Abstract
Members of the Abies genus (Pinaceae) have been used in traditional medicines in their natural ranges, and essential oils derived from Abies species have found use in aromatic therapy. In this work, the foliar (leaves and twigs) essential oil of Abies concolor have been obtained from mature trees growing in southwestern Idaho and analyzed using gas chromatographic techniques. The essential oils were rich in α-pinene (15.2-20.5%), camphene (7.5-10.2%), β-pinene (24.2-25.6%), δ-3-carene (5.5-6.5%), limonene (5.4-6.9%), and bornyl acetate (14.6-22.1%). The dominant enantiomers for the major terpenoids were (+)-α-pinene, (–)-camphene, (–)-β-pinene, (+)-δ-3-carene, (–)-limonene, and (–)-bornyl acetate. The essential oil compositions are qualitatively similar to other Abies species, but the enantiomeric distributions for α-pinene, β-pinene, and camphene did reveal notable variation between Abies essential oils.
Introduction
Several species of Abies (Pinaceae) have been used in traditional medicine. For example, a paste from leaves and cones of Abies spectabilis (D. Don) Mirb. is used in Nepali traditional medicine to treat bone fractures, 1 leaves of Abies pindrow (Royle ex D. Don) Royle are employed in Ayurvedic medicine for fever, respiratory, and inflammatory conditions, 2 Abies sibirica Ledeb. leaves are used in Siberia to treat vomiting, diarrhea, epilepsy, and as a diuretic, 3 preparations of Abies webbiana (Wall. Ex D. Don) Lindl. are used in Siddha and Ayurveda medical systems to treat a variety of ailments, 4 and northern tribes of Native Americans used the resin of Abies balsamea (L.) Mill. externally as an antiseptic salve and internally to treat colds, consumption, menstrual irregularities, and as a laxative. 5 Abies concolor (Gordon) Lindl. ex Hildebr. has been used by several western North American tribes. The Keres used an infusion of the foliage as a bath for rheumatism, the Paiute and Shoshoni used the resin to treat cuts, wounds, and sores, and a decoction of the resin was taken for respiratory troubles. 6 Essential oils derived from Abies species have found utility in aromatherapy, including Abies alba Mill., A. balsamea, A. concolor, Abies koreana E. H. Wilson, Abies lasiocarpa (Hook.) Nutt., Abies nordmanniana (Steven) Spach, Abies procera Rehder, Abies sachalinensis (F. Schmidt) Mast., A. sibirica, and A. spectabilis. 7
Abies concolor is native to western North America. There are two recognized subspecies, A. concolor subsp. lowiana (Gordon) A. E. Murray, the California white fir, ranges from the southern Cascade Range in Oregon, south through the western slopes of the Sierra Nevada in California, and into Baja California. Abies concolor subsp. concolor, the Rocky Mountain white fir, is found naturally in the Wasatch Range of Utah and southeastern Idaho, the Mogollon Rim of Arizona, the southern Rocky Mountains of Colorado and New Mexico, and the San Francisco Mountains of western New Mexico.8,9 In southwestern Idaho, A. concolor subsp. concolor is planted as an ornamental shade tree. 10 In this work, the essential oil from the leaves and twigs of A. concolor, collected from two individuals growing in suburban Kuna, Idaho, were obtained and analyzed using gas chromatographic techniques.
Results and Discussion
Essential Oil Composition
The foliar essential oils from two different A. concolor trees were obtained as colorless liquids in 0.737% and 0.895% yield. The essential oil compositions of two different samples of A. concolor are presented in Table 1. The major components were α-pinene (20.5%, 15.2%), camphene (7.5%, 10.2%), β-pinene (25.6%, 24.2%), δ-3-carene (6.5%, 5.5%), limonene (6.9%, 5.4%), and bornyl acetate (14.6%, 22.1%). The essential oil compositions of the Kuna A. concolor samples are in qualitative agreement with several essential oils growing in urban locations in Belarus, which showed α-pinene (7.3-13.2%), camphene (12.8-17.6%), β-pinene (28.0-35.9%), δ-3-carene (0.2-1.3%), limonene (5.4-7.2%), and bornyl acetate (17.5-25.4%). 11 Likewise, A. concolor foliar essential oil from the Kaibab Plateau, Arizona, showed α-pinene (21.0-25.8%), camphene (0.9-5.8%), β-pinene (45.6-57.4%), δ-3-carene (0.0-0.2%), and limonene (10.7-13.5%); neither β-phellandrene nor bornyl acetate were reported, however. 12
Chemical Composition of Abies concolor Foliar Essential Oil.
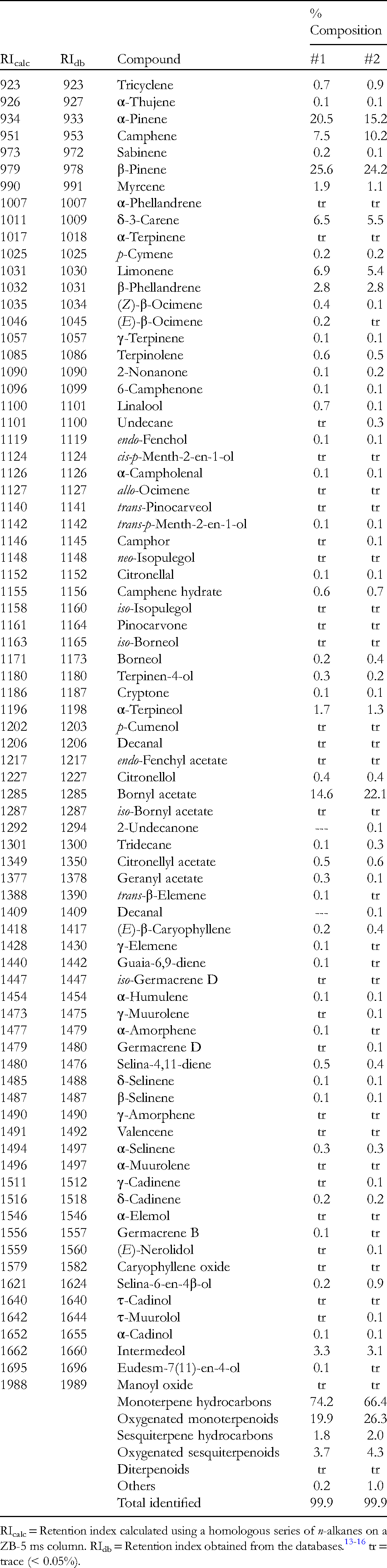
Based on leaf morphology 17 the collected plants in this study are A. concolor subsp. concolor. However, based on geographical location, 18 they may actually be a hybrid of A. concolor and Abies grandis (Douglas ex D.Don) Lindl. A comparison of the concentrations of the major components of A. concolor and A. grandis is summarized in Table 2. The α-pinene concentrations in A. concolor from this study are higher than those found in A. concolor subsp. lowiana from California or A. concolor subsp. concolor from Utah, 19 but comparable to those reported for A. concolor from Arizona. 12 α-Pinene concentration was also low in A. grandis from Idaho. 20 Conversely, β-pinene concentrations were lower in A. concolor in this study compared to A. concolor from Arizona, 12 from California or from Utah, 19 but comparable to A. concolor from Belarus 11 or A. grandis from Idaho. 20 Importantly, reported limonene concentrations are greater than β-phellandrene concentrations in A. concolor 19 whereas β-phellandrene concentrations are greater than limonene concentrations for A. grandis. 20 In this present study, limonene concentrations were slightly greater than β-phellandrene and suggests an intermediate ratio. Likewise, the concentrations of bornyl acetate in this study are intermediate between those of A. concolor (from Arizona, California, or Utah) and that from A. grandis from Idaho. The volatile phytochemistry of A. concolor from southwestern Idaho in this study, then, may suggest these two samples are actually hybrids of A. concolor and A. grandis.
Comparison of Concentration Ranges (%) of Major Components of Abies Essential Oils.
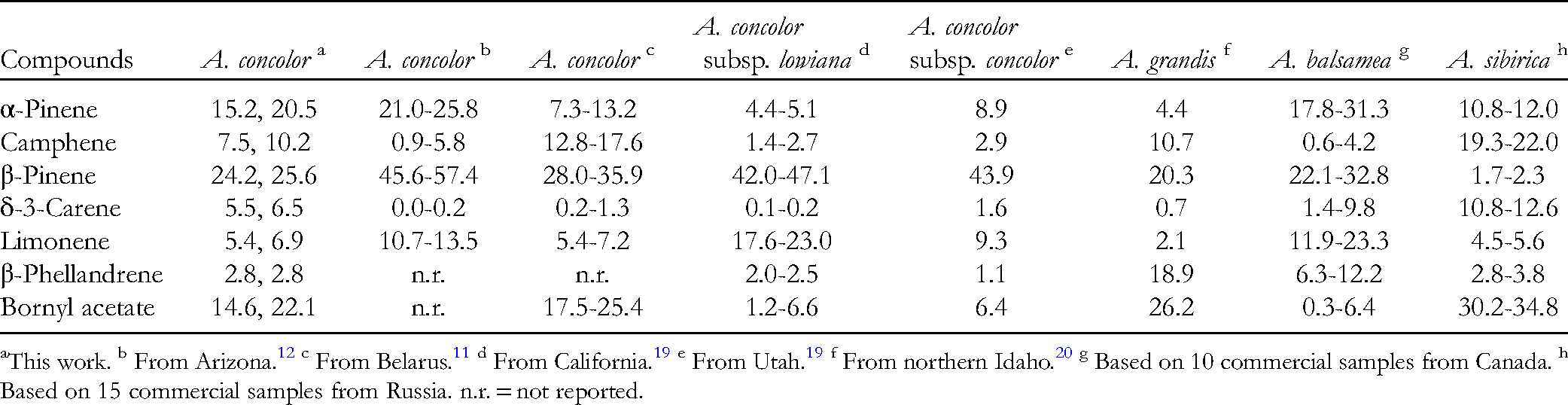
As a comparison, the major components of the leaf essential oils of commercial samples of A. balsamea from Canada and A. sibirica from Russia (unpublished data from samples from the collection of the Aromatic Plant Research Center, Lehi, Utah) are also summarized in Table 2. Based on these data, the essential oil compositions of A. concolor from southwestern Idaho are more similar to those of A. balsamea than to A. sibirica oils.
Enantiomeric Distribution
Chiral gas chromatographic analysis was carried out to determine the enantiomeric distributions of the terpenoid constituents (Table 3). The ( + )-enantiomer was predominant for α-pinene, in contrast to that found in the seed and cone essential oils of A. concolor from Łódź, Poland. 21 (–)-α-Pinene also predominated in the leaf essential oil of A. alba from Poland, 22 the seed essential oil of A. koreana from Poland, 23 and the leaf and shoot essential oils of Abies veitchii Lindl., Abies mariesii Mast., A. sachalinensis from Japan, 24 and A. balsamea from Canada (Table 3). On the other hand, ( + )-α-pinene was the dominant enantiomer in the leaf essential oil of A. sibirica from Austria, 22 A. sibirica from Russia (Table 3), Abies firma Siebold & Zucc. leaf essential oil from Japan, 24 and the leaf essential oil of Abies spectabilis subsp. langtangensis (Silba) Silba from Nepal. 25 Thus, the enantiomeric distribution of α-pinene seems to vary between and within Abies species. (–)-β-Pinene was the dominant enantiomer in A. concolor essential oil from Kuna, as well as essential oils of A. alba, A. sibirica, 22 A. spectabilis, 25 A. koreana, 26 A. sachalinensis, 27 A. balsamea (Table 3), and A. procera. 28 Interestingly, (+)-β-pinene was more abundant than the (–)-enantiomer in the seed and cone essential oils of A. concolor from Poland. 21
Enantiomeric Distribution of Chiral Terpenoid Components in the Essential Oils of Abies Species.

This work. b Commercial samples from Canada. c Commercial samples from Russia.
The (–)-enantiomer of limonene and camphene predominated in A. concolor foliar essential oil in this study as well as in A. concolor seed and cone essential oils, 21 A. balsamea (Table 3), A. alba and A. sibirica leaf essential oils, 22 A. koreana seed essential oil, 23 and A. firma, A. veitchii, A. mareisii, and A. schalinensis leaf essential oils. 24 On the other hand, (+)-camphene was the major enantiomer in A. spectabilis, 25 A. procera, 28 and Russian A. sibirica (Table 3) essential oils. The only enantiomer of δ-3-carene in A. concolor foliar essential oil was (+)-δ-3-carene, consistent with that observed for A. firma, A. mariesii, 24 A. spectabilis subsp. langtangensis, 25 A. procera, 28 A. balsamea and A. sibirica (Table 3) essential oils. Both (–)-borneol and (–)-bornyl acetate were the exclusive enantiomers in A. concolor essential oil, which was also observed in A. procera essential oil 28 as well as A. balsamea and A. sibirica commercial essential oils (Table 3).
Conclusions
The essential oil compositions and enantiomeric distributions of terpenoids for Abies concolor, growing in a suburban environment in southwestern Idaho have been presented. The compositions are qualitatively similar to other Abies essential oils. However, a quantitative comparison of the major components with other Abies concolor and Abies grandis may indicate the southwestern Idaho samples to be hybrids of A. concolor and A. grandis. The enantiomeric distributions for α-pinene, β-pinene, and camphene did reveal variation between Abies species. The similarities between A. concolor and A. balsamea essential oils are notable and account for the similar traditional uses of these two species.
Materials and Methods
Plant Material
Branch tips (leaves and stems) of A. concolor were obtained from two different mature trees growing in Kuna, Idaho (43°30'11″N, 116°25'36″W, 823 m elevation) on 26 July, 2021. The trees were identified by Kathy Swor and William Setzer in the field and verified by Daniel Murphy, Collections Curator, Idaho Botanical Garden, based on leaf morphology. 17 Geographical location, however, suggests that this may be an A. concolor – A. grandis hybrid. 18 A voucher specimen (WNS22143658) has been deposited in the herbarium of the University of Alabama in Huntsville (HALA). The fresh foliar materials (148.8 and 214.0 g, respectively) were hydrodistilled using a Likens-Nickerson apparatus with continuous extraction with dichloromethane for 3 h to give, after evaporation of the solvent, colorless essential oils (1.097 and 1.915 g, respectively).
Gas Chromatography – Mass Spectrometry
The GC-MS analysis of the foliar essential oils of A. concolor samples were performed using a Shimadzu GCMS-QP2010 Ultra Gas Chromatography-Mass Spectrometry instrument (Shimadzu Scientific Instruments, Columbia, MD, USA) fitted with a ZB-5 ms fused silica capillary GC column, (5% Phenyl polymethylsiloxane, 60 m × 0.25 mm i.d. × 0.25 μm film thickness) (Phenomenex, Torrance, CA, USA). Helium (99.9%) was used as the carrier gas at a flow rate of 2.0 mL/min. Mass spectra were recorded at 70 eV, the mass range was from 40 to 400 m/z. The ion source, interface, and injector port temperatures were 260 °C. For each sample, a 5% w/v solution in CH2Cl2 was prepared, and 0.1 μL was injected using a split ratio of 1:24. The oven temperature was initially 50 °C, increased to 260 °C at a rate of 2 °C/min. The components of the essential oils were identified by comparison of their mass spectral fragmentation patterns and on the basis their GC retention indices (RI), calculated using a homologous series of n-alkanes (C8-C40) injected under the same chromatographic conditions, with those available in the MS databases.13-16 The data were handled using the LabSolutions GCMS solution software version 4.45 (Shimadzu Scientific Instruments, Columbia, MD, USA).
Gas Chromatography with Flame Ionization Detection
The GC-FID analyses were carried out using a Shimadzu GC 2010 (Shimadzu Scientific Instruments, Columbia, MD, USA) equipped with flame ionization detector, a split/splitless injector, and Shimadzu autosampler AOC-20i, with a ZB-5 capillary column (60 m × 0.25 mm i.d.; film thickness 0.25 μm) (Phenomenex, Torrance, CA, USA). The oven temperature was programmed as above for GC-MS. Injector and detector temperatures were maintained at 260 °C, respectively. The carrier gas was helium and the flow rate was 1.0 mL/min. For each sample, a 5% w/v solution in CH2Cl2 was prepared, and 0.1 μL was injected using a split ratio of 1:31. The percent compositions of the essential oils were calculated from peak areas with quantification using the external standard method; calibration curves of representative compounds from each class were used for quantification.
Chiral Gas Chromatography – Mass Spectrometry
The chiral GC-MS analysis was performed using a Shimadzu GCMS-QP2010S (Shimadzu Scientific Instruments, Columbia, MD, USA) gas chromatograph equipped with a Restek B-Dex 325 chiral capillary GC column (Restek Corp., Bellefonte, PA, USA) (30 m × 0.25 mm ID × 0.25 μm film thickness). The temperature of the injection was 260 °C. The samples were diluted to 3% w/v with CH2Cl2 and the injection volume was 0.1 μL using a split ratio of 1:25. Helium was used as carrier gas at a constant flow rate of 1.8 mL/min. The initial oven temperature was 50 °C and then increased at a rate of 1.5 °C/min to 120 °C, then heated to 260 °C at a rate of 2 °C/min, and then held at 200 °C for an addition 5 min. The temperature of the detector was 260 °C. The peaks were identified by comparing retention times (RT) with authentic samples obtained from Sigma-Aldrich (Milwaukee, WI, USA). Relative enantiomer percentages were determined based on peak areas.
Footnotes
Acknowledgments
Author Contribution Statement
W.N.S. designed the study; K.S. and W.N.S. collected the plant material; W.N.S. carried out the hydrodistillations; A.P. and A.R. obtained the GC-MS, GC-FID, and chiral GC data; P.S. and W.N.S. analyzed the GC data. All authors contributed to the writing and editing of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
