Abstract
Thrombin activation after cerebral hemorrhage induces the death of neurons and astrocytes. Pyrroloquinoline quinone (PQQ) shows nutritional functions and cell protection. This study aimed to clarify the protective effects of PQQ on thrombin-induced cell death in astrocytes. Murine SMA560 astrocytoma cells were used in this study. The cell viability was measured by 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2-H-tetrazolium bromide assay. The changes in reactive oxygen species (ROS) and mitochondrial membrane potential (MMP) were observed by CellROX® Deep Red and JC-1, respectively. The expression of apoptotic genes was measured by reverse transcription-quantitative polymerase chain reaction. Thrombin dose- and time-dependently induced SMA560 cell death. PQQ significantly repressed thrombin-induced SMA560 cell death in a dose-dependent manner. Thrombin led to the diminishment of MMP, increased production of ROS, and the upregulated expression of apoptotic genes including c-Jun, TP53, Bim, Puma, and Noxa in SMA560 cells. Meanwhile, PQQ treatment significantly attenuated the effects of thrombin on ROS, MMP, and gene expression in SMA560 cells. In conclusion, PQQ protects SMA560 astrocytes against thrombin-induced cell death by inhibiting oxidative stress and improving mitochondrial function in vitro.
Keywords
Introduction
Cerebral hemorrhage leads to the death of both neurons and astrocytes. Astrocytes play an important role in preserving the viability of neurons through multiple pathways including the release of paracrine molecules, the buffering of ion changes, and the direct connection with neurons. 1 It has been shown that the astrocyte–neuron interaction is critical for the maturation of neurons and synaptic circuits. 2 Therefore, to investigate the mechanism of astrocytes’ death and their protective methods may be of great significance for the prevention of neuronal loss after cerebral hemorrhage.
It is well known that thrombin is able to induce the death of the cells in the hemorrhagic region when it is activated after cerebral hemorrhage to initiate coagulation. 3 Thrombin activation influences the pathogenesis of secondary injury progression of cerebral hemorrhage. 4 Thrombin acts through protease-activated receptors (PARs) to elicit intracellular signaling transduction pathways. 5 It was reported that thrombin could activate c-Jun N-terminal kinase (JNK) and caspase 3 to induce neuronal apoptosis via PAR1 signaling.6,7 Meanwhile, PAR1 is expressed in astrocytes, and thrombin also induces astrocyte apoptosis.6,8 Repression of thrombin-induced apoptosis of astrocytes can be beneficial to the protection of neural function since astrocytes improve the viability of neurons.
Pyrroloquinoline quinone (PQQ) is an aromatic tricyclic o-quinone and shows a wide range of nutritional functions and improves the viability of many kinds of cells. 9 PQQ regulates the activities of enzymes that are important for cell metabolism. For example, it binds to lactate dehydrogenase in mammalian cells and augments the conversion of lactate to pyruvate, which may enhance adenosine triphosphate production via the mitochondrial tricarboxylic acid cycle and increase cell viability. 10 PQQ also possesses the ability to influence the cellular redox state, which plays an important role in regulating cell viability.11,12 PQQ scavenges reactive oxygen species (ROS) to attenuate oxidative stress and stimulates mitochondrial biogenesis, and its deficiency influences the normal energy metabolism and mitochondrial function.13,14 Since mitochondria and redox state play a central role in cell apoptosis, 15 PQQ is proposed to be a therapeutic agent to reverse the neural damage that is closely linked to mitochondrial dysfunction after cerebral hemorrhage. It has been shown that PQQ promotes neurite growth of mouse neurons and protects primary cultured rat midbrain neurons against rotenone-induced injury. 16 On the other hand, the effect of PQQ on the survival of astrocytes after cerebral hemorrhage is not clear. In this study, the beneficial effects of PQQ on the survival of astrocytes under treatment with thrombin, an important mediator of cellular damage after cerebral hemorrhage, were observed and the molecular mechanism was investigated.
Materials and Methods
Materials
PQQ and thrombin were purchased from Sigma-Aldrich (Merck KGaA). JC-1, ROS assay probe CellROX® Deep Red, Dulbecco’s modified Eagle’s medium (DMEM), fetal bovine serum (FBS), and reagent for cell culture were the products of Thermo Fisher. RNA extractive kit, reverse transcription (RT), and quantitative polymerase chain reaction (qPCR) reagents were obtained from Takara Bio USA Inc.
Cell Culture
Murine SMA560 astroglioma cells were cultured in an incubator with DMEM containing 10% FBS, 100 U/mL penicillin, and 100 μg/mL streptomycin in 5% CO2 at 37 °C. The medium was regularly changed every 3 days. SMA560 cells were subcultured after 80% confluence and planted into either plates or dishes for different measurements.
3-(4,5-Dimethyl-2-thiazolyl)-2,5-diphenyl-2-H-tetrazolium bromide Assay
SMA560 cells, grown up to 70% confluence in 96-well plates, were treated by thrombin and the other compounds for the fixed time period. 3-(4,5-Dimethyl-2-thiazolyl)-2,5-diphenyl-2-H-tetrazolium bromide (MTT) was added into medium (0.5 mg/mL) for 4 h. Then the medium was discarded and 100 μL isopropanol with 0.01 M hydrochloric acid was added to each well. MTT crystals were dissolved and the absorbance values per well at 560 nm wavelength were measured.
ROS Measurement
SMA560 cells were treated with thrombin and the other compounds for a fixed period. Then CellROX® Deep Red (Thermo Fisher Scientific) was added to the medium at a final concentration of 5 μM for 30 min at 37 °C. The cells were washed 3 times with phosphate-buffered saline and visualized under a confocal microscope (Leica SP8). The cellular fluorescence intensity with excitation/emission (Ex/Em) at 640 nm/665 nm was recorded and used for data analysis.
Mitochondrial Membrane Potential Measurement
JC-1 exhibits mitochondrial membrane potential (MMP)-dependent accumulation in mitochondria with the shift of emission wavelength from green to red. 17 SMA560 cells were stained with JC-1 (5 μg/mL) for 15 min at 37 °C. The cells were washed and the fluorescence was recorded using a confocal microscope (Leica TCS SP8). The red fluorescence (Ex/Em at 560 nm/600 nm) was measured and the intensity of each cell was analyzed by LAS LITE software (Leica Biosystem).
Reverse Transcription-Quantitative Polymerase Chain Reaction
Total RNA of SMA560 cells was extracted and RT using cellular RNA extraction and RT kits (Takara Biotechnology), and the complementary DNA was used for qPCR. The reaction mixture was denatured for 5 min at 95 °C for 30 s and then went to 38 cycles of denaturing at 95 °C for 10 s, annealing, and extending at 60 °C for 15 s. The sequences of primers are listed in Table 1.
The Sequences of Primers Used in the Study

Statistical Analysis
The data are presented as mean ± SEM. The data were analyzed by one-way analysis of variance and Bonferroni posthoc test to calculate the differences of the groups. P < .05 was considered as being a statistically significant difference.
Results
Effects of Thrombin and PQQ on the Death of SMA560 Cells
First, the dose- and time-dependent effects of thrombin on SMA560 cell viability were observed. Thrombin at a concentration below 30 U/mL did not show significant influence on SMA560 cell viability in 48 h incubation, but at concentrations of 300 and 1000 U/mL inhibited SMA560 cell viability after 12 h treatment and showed stronger effects at 48 h (P < .05, n = 12, Figure 1A). PQQ dose-dependently attenuated thrombin-induced death of SMA560 cells, as indicated by MTT assay. PQQ at the concentration above 10 μmol/L significantly improved the thrombin (300 U/mL) induced decrease in cell viability after 24 h incubation (Figure 1B).
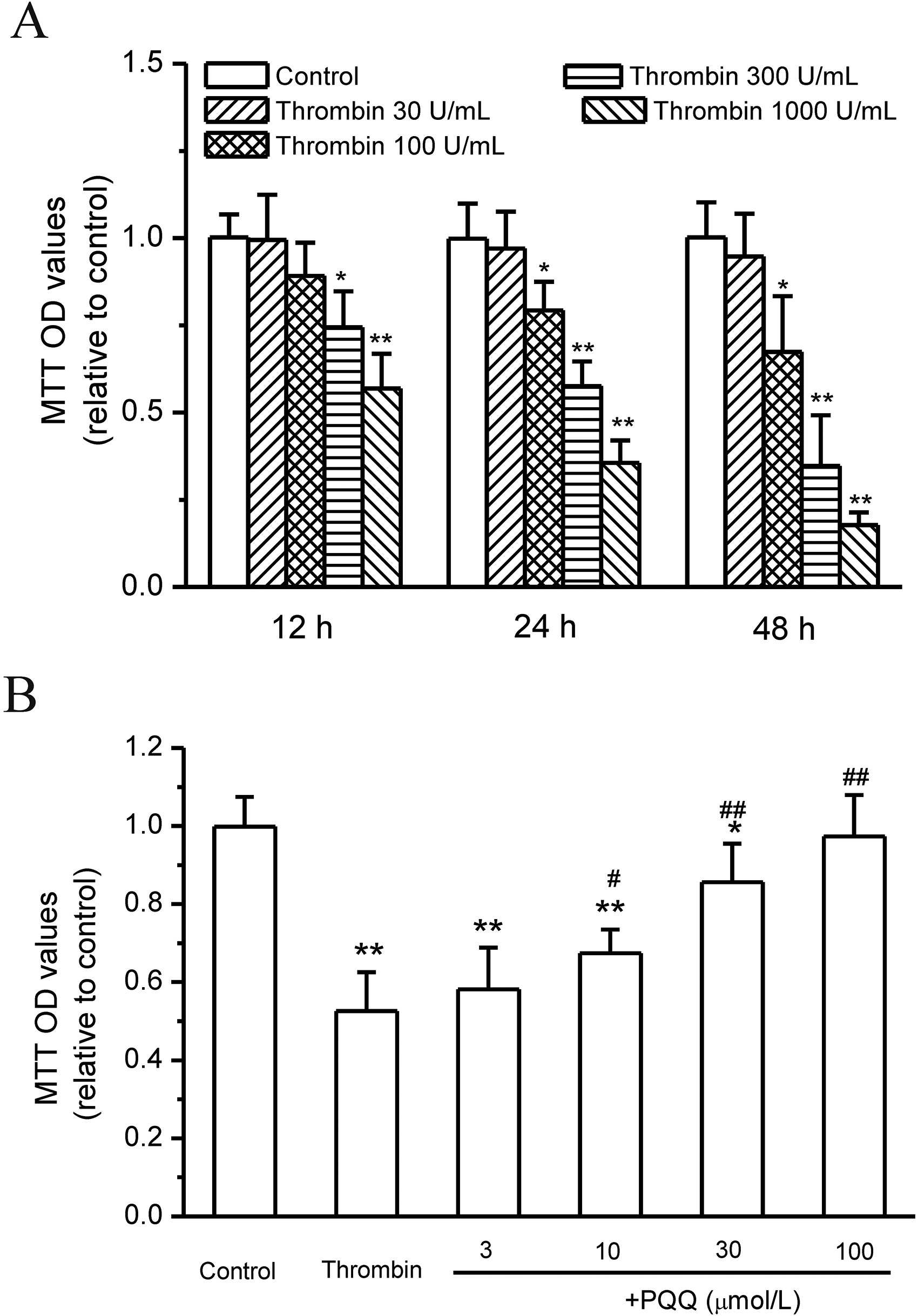
Effects of thrombin and pyrroloquinoline quinone (PQQ) on the death of SMA560 cells. (A) Thrombin induced the death of SMA560 cells. (B) PQQ attenuated thrombin-induced SMA560 cell death (*P < .05 and **P < .01 vs control, #P < .05 and ##P < .01 vs thrombin, n = 12).
Effects of Thrombin and PQQ on ROS Levels in SMA560 Cells
The ROS levels significantly increased in SMA560 cells after thrombin treatment (300 U/mL) for 12 h, as indicated by the intensity of CellROX® Deep Red. PQQ (30 μmol/L) treatment significantly attenuated thrombin-induced increase in ROS in SMA560 cells (P < .05, n = 3, Figure 2).

Effects of thrombin and PQQ on ROS levels in SMA560 cells. (A) Representative staining of ROS indicator CellROX® Deep Red (Bar means 50 μm). (B) Statistical analysis of the intensity of CellROX® Deep Red (**P < .01 vs control, ##P < .01 vs thrombin, n = 6).
Effects of Thrombin and PQQ on Mitochondrial Function of SMA560 Cells
Driven by MMP, JC-1 accumulates in mitochondria to emit red fluorescence. Thrombin treatment (300 U/mL) for 12 h resulted in a significant decrease in JC-1 red fluorescence intensity in SMA560 cells, indicating damage to mitochondrial function. PQQ (30 μmol/L) treatment significantly attenuated this thrombin-induced decrease in JC-1 red fluorescence intensity in SMA560 cells (Figure 3).

Effects of thrombin and PQQ on MMP of SMA560 cells. (A) Representative staining of JC-1 (bar means 50 μm). (B) Statistical analysis of the intensity of JC-1 (**P < .01 vs control, ##P < .01 vs thrombin, n = 6).
Effects of Thrombin and PQQ on Apoptotic Gene Expression in SMA560 Cells
The expression of apoptotic genes was stimulated by thrombin. Thrombin treatment (300 U/mL) for 12 h induced a significant increase in expression of c-Jun, TP53, Bim, Puma, and Noxa, but the expression of Bax and Bcl-2 was not significantly influenced. PQQ (30 μmol/L) treatment significantly attenuated thrombin-induced increase in the expression ROS in c-Jun, TP53, Bim, Puma, and Noxa in SMA560 cells (P < .05, n = 3, Figure 4).

Effects of thrombin and pyrroloquinoline quinone (PQQ) on apoptotic gene expression in SMA560 cells (*P < .05 vs control, #P < .05 vs thrombin, n = 3).
Discussion
This study found that thrombin dose- and time-dependently decreased SMA560 cell viability and induced cell death. Thrombin led to SMA560 cell apoptosis at a concentration above 100 U/mL at 12 h treatment and showed stronger effects after 24 and 48 h treatment. After 48 h treatment, there were no cytotoxic effects of thrombin at 30 U/mL and below. It was reported that low concentrations of thrombin are neuroprotective and high concentrations stimulate apoptosis of neuronal cells.18,19 Although the neuroprotective effects of thrombin at low concentration did not appear in SMA560 astrocytes, the present results are in accord with the previous reports that showed cytotoxicity on astrocytes of thrombin at high concentration. 6
It is well established that thrombin activates PARs to exhibit a role in the coagulation cascade and regulation of cell viability by stimulating different subtypes of G-proteins and subsequent diverse signaling molecules.20,21 Different signaling pathways have been reported to mediate thrombin-induced cell apoptosis, which include molecules such as JNK, the extracellular signal-regulated kinases 1 and 2, ras homolog family member A, nuclear factor-κB, and signal transducer and activator of transcription.6,7,22 In this study, thrombin stimulated the expression of c-Jun and TP53 in SMA560 cells. The products of c-Jun and TP53 are 2 transcription factors that induce apoptosis after being activated by apoptotic factors. 23 Puma and Noxa are the target genes of p53 and Bim that of gene c-Jun. 24 The expression of Puma, Noxa, and Bim in SMA560 cells was upregulated by thrombin in SMA560 cells. This indicates that thrombin induces SMA560 cell apoptosis by activating the PAR1 and c-Jun/P53 signaling pathways. However, other signaling pathways cannot be excluded since they may also regulate the expression of Puma, Noxa, and Bim. The exact role of other signaling pathways and the interactions among the signaling molecules in thrombin-induced SMA560 cell apoptosis is not well defined and remains to be debated in the future.
Thrombin-induced SMA560 cell apoptosis was blocked by PQQ treatment. PQQ is a nutritionally important growth factor to improve reproduction ability and stimulate neonatal growth. 25 The present study found that PQQ dose-dependently reversed thrombin-induced apoptosis in SMA560 astrocytes. PQQ also protects against apoptosis in other kinds of cells, such as myocardial and neural cells. 26 Antioxidation is suggested to be the molecular mechanism of the cytoprotective role of PQQ. 29 In the present study, thrombin led to oxidative stress in SMA560 cells, and PQQ completely inhibited thrombin-induced ROS elevation. Antioxidation may be the reason for PQQ to attenuate thrombin-induced SMA560 cell apoptosis. On the other hand, thrombin resulted in MMP loss and mitochondrial dysfunction, which were fully prevented by PQQ treatment. The dysfunction of mitochondria causes excessive ROS production, and this results in further mitochondrial dysfunction. This deterioration of mitochondrial function finally leads to the activation of the apoptotic pathway. PQQ protects mitochondria by attenuating ROS. Meanwhile, PQQ is able to promote mitochondria biogenesis by stimulating the expression of peroxisome proliferator-activated receptor-γ coactivator-1α.9,14,30,31 Thus, a mutual improvement between ROS reduction and mitochondrial biogenesis under PQQ stimulation protects SMA560 astrocytes against thrombin-stimulated cell apoptosis. The detailed mechanism of PQQ to elicit the mutual improvement between ROS and mitochondria remains to be clarified in future studies.
The favorable prognosis after cerebral hemorrhage depends on the survival of neurons. A previous study has shown that PQQ protects neurons against cerebral hemorrhage-induced apoptosis. 32 The present study demonstrated that PQQ also protects astrocytes against cell death that is induced by thrombin, an important mediator of cerebral hemorrhage-induced cell apoptosis. It has been known that astrocytes interact with neurons through multiple pathways to preserve neural function. 33 Thus, it is suggested that PQQ benefits neural function not only by direct action on neurons, but also by protection of astrocytes after cerebral hemorrhage. The present study further establishes the foundation for PQQ usage in the early stage of cerebral hemorrhage when thrombin is activated.
In conclusion, PQQ protects SMA560 astrocytes against thrombin-induced cell death by inhibiting oxidative stress and improving mitochondrial function in vitro.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: This work was supported by the Natural Science Basic Research Program of Shaanxi (Program No. 2019JZ-38).
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
