Abstract
The aim of this study was to analyze the content of hypnotic components in the essential oil from Citrus aurantium flowers (EDD), extracted by different methods, and to characterize its sedative-hypnotic effects. The sedative-hypnotic capacity of EDD was evaluated using pentobarbital-induced sleeping assays, locomotor activity tests and GABAA receptor antagonists. The results showed that EDD extracted by steam and water distillation (SWD), hydrodistillation (HD), and ultrasound-assisted hydrodistillation (UHD) had as their main components linalool, linalyl acetate, and limonene, comprising more than 55% of the total peak area. Compared with EDD extracted by HD and UHD, the total content of linalool and linalyl acetate in EDD obtained by SWD was highest, whereas the content of limonene in EDD extracted by the 3 different methods was not different. Oral and intraperitoneal administration of EDD resulted in reduced sleep latency and increased sleep duration of mice, as well as reduced locomotor activity, which was proven by decreases in the total distance travelled, average velocity, number of activities, and central distance. Interestingly, intraperitoneal injection of EDD had better sedative and hypnotic effects than oral ingestion. In vitro assays using SH-SY5Y cells showed that EDD dose-dependently increased Cl− influx, which could be blocked by the GABAA receptor antagonists, picrotoxin, bicuculline, and flumazenil, suggesting that EDD promoted sedative-hypnotic activity by potentiating GABAA receptor-mediated Cl− current responses. Altogether, these results suggest that the important hypnotic-sedative activity of EDD appears to be due to the effects of limonene, and particularly the high contents of linalool and linalyl acetate, which were effectively extracted by SWD.
Sleep, which accounts for almost 30% of human life, has a remarkable impact on repairing and maintaining bodily functions. 1 The quantity and quality of sleep greatly contributes to perception, memory, consciousness, mood stability, and immunity. 2 However, insomnia is a prevalent health problem in clinical practice affected by several perpetuating, precipitating and predisposing factors, and has become a frequent complaint among adults in modern industrial societies. 3 It is generally characterized by interrupted sleep, sleep loss, early waking and delayed sleep onset caused by mood-, anxiety-, and substance-related disorders, leading to an increased risk of mental and physical health hazards and interference with normal life and work. 4 The insomnia prevalence rates were 22.2% in men and 34.2% in women in 2018. 5
Current patients often receive drug-based therapy to relieve insomnia. Pharmaceutical treatment primarily includes benzodiazepines, melatonin, antipsychotics, and antihistamines. However, these hypnotics are usually associated with central nervous system dysfunction, and liver and kidney damage, and not recommended for insomnia treatment according to the European guidelines for the diagnosis and treatment of insomnia. 6 In particular, rebound insomnia, physical dependence and addiction have limited their appropriate use. Compared with hypnotics, complementary and alternative natural ingredients for promoting sleep have gained gradual global acceptance because of their safety and efficiency over the past 2 decades. 7
Citrus aurantium L. var. daidai Tanaka, family Rutaceae, grows in the Jiangsu, Zhejiang, Guangdong, Hunan, and Guizhou provinces in southeast China. Their fruits become ripe in autumn and are edible, and their flowers, with distinctive fragrance, are used as medicinal tea, both of which are rich in essential oil. Recent research has focused on biofunctional properties of the essential oil of C. aurantium because it does not affect either maternal reproduction or cause teratogenicity in rats. 8 Essential oils from flowers of C. aurantium var. daidai (EDD) induced endothelium- and smooth muscle-dependent vasodilator effects by modulation of the NO-sGC and RyR signaling pathways. 9 Inhalation of C. aurantium essential oil helps relieve stress and modulate the endocrine system, resulting in improved menopausal symptoms, blood pressure reduction, and increased sexual desire in postmenopausal women. 10 Essential oil from C. aurantium displayed moderate to strong antimicrobial properties and excellent DPPH scavenging properties. 11
To the best of our knowledge, no studies have compared the content of hypnotic components in EDD isolated by different extraction methods. In this study, EDD was obtained by separate steam and water distillation (SWD), hydrodistillation (HD) and ultrasound-assisted hydrodistillation (UHD), and the chemical composition and content of the oils were compared by GC-MS analysis. Additionally, the sedative-hypnotic effects of EDD obtained under optimized conditions were assessed using pentobarbital-induced sleeping assays and locomotor activity tests in mice. To define further the mechanism of EDD on γ-aminobutyric acid-A (GABAA) receptor function, pharmacological tools (eg, picrotoxin, bicuculline and flumazenil) were used, and the intracellular Cl− concentration was evaluated in cultured SH-SY5Y human neuroblastoma cells.
Materials and Methods
Materials
Flowers of Citrus aurantium var. daidai were obtained from Lianyuan Kanglu Biological Technology Co., Ltd. (Loudi, Hunan, China), carboxymethyl cellulose sodium (CMC-Na) and pentobarbital from Sigma-Aldrich (St. Louis, MO, USA), diazepam (purity 99%) from Lisheng Pharmaceutical Co., Ltd. (Tianjin, China), N-(ethoxycarbonylmethyl)-6-methoxy-quinolinium bromide (MQAE) and 2-(4-(2-hydroxyethyl)piperazin-1-yl)ethanesulfonic acid (HEPES) from Beyotime (Shanghai, China), fetal calf serum (FBS), and DMEM medium from Gibco (Carlsbad, CA, USA), and picrotoxin (purity 94%), bicuculline (purity 98%) and flumazenil (purity 99%) from Sinopharm Chemical Reagent Co., Ltd. (Shanghai, China). All other chemicals and reagents were of the highest grade available.
Essential Oil Extraction
Extraction by SWD: Flowers were dried and crushed, and the essential oil extracted by steam and water distillation, as described by Memarzadeha et al. 12 Three hundred g was supported above 1 L boiling water on a perforated grid. After steam and water distillation for 2 h, the essential oil was collected and dried over a small amount of Na2SO4 to absorb the remaining water and stored without light exposure at −80 °C before analysis.
Extraction by HD: The essential oil was extracted from the dried, crushed flowers using an all-glass Clevenger apparatus, as described by Hashemi et al. 13 One L of distilled water and 300 g sample were added to the 2 L round flask and heated using a temperature controlled electric mantle until essential oil was obtained. This was collected, the water was removed and the sample stored without light exposure at −80 °C.
Extraction by UHD: The dried, crushed flowers (300 g) were placed in a 2 L round flask containing 500 mL of distilled water and sealed with a stopper, and then EDD was extracted using ultrasound. The ultrasonic wave energy input by an ultrasonic transducer had a fixed power of 120 W for 30 min. Subsequently, another 500 mL of distilled water was added to the sample and hydrodistilled for 2 h. The essential oil obtained was dried with anhydrous sodium sulfate, and stored without light exposure at −80 °C.
Essential Oil Analysis
A GC-MS instrument (7890A, Agilent Technologies, Santa Clara, CA, USA) equipped with a mass selective detector operating in the electron impact mode (70 eV) was used to study the compositions of EDD. The GC part (5975C, Agilent Technologies, Santa Clara, CA, USA) was equipped with a HP-5MS capillary column (30 m × 0.25 mm i.d. × 0.25 µm, film thickness). The injector temperature of the instrument was set at 280 °C and MS scanning was from 50 to 500 amu. Temperature-programming of the oven included an initial hold at 70 °C for 1 minute, an increase from 70 °C to 150 °C (3 °C/min), maintained at 150 °C for 2 minutes, an increase from 150 °C to 220 °C (2 °C/min), a subsequent increase from 220 °C to 236 °C (1 °C/min) and an increase from 236 °C to 240 °C (2 °C/min), maintained at 240 °C for 13 minutes. Alkanes (C5-C24) were used as reference points in the calculation of RI. The sample was injected in split mode (1:10) with an injection volume of 1 µL. The EDD compounds were identified based on comparison of the RI with a database (Wiley/NIST library) and literature data.
Experimental Animals
ICR mice (25-30 g, male and female) were purchased from Tianqin Biotechnology Co., Ltd. (Changsha, Hunan, China) and used at 5-6 weeks of age. All procedures involving mice were conducted in accordance with the animal care and use guidelines of the Science and Technology Department of Hunan Province (permission number: SCXK 2016-0002). The animals were housed in individual cages in a room with a temperature of 23 ± 0.5 °C, a 12 h light/dark cycle and with ad libitum access to food and water. The mice were fasted for approximately 8 hours before the analysis.
Determination of Pentobarbital-Induced Hypnosis
Sixty mice were randomly divided into 6 groups with 10 mice in each group. The mice were administered intraperitoneally (i.p.) with different concentration of sodium pentobarbital (20, 23, 25, 27, 30, and 35 mg/kg) dissolved in 0.9% physiological saline to induce sleep, followed by being placed in individual cages; the number of sleeping mice, sleep latency and sleep duration was recorded. Sleep duration was recorded as the time interval from loss of righting reflex to recovery.
Effect of EDD on Pentobarbital-Induced Sleeping Tests
Ninety mice were randomly divided into 9 groups (half males and half females in each group) with 10 mice per group. The groups were comprised of: control group Ⅰ (oral saline), control group Ⅱ (i.p. saline), oral EDD groups (40, 80, and 120 mg/kg), i.p. injection of EDD groups (40, 80, and 120 mg/kg) and diazepam group (0.65 mg/kg, oral). These EDD groups were either orally or intraperitoneally administered with the corresponding drug solution dissolved in 0.5% CMC-Na saline solution for 14 days. Mice in each group were i.p. injected with 30 mg/kg pentobarbital 30 min after the last drug administration. Then the sleep latency, sleep duration, and number of sleeping mice were recorded. Sleep latency was recorded from the time of pentobarbital injection to the time of sleep onset.
Measurement of Locomotor Activity
Ninety mice were randomly divided into 9 groups (half males and half females in each group) with 10 mice per group. Sample grouping was the same as in the pentobarbital-induced sleeping test. The groups were either orally or intraperitoneally administered different doses of EDD (40, 80, and 120 mg/kg) dissolved in 0.5% CMC-Na saline solution for 14 days. The spontaneous locomotor activity of the mice was assessed at the end of the diazepam and EDD treatment using an animal behavior analysis system with a spontaneous activity video analysis system (Jiliang, Shanghai, China) for 5 min. The spontaneous locomotor activity was analyzed by recording the total distance travelled, average velocity, number of activities, time immobile, mobility time, central distance, central time, peripheral distance and peripheral time.
Measurement of Intracellular Cl− Influx
The human neuroblastoma SH-SY5Y cells were purchased from the Chinese Academy of Sciences (Shanghai, China) and cultured in an incubator containing 5% CO2 at 80% humidity and 37 °C. DMEM medium supplemented with 10% FBS was used for the cell culture. The effects of the EDD, EDD + bicuculline, EDD + picrotoxin, and EDD + flumazenil on intracellular Cl− influx ([Cl−]i) in SH-SY5Y cells were analyzed using the fluorescent Cl− sensitive indicator MQAE. The experiments were performed as previously described by de la Peña et al. 14 Cells were re-suspended at a concentration of 4 × 105 cells/mL in Hank’s buffer after washing twice with Hank’s buffer. Then MQAE was added to stain the cells at a final concentration of 5 mM at 37 °C for 2 h. Fluorescence was monitored using a multifunctional microplate reader (TECAN, Infinite 200 PRO, Grodig, Austria) at an excitation/emission wavelength of 365 nm/450 nm. Contents of influxed Cl− ion is presented as relative fluorescence F/F0, where F is the fluorescence as a function of time, and F0 was evaluated by incubating the cells with Cl−-free (KNO3) solution containing 10 mM nigericin and 10 mM tributyltin in a separate experiment. All fluorescence values were corrected for background fluorescence, which was tested using 20 mM HEPES buffered containing 150 mM potassium thiocyanate and 5 μM valinomycin to maximally quench the MQAE ion-selective signal at the end of each experiment.
Statistical Analysis
Data were analyzed using SPSS v.23.0 and are represented as the mean standard deviation (SD). Results were analyzed using a one-way analysis of variance (ANOVA), followed by Tukey’s Post hoc test. P < 0.05 and P < 0.01 were considered significant and extremely significant in all cases, respectively. The data matrix of the volatile components detected by GC-MS were then submitted to principal component analysis (PCA) in order to determine the volatile compounds able to differentiate the samples by different extraction methods.
Results and Discussion
Comparison of the Yields and Composition of EDD Obtained by SWD, HD, and UHD
The yield of EDD extracted by UHD was higher than by SWD (P < 0.01), while the yield of EDD isolated by SWD and HD were not significantly difference (Figure 1). Ultrasound-assisted hydrodistillation offered obviously higher yields of volatile oil from citrus peel than supercritical fluid extraction, 15 suggesting that ultrasonic assisted extraction is a good method to obtain Citrus essential oil. Herein, we studied the effect of ultrasound-assisted extraction on the yield and composition of EDD.
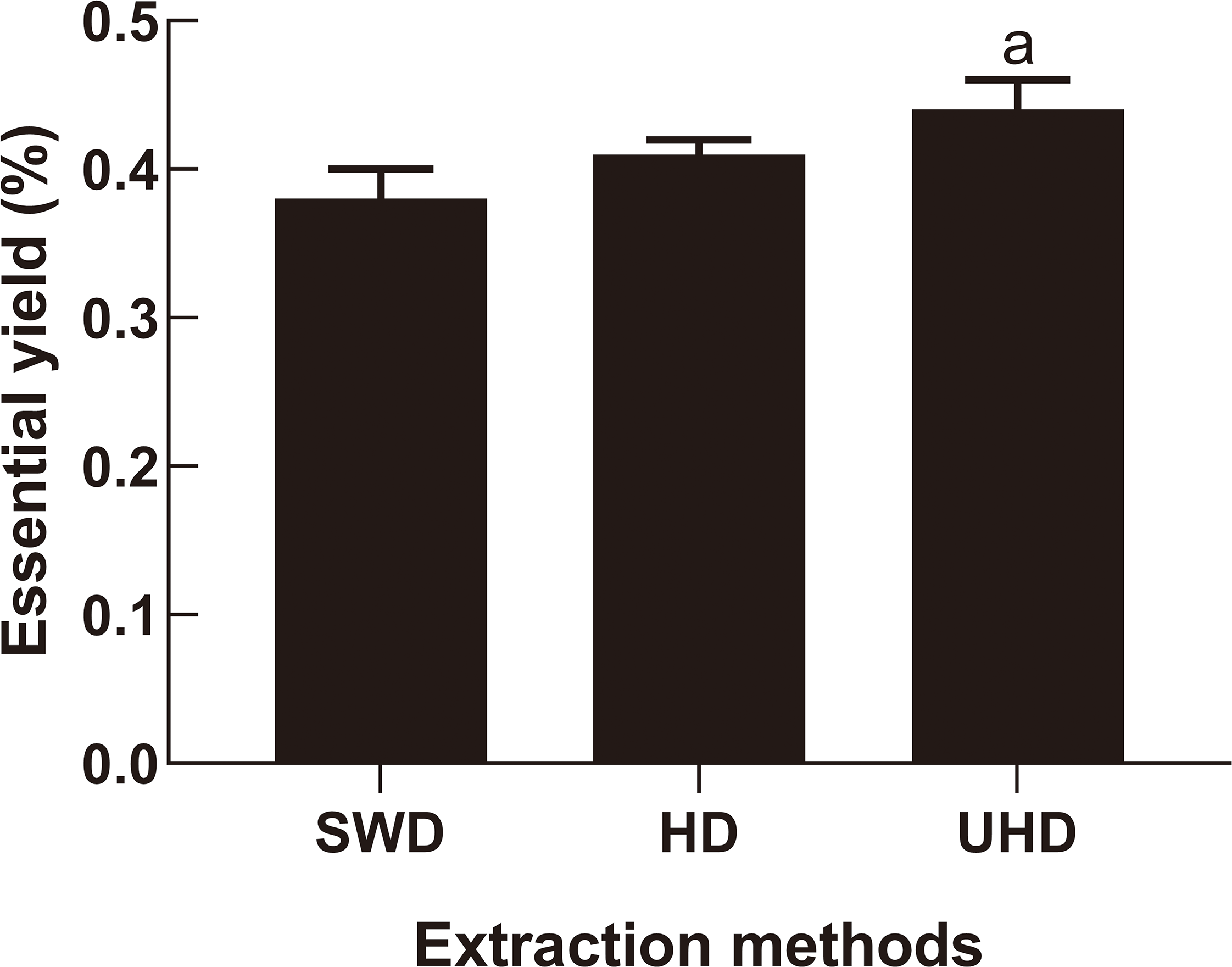
The yields of essential oil from Citrus aurantium flowers extracted by different methods. P < 0.01 vs SWD.
As shown in Table 1, EDD extracted by SWD, HD and UHD had 43, 39, and 35 components, respectively, representing 98.3%, 97.8%, and 99.7% of the corresponding oils, respectively. Oxygenated monoterpenes were the main components of EDD, followed by monoterpene hydrocarbons. The contents of sesquiterpene hydrocarbons and oxygenated sesquiterpenes were relatively lower. Compared to other extraction methods, EDD extracted by UHD contained more monoterpene hydrocarbons, for example, β-myrcene (1.27% in UHD oil >1.16% in HD oil >0.73% in SWD oil), β-ocimene (4.29% > 2.01% > 1.97%), and terpinolene (0.37% > 0.2% > 0.07%). On the contrary, EDD extracted by SWD showed higher content of sesquiterpene hydrocarbons and oxygenated sesquiterpenes than other extraction methods, for example, caryophyllene (3.93% in SWD oil >3.37% in UHD oil >2.72% in HD oil), E-nerolidol (1.67% > 1.29% > 1.06%) and cis, cis, cis-1,1,4,8-tetramethyl-4,7,10-cycloundecatriene (0.58% > 0.45% > 0.39%). Compared with HD extraction, UHD had no obvious effect on monoterpene hydrocarbon content of EDD, but increased oxygenated monoterpene and sesquiterpene hydrocarbon content. This result is different to the report that ultrasound-assisted extraction reduced the monoterpene content of the essential oil from Carum carvi L. seeds compared with hydrodistillation, 16 suggesting that ultrasound-assisted extraction has different effects on the components of essential oil from different plants.
Chemical Compositions of the Essential Oils From Citrus aurantium Flowers by SWD, WD and UHD Analyzed by GC-MS.

Retention index of compounds on HP-5MS.
Retention index of compounds in reference.
MS comparison using NIST Mass Spectral Library and Adams (2007), 46 RI retention index in agreement with the literature value. ND, not detected. Data are mean ± SE (n = 3).
Importantly, the total content of linalool and linalyl acetate in SWD oil (54.1%) was higher than that in HD oil (48.3%) and UHD oil (45.2%) (P > 0.01). Limonene is also one of the main components of EDD, and its content was not significantly different in EDD extracted by the 3 extraction methods (12.8% in SWD oil, 12.7% in UHD oil, 12.7% in HD oil). These results implied that SWD afforded considerable amounts of linalool and linalyl acetate. For steam and water distillation, Citrus aurantium flowers cannot be in direct contact with boiling water. The vapor is passed through the flowers before being condensed. Hence, the components of SWD oil are less hydrolyzed than in the case of HD. 47 This may be the reason why the total content of linalool and linalyl acetate in SWD oil was higher than that in HD oil.
Similar findings have been reported by Değirmenci et al., and Sarrou et al. who reported that the essential oil of C. aurantium flowers consists mainly of linalool, limonene, linalyl acetate and β-pinene. 48,49 However, Hsouna et al. and Salma et al. reported that the main component of the essential oil of C. aurantium flowers from Tunisia and Morocco can be characterized by the dominance of limonene (27.5% to 40.81%). 50,51 These differences in the essential oil composition might arise from several environmental (geographical, climatic, soil), gene and extraction method differences.
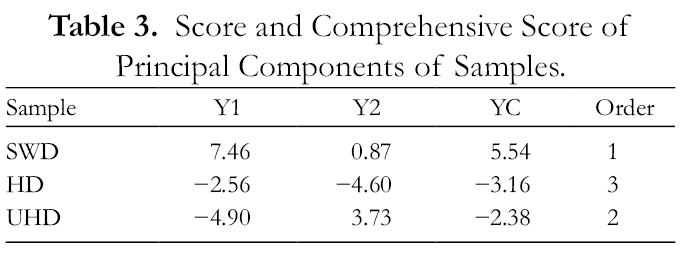
For a better visualization of data, principal component analysis (PCA) was applied to identify patterns of correlation with single composition variables involved in the discrimination among samples obtained by different extraction methods. As shown in Table 2 and Figure 2, separation along the first principal component (PC1) explained 70.7% of the total variation in the sample set and accounted for the positive contributions from α-pinene, linalyl acetate, sesquirosefuran and phytone and negative contributions from β-myrcene, linalool, myrtenal, neryl acetate, and geranyl acetate. Separation along the second principal component (PC2) accounted for 29.3% of the total variance and for the negative contributions from trans-p-mentha-2,8-dien-1-ol and hexadecanoic acid. The variance explained by the PCA was 100% for the 2 principal components. The principal component scores “Y1, Y2” and the comprehensive principal component score “YC” were calculated as YC = 0.707Y1 +0.293Y2. As shown in Table 3, the order referred to YC was obtained: SWD oil >UHD oil >HD oil. Based on the analysis of linalool, linalyl acetate and limonene content and evaluation of the comprehensive EDD score, this study proved that SWD is an effective method of extracting EDD with hypnotic activity. Depending on the interest in a certain kind of functional components, specific extraction methods could be utilized according to different target compositions. 52

PCA plots of volatile compositions of essential oil from Citrus aurantium flowers extracted by different methods.
Principal Components Data Based on Predominant Constituents in Essential Oils From Citrus Aurantium Flowers.

Score and Comprehensive Score of Principal Components of Samples.
EDD Potentiated Pentobarbital-Induced Sleep
To evaluate the hypnotic activity of EDD isolated by SWD, we first investigated the hypnotic dosage of pentobarbital on mice. The range of pentobarbital doses from 20 to 30 mg/kg, i.p. did not make the mice sleep, while 35 mg/kg pentobarbital (i.p.) made 2 mice sleep (sleep duration 12.8 ± 3.5 m). Because 30 mg/kg was the maximum dose of pentobarbital that did not cause any of the mice to go to sleep, it was used as a sub-hypnotic dose (rate of sleep onset <10%).
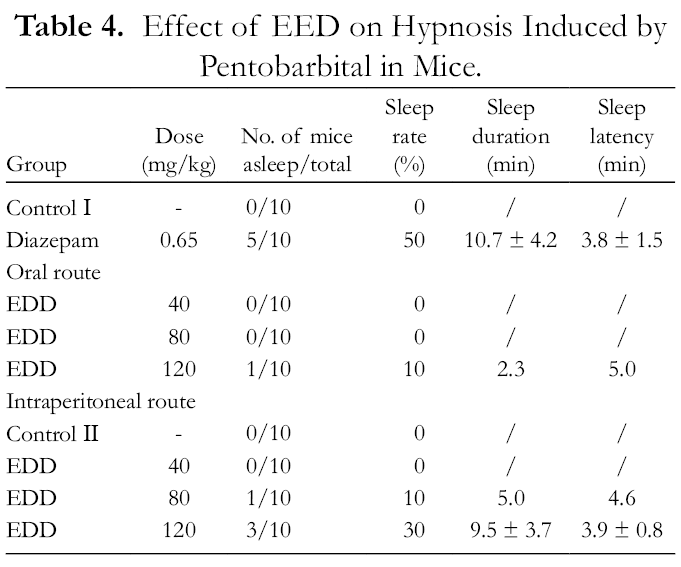
We examined the sedative-hypnotic activities of EDD using a pentobarbital-induced sleep test in mice and the results are shown in Table 4. Compared with the control group (30 mg/kg pentobarbital, i.p.), oral administration of 120 mg/kg EDD induced one mouse to fall asleep, while oral administration of 40 and 80 mg/kg EDD showed no obvious effectiveness in improving pentobarbital-induced sleep. EDD at 120 mg/kg (i.p.) caused 3 mice to sleep with a sleep duration and sleep latency of 9.5 ± 3.7 min and 3.9 ± 0.8 min, respectively, and the effect was similar to that of diazepam (0.65 mg/kg, oral). Compared with oral administration, i.p. injection of EDD is more effective for promoting sleep in mice.
Effect of EED on Hypnosis Induced by Pentobarbital in Mice.
Previous studies have also shown that some plants in the Rutaceous family possess sedative/hypnotic activities. Treatment with essential oil from C. aurantium peel increased the time spent in the open arms of the elevated plus maze and the sleeping time induced by barbiturates. 53 Aqueous extract of C. aurantium flowers (i.p.) produced a significant increase in the sleeping time and time spent on open arm entries, as well as a decrease in anxiety levels in mice. 54 Studies have reported that linalool and linalyl acetate apply directly to the synaptosomes, resulting in the induction of calmness and sleep enhancement. 55 Limonene was also reported to present sedative effects and produce a potentiation of the pentobarbital-induced sleeping time but does not increase the onset of sleep, which may be due to the activation of adenosine A2A receptors and an increase in cytosolic calcium concentration. 56 In this study, the total content of linalool and linalyl acetate in EDD extracted by SWD exceeded 50% of the total oil, and the content of limonene in EDD extracted by the 3 extraction methods exceeded 10%. Therefore, the 3 components of EDD may be the main reason why EDD possesses sleep-promoting functions.
EDD Reduced Locomotor Activity
The sedative activity of EDD was evaluated by a locomotor activity test. As seen in Figure 3, compared with the control group, oral EDD significantly decreased the total distance travelled, average velocity, number of activities, central distance, time immobile and peripheral time (P < 0.01 or P < 0.05), but did not obviously change the mobility time, peripheral distance or central time. Similarly, EDD (i.p.) significantly decreased total distance travelled, average velocity, number of activities, central distance, time immobile, mobility time, and peripheral distance compared with the control group (P < 0.01), while central time and peripheral time were not significantly different from those of the control group (Figure 4). Remarkably, i.p. administration of EDD was more effective than oral administration in reducing locomotor activity, but the dose effect was not obvious. This result may be the reason why the absolute bioavailability of orally administered EDDs is better than that of intraperitoneally administered EDDs. These results suggested that locomotor activity was effectively reduced after oral and i.p. administration of EDD, which is similar to that of the positive control, diazepam.
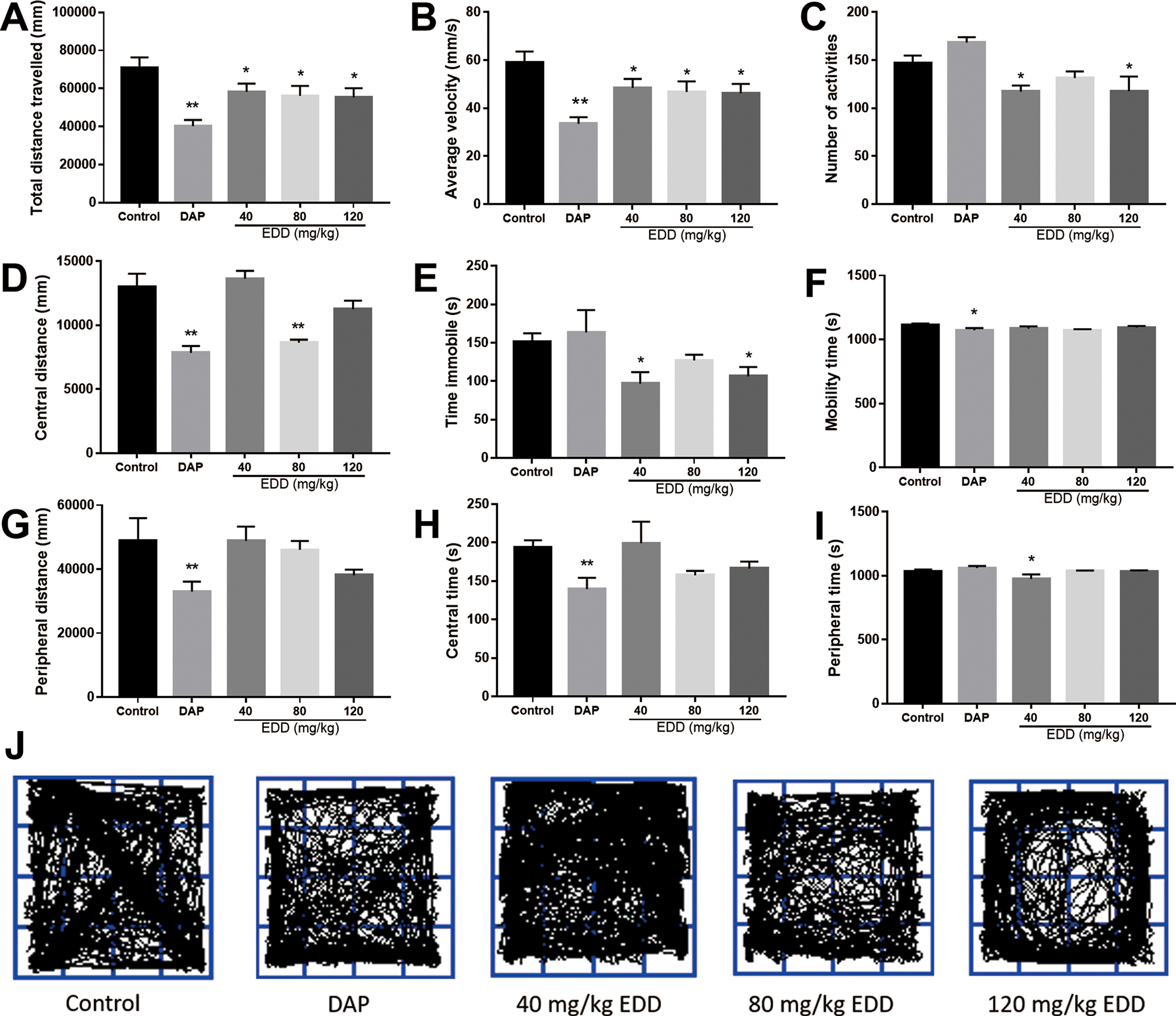
Locomotor activity of ICR mice after oral administration of EDD. After the groups were orally administered with different doses of EDD (40, 80 and 120 mg/kg) dissolved in 0.5% CMC-Na saline solution for 14 days, the indexes of locomotor activity were assessed in a test of 5 minutes. (A) Total distance travelled, (B) average velocity, (C) number of activities, (D) central distance, (E) time immobile (I) peripheral time, (F) mobility time, (H) central time, (G) peripheral distance, and (J) representative tracks of mice in each group, drawn by an animal behavior analysis system. All data are represented as the mean ± SD (n = 10), *P < 0.05 and **P < 0.01 vs control group.

Locomotor activity of ICR mice after intraperitoneal administration of EDD. After the groups were either orally or intraperitoneally administered with different doses of EDD (40, 80, and 120 mg/kg) dissolved in 0.5% CMC-Na saline solution for 14 days, the indexes of locomotor activity were assessed in a test of 5 minutes. (A) Total distance travelled, (B) average velocity, (C) number of activities, (D) central distance, (E) time immobile, (I) peripheral time, (F) mobility time, (H) central time, (G) peripheral distance, and (J) representative tracks of mice in each group, drawn by an animal behavior analysis system. All data are represented as the mean ± SD (n = 10), *P < 0.05 and **P < 0.01 vs control group.
Recent research reports that some active components of C. aurantium could elevate or inhibit locomotion activity. For example, long-term 3% naringenin supplementation markedly decreased muscle diacylglycerol content, attenuated muscle loss, and increased locomotor activity monitored by infrared beam breaks in obese ovariectomized mice. 57 In contrast, according to the report by Arbo et al., 58 acute oral administration of extracts (300 to 5000 mg/kg) from C. aurantium leaves and fruits containing a high content of p-synephrine remarkably reduced locomotor activity in mice. Essential oil is widely used to promote comfort, relax the mind and for hypnosis purposes, for example, essential oil from Juniperus virginiana L. (EJV). Cedrol, a main component of EJV, significantly increased locomotor activity in the light-dark box and elevated plus maze. Interestingly, another EJV ingredient, cedrane, obviously inhibited the locomotor activity and increased anxiety-related behaviors. 59 These findings indicate that the ingredients in essential oil can either positively or negatively regulate locomotor activity. Therefore, according to the purpose of the application, the choice of method to extract effectively the active ingredients is very important.
Effect of EDD on Intracellular Cl− Influx
To demonstrate the effect of EDD on GABAA receptors, the intracellular Cl− concentration was tested using the Cl− fluorescence probe MQAE. As shown in Figure 5(A), EDD dose-dependently increased intracellular Cl− influx in SH-SY5Y cells (P < 0.01). Furthermore, 2.5 and 5 μL/mL EDD mediated the highest Cl− concentrations at 100 s and 150 s, respectively, which was weakened over time. In contrast, 3 GABAA receptor antagonists, picrotoxin, bicuculline and flumazenil, inhibited the increase in Cl− influx from 50 s to 300 s caused by 5 μL/mL EDD (Figure 5(B)).

Differential effects of EDD and GABAA receptor antagonists on Cl− influx in SH-SY5Y cells. Cells were treated with EDD and/or GABAA receptor antagonists (10 µM picrotoxin, bicuculline or flumazenil), then intracellular Cl− concentration was tested using a Cl− fluorescence probe MQAE. The results are presented as mean ± SD with n = 6. (A) a, P < 0.01 vs control cells; b, P < 0.05 vs pentobarbital group. (B) a, P < 0.01 vs 5 µL/mL EDD + picrotoxin; b, P < 0.01 vs 5 µL/mL EDD + bicuculline; c, P < 0.01 vs 5 µL/mL EDD + flumazenil.
The GABAA receptor is a GABA/benzodiazepine receptor-Cl− channel complex. 60 GABAA-receptor agonists, located in the tuberomammillary nucleus cortical EEG delta and spindle, are used in sedation and anesthesia practice. 61 However, with increasing clinical application of GABAA receptor agonists, a growing side effect profile has been found. Similar to our results, diazepam exhibits sedative activity and attenuates seizure behavior, but induces hypotension and dizziness. 62 In contrast, GABAA antagonists, including propofol, etomidate, picrotoxin/picrotoxinin, flumazenil, sevoflurane, pentobarbital, isoflurane, and bicuculline, prevent GABA from binding to GABAA receptors. 63 The competitive antagonist bicuculline acts as a negative allosteric modulator of GABAA receptor function by binding to orthostatic sites. 64 Picrotoxin acts as a noncompetitive antagonist on GABAA receptors, which blocks the GABA-gated Cl− channel by binding to the picrotoxin site. 65 Our study showed that EDD could affect the functions of GABAA receptors to induce Cl− channel opening via benzodiazepine, orthostatic, and picrotoxin binding sites.
Conclusion
The present study suggests that limonene, linalool and linalyl acetate are the major compounds of EDD effectively extracted by SWD that contribute to its sedative-hypnotic effects. The underlying mechanism of EDD might be to enhance GABAA-mediated chloride flux via benzodiazepine, orthosteric, and picrotoxin binding sites. Our results suggest that EDD could be used as a natural sleep aid to relieve insomnia. Further study is necessary to determine which has a stronger hypnotic and sedative activity between a single component and a combination of multiple components of EDD. It is also necessary to compare the hypnotic and sedative effects of limonene, linalool and linalyl acetate at the same concentration.
Footnotes
Statement of Human and Animal Rights
All of the experimental procedures involving animals were conducted in accordance with the Guidelines for the Care and Use of Laboratory Animals and the related ethical regulations of Hunan Agricultural University approved by the Administration Committee of Experimental Animals, Hunan Province, China.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: All authors acknowledge the National Key Research and Development Project (2020YFD1001100), Key Research and Development Program of Hunan Province (2019NK2041), Hunan Agricultural Science and Technology Innovation Fund Project (2019JG01) for its financial support.
