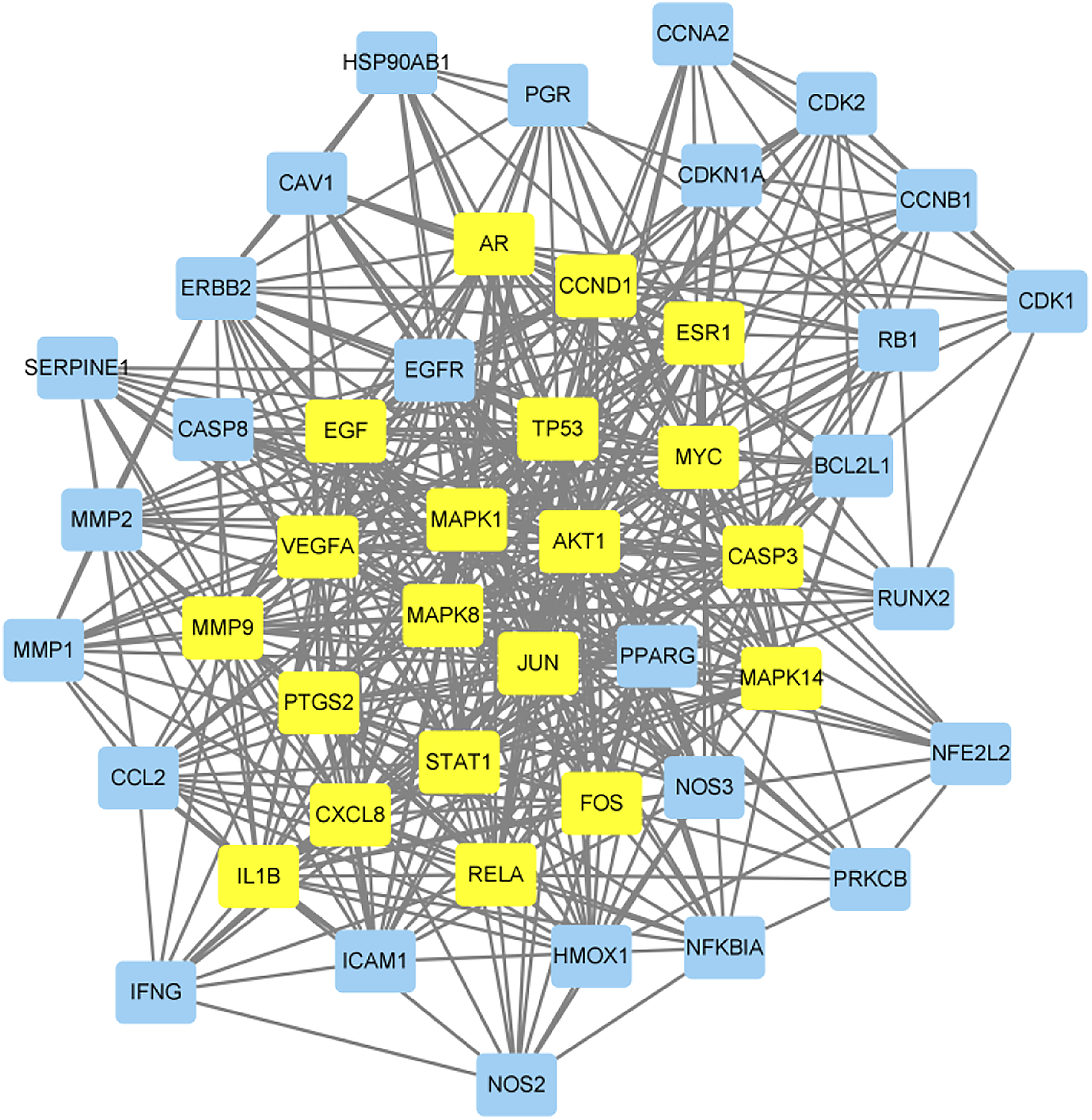
Construction of PPI network. The core proteins of the PPI network are indicated in yellow.
GO Enrichment Analysis
Through GO database analysis, 2425 GO items were enriched for targets related to DBD in patients with renal anemia. These items were subsequently sorted according to their q-value. The top 30 items, with q < 0.05 serving as the threshold, are displayed (Figure 4a). As shown in Figure 4a, the inflammation-related GO terms associated with DBD included lipopolysaccharide, response to molecules of bacterial origin, cellular response to chemical stress, response to metal ions, and oxidative stress-related reactions.

(a) GO function enrichment indicates the key targets of DBD in the treatment of renal anemia. (b) KEGG function enrichment indicates potential key targets of DBD in the treatment of renal anemia.
KEGG Enrichment Analysis
The targets related to DBD in the context of renal anemia were mapped to the KEGG pathways. A total of 167 pathways were enriched and sorted according to their q-values. The top 30 pathways, with q < 0.05 serving as the threshold, are displayed (Figure 4b). The core inflammatory pathways of DBD in renal anemia therapy included the AGE-RAGE pathway of diabetic complications, IL-17 signaling pathway, tumor necrosis factor (TNF) signaling pathway, phosphoinositide 3-kinase-protein kinase B (P13K-Akt) signaling pathway, Th17 cell differentiation, MAPK signaling pathway, intracellular senescence, and hypoxia-inducible factor (HIF) signaling pathway.
Molecular Docking Analysis
Quercetin, which was selected by the “ingredient-target” network, was subjected to molecular docking with inflammation-related proteins (VEGFA, STAT1, CXCL8, IL-1β, and PTGS2) that were screened by the PPI network. Roxadustat, which is used to treat renal anemia, was also subjected to molecular docking with inflammation-related proteins.
The more stable the binding between the ligand and receptor, the lower the binding energy. 13 The results showed that the binding energies of quercetin and roxadustat to the 5 core proteins were less than −6 kcal·mol−1 (Table 3). The results were imported into the PyMOL software for visual optimization and presented as a 3D map of molecular docking. The green and yellow parts of the map represent the molecular ligands and macromolecular receptors, respectively (Figure 5). The binding affinities obtained using MD simulation are shown in Supplements 1 and 2.

Molecular docking diagram of inflammation-related proteins, quercetin, and roxadustat: (a, b, c, d, and e) molecular docking diagram of CXCL-8, IL-1β,PTGS2, STAT1, and VEGFA with quercetin; (f, g, h, I, and j) molecular docking diagram of CXCL-8, IL-1β,PTGS2, STAT1, and VEGFA with roxadustat.
Binding Energy of Quercetin, Roxadustat, and Core Proteins.

Abbreviations: VEGFA, vascular endothelial growth factor A; STAT1, signal transducer and activator of transcription 1; CXCL8, C-X-C motif chemokine ligand 8 (interleukin 8); IL-1β, interleukin-1beta; PTGS2, prostaglandin-endoperoxide synthase 2.
Discussion
We found that the core components of DBD for the treatment of renal anemia are quercetin, kaempferol, 7-O-methylisomucronulatol, formononetin, and isorhamnetin based on network pharmacology, and the core target proteins are STAT1, CXCL8, PTGS2, IL-1β, and other inflammatory factors. Further molecular docking revealed that the binding of quercetin and these core proteins is stable. This result suggests that DBD improves renal anemia by regulating not only the cell cycle, 14 but also inflammatory proteins9,10,15,16 This finding is consistent with those reported in previous studies.6,7
GO and KEGG enrichment analyses also confirmed that the treatment of renal anemia with DBD involved lipopolysaccharide reactions, metal ion reactions, nutritional reactions, reactive oxygen free radical reactions, reactive oxygen metabolic processes, and oxidative stress reactions. The core inflammatory pathways of DBD in renal anemia therapy included the RAGE, IL-17, TNF, P13K-Akt, MAPK, and HIF signaling pathways; Th17 cell differentiation; and other inflammatory reactions or pathways.
CKD patients experience chronic inflammation because of the gradual decrease in renal function, and the incidence of malnutrition-inflammation complex syndrome is higher in patients with end-stage renal disease. 17 This persistent inflammatory state can cause inflammatory anemia by preventing proliferation of myeloid precursor cells, activating macrophages to phagocytose red blood cells, shortening the lifespan of red blood cells, and inducing synthesis of hepcidin. 18 Among inflammatory proteins, chemokines, such as CXCL8 and IL-1β, could attract leukocytes, and IL-1β could reduce the number of red blood cell precursors and shorten erythrocyte lifespan. 4 In most acute infections, the longer life span of red blood cells can buffer the reduction in red blood cell numbers caused by infection. However, in a chronic inflammatory state, the erythrocyte count is usually reduced to a stable state of anemia, in which the destruction of red blood cells matches the reduction in red blood cell production. 19 In addition, the increase in white blood cells in the bone marrow can induce an increase in the number of myeloid precursor cells, and the ratio of myeloid precursors to erythroid precursors can be greater than 4:1. TNFα mediates this bone marrow remodeling process, 20 and the activating transcription factor PU.1 promotes bone marrow production and lymphogenesis at the expense of erythrocyte production. Furthermore, the ability of erythroid burst-forming units to produce differentiated erythroid cells is inhibited by inflammatory cytokines. 21
Inflammation significantly increases hepcidin synthesis, and hepcidin inhibits iron absorption in the intestines and the release of iron from the mononuclear phagocyte system. This results in an imbalance in iron metabolism in the body as well as hypoglycemia, which may lead to chronic inflammatory anemia due to iron loss. 22 IL-6 may increase hepcidin synthesis in hepatocytes through the JAK2-STAT3 pathway. 23 Akchurin et al. found that IL-6 induced anemia in adolescents with CKD through mechanisms such as hypoferremia, aggravation of renal fibrosis, and changes in the erythropoietin (EPO) axis. 23 STAT1 and STAT3 have opposing functions. STAT1 inhibits cell proliferation and promotes apoptosis, whereas STAT3 promotes cell survival and proliferation. The present study suggests that the core mechanisms of DBD in the treatment of renal anemia involve STAT1, although specific effects need to be examined further. In addition, PTGS2 encodes cyclooxygenase-2 (COX-2). COX-2 can be highly induced in various cells by pro-inflammatory cytokines and participate in inflammatory response. 24 VEGFA is a known pro-angiogenic factor 25 ; its expression is reduced in the bone marrow of patients with severe aplastic anemia, and it is related to bone marrow hematopoietic function. 26 The core targets involved in the treatment of renal anemia with DBD include PTGS2 and VEGFA, which may be related to the regulation of inflammation or bone marrow hematopoietic function.
Inflammation may also inhibit production of EPO in renal tubular cells and destroy the response of erythrocyte precursors to EPO. The demand for exogenous EPO in patients with CKD increases significantly. Consistent with this trend, EPO levels are reduced in systemic inflammatory diseases, such as cancer and juvenile chronic arthritis. Our results revealed that the core components of DBD affected the HIF-1 pathway. HIF-1 is a heterodimer consisting of HIF-1α and HIF-1β, 27 and a low-oxygen environment can induce accumulation of HIF-1α. Increased HIF-1 can bind to the enhancer located at the 3′ end of the EPO gene to induce EPO transcription. 28 EPO protein binds to specific receptors to promote the synthesis of red blood cells in the bone marrow, thereby improving anemia.
Chronic inflammation in patients with CKD consumes qi and damages blood. A large dose of Astragalus in DBD can quickly supply invisible qi, which then transforms into tangible blood. While replenishing qi and producing blood, Astragalus may indirectly or directly improve chronic inflammation in patients with CKD. 29 Our results also demonstrated that the core proteins and pathways related to DBD treatment of renal anemia involved inflammatory proteins and pathways, suggesting that DBD treats renal anemia by regulating the inflammatory state of CKD patients.
In summary, this study indicates that the active ingredients in DBD may improve renal anemia by regulating inflammatory proteins and pathways by creating a “drug-component-target-disease” network composed of various components, diverse targets, and multiple pathways. Further in-depth studies are needed to confirm this mechanism.
Limitations
In this study, various databases were used to mine the core components of DBD and the core targets and pathways for the treatment of renal anemia. However, further verification with in-vivo or in-vitro experiments was not performed. Moreover, DBD is a compound preparation, and its active ingredients are constantly updated. If these active ingredients are not included in the database, some potential mechanisms will not be identified.
Conclusions
DBD may have a protective effect against renal anemia by improving inflammation. Quercetin, as the main active ingredient of DBD, may modulate multiple inflammatory proteins and inflammatory pathways.
Supplemental Material
sj-xlsx-1-npx-10.1177_1934578X221093905 - Supplemental material for Exploring Mechanisms by Which Danggui Buxue Decoction Regulates Inflammation and Improves Renal Anemia Based on Network Pharmacology
Supplemental material, sj-xlsx-1-npx-10.1177_1934578X221093905 for Exploring Mechanisms by Which Danggui Buxue Decoction Regulates Inflammation and Improves Renal Anemia Based on Network Pharmacology by Can Tu, Guirui Huang, Chuang Li, Li Cheng, Yonglong Min, Hongbo Li, Dongdong Mao and Fei Xiong in Natural Product Communications
Supplemental Material
sj-xlsx-2-npx-10.1177_1934578X221093905 - Supplemental material for Exploring Mechanisms by Which Danggui Buxue Decoction Regulates Inflammation and Improves Renal Anemia Based on Network Pharmacology
Supplemental material, sj-xlsx-2-npx-10.1177_1934578X221093905 for Exploring Mechanisms by Which Danggui Buxue Decoction Regulates Inflammation and Improves Renal Anemia Based on Network Pharmacology by Can Tu, Guirui Huang, Chuang Li, Li Cheng, Yonglong Min, Hongbo Li, Dongdong Mao and Fei Xiong in Natural Product Communications
Footnotes
Author Contributions
CT and FX conceived and designed the study. GRH and CL analyzed and interpreted the network pharmacology. LC, YLM, HBL, and DDM provided additional guidance for the analysis of this study. All authors critically revised the manuscript for important intellectual content and approved the final version for publication.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Project of Science and Technology Department of Guangdong Province (grant number 2016A020226042).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
