Abstract
Ellagic acid (EA), a natural food component, has been identified as a functional food because of its role in disease prevention. In our previous study on the chemical component of the seeds of Cornus officinalis Sieb. et Zucc., ellagitannins (ETs) were firstly isolated and identified; these compounds can yield EA by acidic hydrolysis. Therefore, to evaluate the seeds as a potential source of EA, the free and total EA contents of the seeds, sarcocarps, and barks were evaluated separately. The highest total EA content was found in seeds (14.51-21.58 mg/g), followed by the sarcocarp (1.97-4.51 mg/g) and bark (trace amounts). EA mainly existed as ET in both the seeds and sarcocarps, while a small portion existed in free form. The findings of this study prove that the seeds of C. officinalis are a rich source of EA. This study reports a unique EA profile of C. officinalis for the first time, which may assist in the selection of EA sources for use in research and for the food and pharmaceutical industries.
Introduction
Recently, the consumption of natural plant-based food products has gained considerable interest because of their health benefits. Polyphenols are present in various plant-based products, such as tea, fruits, and medicinal plants, and account for the key functional components present in such foods.1,2 The polyphenol ellagic acid (EA) (Figure 1) is a dimeric derivative of gallic acid that is found in several economically important plants, particularly nuts and fruits. 3 EA can bind to the specific precursor hexahydroxydiphenic acid (HHDP); alternatively, it existed as ellagitannins (ETs). 4
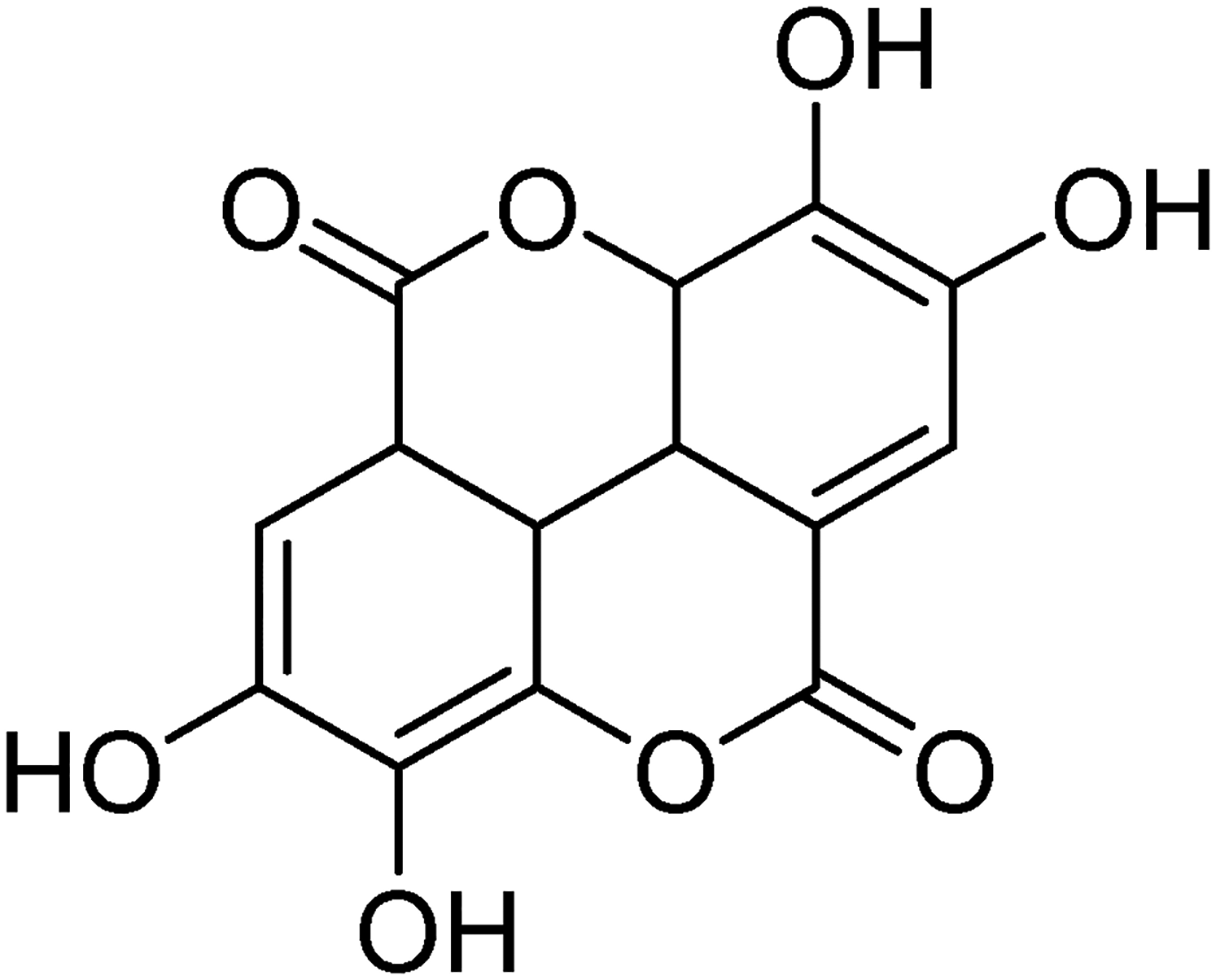
Ellagic acid (EA).
EA shows skin whitening, antioxidant, anti-mutagenesis, and anti-carcinogenesis activities. 5 EA consumption is additionally associated with health benefits in the prevention of oxidative-linked chronic diseases, including cancer, diabetes, and cardiovascular and neurodegenerative diseases 6 , and, therefore, EA consumption has attracted increasing attention in the past few years.
Cornus officinalis Sieb. et Zucc., also known as Shan Zhu Yu, Japanese Cornel Dogwood or Asiatic Dogwood in China, is a deciduous shrub that belongs to the family Cornaceae. The dried and ripe sarcocarp of C. officinalis fruits is used to prepare Chinese herbal medicine, which is generally referred to as Corni Fructus (CF). Both in vitro and in vivo studies have shown that CF exhibits various pharmacological effects, including anticancer, hypoglycemic, hepatoprotective, antioxidant, neuroprotective, anti-inflammatory, and nephroprotective activities. 7 CF is usually combined with Moutan Cortex, Radix Rehmanniae Praeparata, Alismatis Rhizoma, Dioscoreae Rhizoma, and Poria to prepare Liuwei Dihuang Wan, a famous Chinese patent medicine, used to treat clinical symptoms, like vertigo, tinnitus, and weakness in the waist and knees. Moreover, the sarcocarp of C. officinalis is used to produce fruit juice, preserved fruit, vinegar, and fruit wine.
The use of C. officinalis seeds, which are the by-products of CF, has received limited attention because of lack of data on their functional components. The seed is generally discarded after the sarcocarp is successfully separated from the ripe fruit. Therefore, we previously studied the chemical components of C. officinalis seeds to enable their use. The hydrolysable ETs, such as tellimagrandin I (Figure 2(A)), 12,3-tri-O-galloyl-4,6-(S)-HHDP-β-D-glucopyranose (Figure 2(B)), and 14,6-tri-O-galloyl-2,3-(S)-HHDP-β-D-glucopyranose (Figure 2(C)), have been successfully separated and identified from the seeds of C. officinalis for the first time 8 .
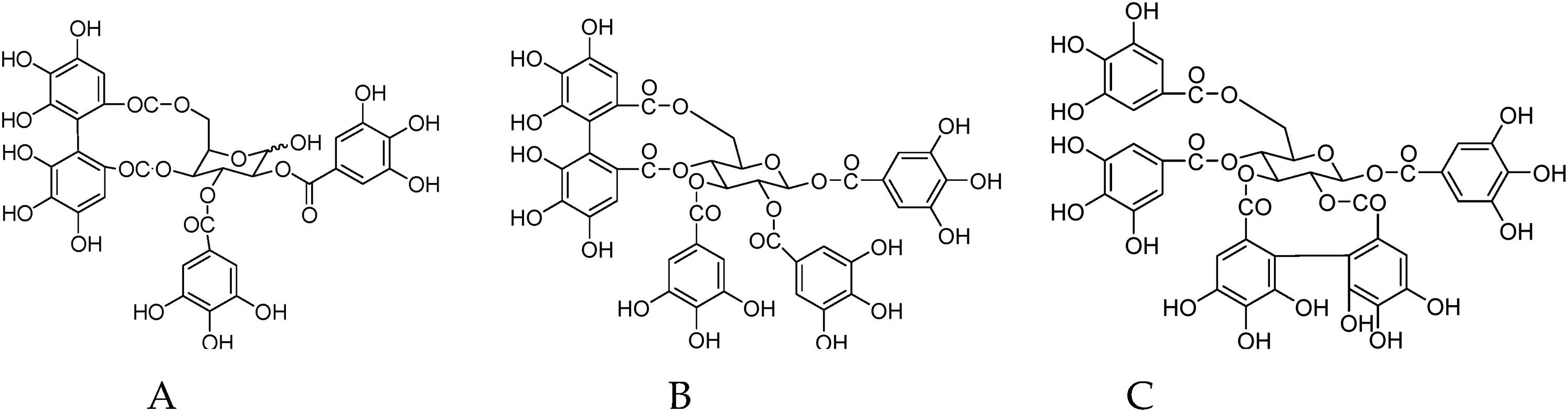
Ellagitannins (ETs) isolated and identified from the seeds (A) tellimagrandin I, (B) 12,3-tri-O-galloyl-4,6-(S)-HHDP-β-D-glucopyranose, and (C) 14,6-tri-O-galloyl-2,3-(S)-HHDP-β-D-glucopyranose.
ET hydrolysis usually generates HHDP, and the latter can be simultaneously lactonized to form EA (Figure 3). To evaluate C. officinalis comprehensively as a potential EA source that can be utilized as a food additive, the contents of total and free EA should be determined in different plant parts obtained from different production areas. In this study, the EA content of the seeds and sarcocarps of fruits and the barks from plants growing in the five main CF production areas in China was extracted and determined.

ETs (A) hydrolysis into EA (C) by producing HHDP (B), and HHDP is also subjected to lactonization to EA (C)at the same time.
Materials and Methods
Chemicals and Standards
EA was obtained from the National Institutes for Food and Drug Control (Beijing, China), trifluoroacetic acid and high-performance liquid chromatography (HPLC)-grade methanol from Dikma Scientific (Tianjin, China), and the remaining chemicals, which were analytically pure, from China Tianjin Chemical Factory (Tianjin, China).
Plant Source
Mature fruits of C. officinalis were harvested from the five main CF production areas in China. Fruits and barks were collected from Luanchuan, Songxian, Xixia, Nanzhao, Linru, and Nanzhao. The sarcocarps and seeds were manually separated. All samples were dried in an oven at 40°C until a constant weight was achieved, and then stored at −4°C prior to analysis. The samples were identified by Prof. Ximing Lu, Medical College, Henan University of Science and Technology, Luoyang, China. Voucher specimens are maintained in the College Herbarium with the certificate No. 906(1-5).
Instrumentation and Chromatographic Conditions
HPLC quantification was performed using a Shimadzu Prominence LC-20A System (Shimadzu) comprising a degasser (DGU-20A5), diode array detection system (SPD-M20A, Shimadzu), and a binary gradient pump (LC-20AB). Labsolutions (version 5.97, Shimadzu) was used to control equipment, obtain and process data, and manage chromatographs. A reversed-phase C18 Wondasil column (4.6 × 250 mm, 5 μm, GL Sciences) and the matched guard column (Phenomenex) were used for analysis. MeOH:H2O:H3PO4 = (27:73:0.08) was used as the mobile phase. The separation conditions were as follows: flow rate 1.0 mL/min, sample injection volume 20 μL, column temperature 30°C, and UV detector wavelength 254 nm.
Preparation of Standard Solutions
An EA standard stock solution (0.042 mg/mL) was prepared using methanol. Subsequently, the standard stock solution was serially diluted using methanol to prepare 5 standard working solutions (0.0021-0.042 mg/mL) to prepare the respective calibration curves. Each solution was filtered through a 0.45 μm membrane filter. Under the aforementioned conditions, the retention time for EA was determined as 26.3 min. The sample was additionally co-injected with pure EA to confirm the position of the EA peak in chromatographic profiles.
Free EA Sample Preparation for HPLC Analyses
A 100% methanol extraction solution was prepared for each assay. Extraction was performed as described by Williams et al 9 . In brief, 1.0 g ground sample was collected in a 50 mL centrifuge tube containing a screw cap, and diluted using methanol to 25 mL, followed by 30 min of stirring under 25°C. Thereafter, the mixed solution was subjected to 20 min of ultrasonication at 100 W and 10 min of centrifugation at × 3200 g. The supernatant was collected and preserved at −30°C for subsequent analysis. Additionally, a small portion of supernatant was filtered through a 0.45 μm microporous membrane before injection.
Total EA Sample Preparation for HPLC Analyses
Acid hydrolysis of the methanol extracts was performed as described by Vekiari et al 4 . Briefly, 13 mL of supernatant was subjected to evaporation until dryness using a rotary evaporator, whereas the remaining supernatant was dissolved in a solvent comprising trifluoroacetic acid and methanol (2 mL each), followed by 2 h of reflux under 80°C. Subsequently, the mixed solution was subjected to evaporation until dryness in the presence of N2, whereas the remaining portion was redissolved in 5 mL methanol-dimethyl sulfoxide (1:1) and then diluted by 100-folds using methanol. The sample was filtered through a 0.45 μm membrane filter prior to HPLC quantification.
Method Validation
All validation parameters are provided in online Supplemental Material.
Statistical Analysis
All measurements were repeated thrice and data are expressed as mean ± SD; one-way analysis of variance was performed. Duncan's multiple range tests were used to test the significant differences. All statistical analyses were performed using SPSS 11.0 (IBM). A P value < 0.05 was considered significant.
Results and Discussion
Determination of Free EA Content
Free EA has been suggested to function as an antioxidant in vivo and in vitro, and its antioxidant activity is determined based on its chemical structure, especially the available hydroxyl number. 2 Zafrilla et al 10 showed that free EA has excellent antioxidant activity, which is related to the two dihydroxyl groups present in EA (Figure 1). Therefore, the free EA level should be measured prior to hydrolysis. In the present study, the highest level of free EA was present in seeds ranging from 0.25 to 0.94 mg/g (d.w.), followed by the sarcocarp containing 0.09 to 0.30 mg/g (d.w.), and trace amounts of free EA were found in the bark. However, the free EA content in the seeds estimated in the present study was lower than that of 3.03 to 12.04 mg/g (d.w.) reported by Zhang et al 11 . To investigate the inconsistencies between the analyzed and reported values, we evaluated the free EA extraction procedure used by Zhang et al 11 . The rigorous extraction conditions (70°C for 1 h in water, aqueous ethanol, and aqueous methanol) applied by the authors, provide a favorable condition for ET hydrolysis. Additionally, the findings of Okuda et al 12 support this observation since they observed that higher temperatures for extended times mediate the release of free EA from ETs. Therefore, the combination of extraction factors provides a considerable overestimation of the free EA content in seeds. In the present study, an ultrasonication extraction method was used for obtaining free EA; therefore, our results were consistent with the real free EA levels in seeds of C. offcinalis.
The free EA content in seeds determined in our study was higher than that reported by Williams et al 9 who reported a free EA content of 0.055 and 0.048 mg/g (d.w.) in boysenberry (Rubus ursinus x Rubus idaeus) and strawberry (Fragaria x ananassa), respectively. The free EA contents of 0.71, 0.51, and 0.40 mg/g (d.w.) in the flowers, inner skin, and leaves of Japanese chestnut (Castanea crenata) reported by Tuyen et al 13 are similar to the values in C. officinalis seeds, but higher than that in sarcocarps determined in our study. Furthermore, the free EA contents of 23.124 and 6.207 mg/g (d.w.) in grape seeds (Vitis vinifera) and Kakadu plum fruits (Terminalia ferdinandiana) reported by Stanciu et al 14 and Williams et al, 9 respectively, are much higher than those for C. officinalis seeds and sarcocarps in the present study.
Determination of Total EA Content
In the present study, the total EA content of C. officinalis seeds ranged from 14.51 to 21.58 mg/g (d.w.), the mean of which is 5.57-fold higher than that of the sarcocarp with a range from 1.97 to 4.51 mg/g (d.w.). When considering the seeds, sarcocarps, and barks obtained from five different regions, the seeds were the richest source of total EA. The samples collected from diverse CF production regions in China varied in terms of their total EA and ET contents. The samples obtained from Luanchuan contained the highest total EA content in hydrolyzed seed samples, followed by samples obtained from Nanzhao. The differences may be due in part to the difference in environmental factors affecting plant growth and development.
The total EA content of C. offcinalis seeds was higher than that in many other EA-containing fruits. The total EA levels have been reported as 0.59 mg/g (d.w.) in walnut, 0.33 mg/g (d.w.) in pecan 5 , 0.616 mg/g (d.w.) in strawberry fruits 15 , 4.96 mg/g (d.w.) in boysenberries 16 , 8.80 mg/g (d.w.) in Kakadu plum (T ferdinandiana), 17 as well as 5.96 mg/g (d.w.) in blackberry, 18 which were converted to dry weight based on the application of a water content of 90.95% obtained after consultation of the USDA National Nutrient Database (2013) 19 for ease of comparison between the fresh weights. The mean total EA level in C. officinalis seeds of the five main CF production areas was 17.55 mg/g (Table 1). The total EA values of 120.45 mg/g (d.w.) for pomegranate (Punica granatum) rinds reported by Zhou et al 20 and 58.48 mg/g (d.w.) in the leaves of Kakadu plum (T ferdinandiana) reported by Williams et al 17 are higher than our reported value for C. officinalis seeds.
Ellagic Acid (EA) Contents (mg/g in d.w.) of Different Parts of Cornus officinalis.
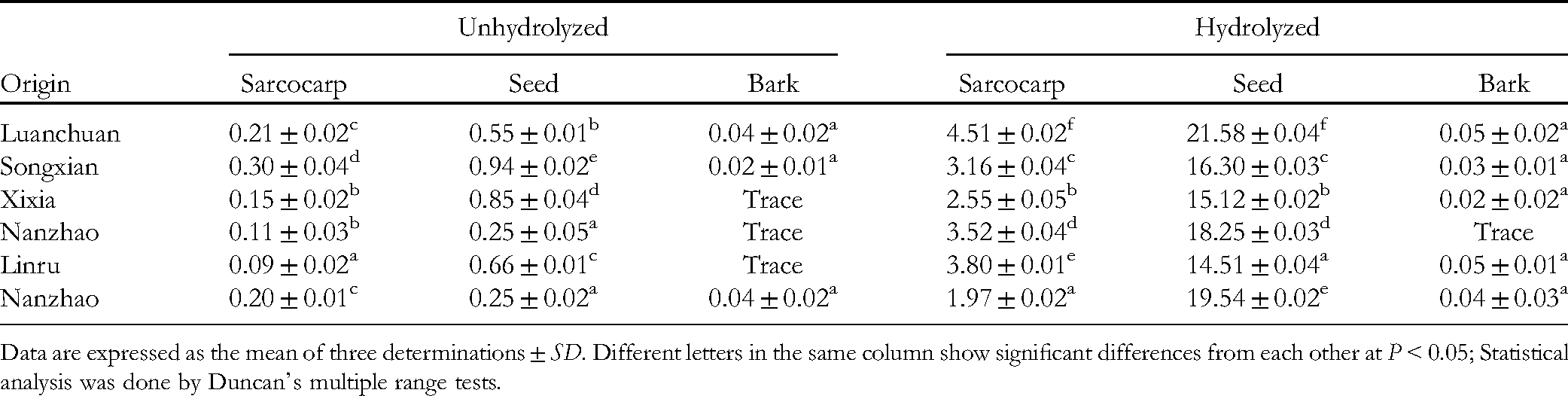
Data are expressed as the mean of three determinations ± SD. Different letters in the same column show significant differences from each other at P < 0.05; Statistical analysis was done by Duncan's multiple range tests.
Free/Total EA Ratio Levels
Apart from the free form, EA exists as ETs in plants, which are complex polymers formed via sugar esterification 20 . When acids are used to hydrolyze water-soluble ETs, an unstable intermediate HHDP is produced, which can form water-insoluble EA simultaneously (Figure 3). Such reactions are used to detect and quantify the total EA content. 9 In this study, the EA level was remarkably increased in the hydrolyzed seed and sarcocarp samples than that in unhydrolyzed samples; thus, it was speculated that a majority of total EA existed as ET. Based on the EA contents estimated in this study (Table 1), the proportion of free EA in total EA in the seeds of C. officinalis was calculated as 1.28% to 5.77%. This value is in agreement with previous findings that reported that most plant EA existed in the ET form, with only a small proportion comprising the free form.9,21,22
Determination of EA Content of Bark
The bark of C. officinalis was evaluated as a source of EA because the bark of chestnut tree has been reported to contain significant concentrations of total EA ranging from 2.83 to 18.4 mg/g. 4 We found that the bark of C. officinalis contained only traces amounts of free and total EA.
Conclusion
In the current study, the total EA levels in the seed, sarcocarp, and bark of C. officinalis were determined, and the seed was identified as a rich source of EA (14.51-21.58 mg/g). Moreover, EA in seeds was found to exist mostly in the ET form, with only a small proportion in the free form. Water-soluble ETs possess a larger number of phenolic hydroxyls in their structure, which are associated with a high antioxidant activity. Therefore, further research should evaluate the functional properties of ETs present in C. officinalis seeds to enable commercialization and nutraceutical applications. The seeds of C. officinalis represent an abundant and economical EA source, which can be applied in scientific research and in the food and pharmaceutical industries.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221080342 - Supplemental material for Cornus officinalis Sieb. et Zucc. Seeds as a Dietary Source of Ellagic Acid
Supplemental material, sj-docx-1-npx-10.1177_1934578X221080342 for Cornus officinalis Sieb. et Zucc. Seeds as a Dietary Source of Ellagic Acid by Ning Zhang, Jun Li and Hua Jiang in Natural Product Communications
Footnotes
Author Contributions
Conceptualization: Jun Li and Hua Jiang; Data curation: Hua Jiang and Ning Zhang; Formal analysis: Ning Zhang; Investigation: Jun Li and Ning Zhang; Methodology: Jun Li; Project administration: Hua Jiang and Jun Li; Writing—original draft, Hua Jiang and Ning Zhang; Writing—review and editing, Hua Jiang, Jun Li and Ning Zhang. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of the Henan Province of China, (grant number 182102311125).
Ethical Approval
Ethical Approval is not applicable for this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
