Abstract
Zingiber officinale Roscoe rhizome plays a vital role in food flavoring as well as utilization in folk medicine. Nepal is categorized among the leading countries of Z. officinale rhizome production and export. Mature Z. officinale rhizome collection is based on the major production sites within Nepal, but we are currently unaware of the chemical composition of essential oil for export. A comparative investigation of Z. officinale rhizome essential oil compositions, collected from 12 different sites of the eastern, mid, and western regions of Nepal, has been carried out. The analysis of essential oils chemical composition by gas chromatographic–mass spectral (GC-MS), enantiomeric composition by chiral gas chromatography–mass spectrometry (CGC-MS), and antimicrobial activity was evaluated. Essential oil yields ranged from 0.28% to 0.34%. The results showed that the essential oils obtained in this study contained α-zingiberene (8.6%-24.1%), camphene (7.2%-12.8%), β-phellandrene (3.8%-10.1%), neral (0.6%-11.8%), geranial (1.0%-17.4%), ar-curcumene (3.0%-10.3%), and β-sesquiphellandrene (3.7%-9.7%). With CGC-MS, the enantiomeric distributions of 21 chiral compounds were determined and showed no contrasting enantiomeric distributions. Two essential oil samples (G3 and G5) possessed good antibacterial activity against Pseudomonas aeruginosa (MIC = 78.1μg/mL) and excellent antifungal activity against Aspergillus niger (MIC = 39.1 μg/mL). Based on the content of α-zingiberene and citral (neral and geranial), samples collected from their respective areas might be used to identify an ideal Z. officinale rhizome production zone in Nepal.
Introduction
Zingiber officinale Roscoe rhizome (ginger) originated in southeast Asia 1 and is now cultivated worldwide. Production of Z. officinale rhizome in Asia contributes around 81% of the worldwide yield. The global Z. officinale rhizome market is expected to reach US$ 4.8 billion by 2027. Nepal is the world’s fourth largest Z. officinale rhizome producer and exporter according to the Food and Agriculture Organization (FAO). However, Nepalese Z. officinale rhizome quality isn’t competitive in the global market and the price is extremely low due to high dependency on the Indian market and lack of sophisticated processing units as well as ignorance about chemical composition and quality. 2 To intensify the trade-related environment and to strengthen the export of Nepalese Z. officinale rhizome in the international market, it is necessary to identify a potential geographical site for production and chemical composition. Nepal has an extreme altitudinal range from 60 to 8848 m with heterogeneous topography, distinct geo-climatic zones, 3 and adapted to Z. officinale rhizome production up to an elevation of 1500 m.1,4 Geographic and topographic variation are major challenges in the formation of major secondary metabolites in plants as they play primary roles of the defense system. 5
Less than 5.5% of essential oil extracted from Z. officinale rhizome has been reported in.6‐8 Supercritical fluid extraction (SFE) of Z officinale has also been carried out and compared to steam and hydrodistillation and has shown superior yields.8,9 Note, however, that SFE does not provide a true essential oil. Gas chromatographic–mass spectral (GC-MS) analysis revealed Z. officinale rhizome essential oil to contain α-zingiberene, ar-curcumene, β-bisabolene, β-sesquiphellandrene, and citral as major constituents. 10 Secondary metabolites from Z. officinale rhizome and their pharmacological activities have been explored in. 11 α-Zingiberene is a thermolabile major compound, however, low α-zingiberene content is may be caused by conventional distillation processes as it uses high temperatures leading to decomposition. 12 The content of α-zingiberene determines the quality of Z. officinale rhizome essential oil. 13 Z. officinale rhizome essential oil analyses have been the subject of a number of studies, which have shown significant differences in concentrations of major constituents.14‐19
Although much research has been carried out on Z. officinale rhizome, there have been no systematic investigations on comparative analysis of essential oils from different geographical locations of Nepal, nor have there been investigations regarding differences in their biological activities. The present study was carried out to examine the quality of Z. officinale rhizome, based on α-zingiberene and citral content as an assessment of essential oil quality, and to identify potential production zones within Nepal. This research will be beneficial to encourage both entrepreneurs and farmers to select the locations for investment as well as production of quality ginger, which should lead to increased exportation with better prices in the future as well as potential sources of quality Z. officinale rhizome essential oil for commercialization.
Results and Discussion
Isolation and Yields of Essential Oils
Hydrodistillation of fresh Z. officinale rhizome from the different geographical locations in Nepal gave relatively low yields of essential oils (0.28%-0.34% v/w). This is in close agreement with previously reported hydrodistillation yields of Z. officinale rhizome,6,7 including a previous report from Nepal. 20 It has been reported that higher yields of essential oil have been obtained from dried Z. officinale rhizomes as opposed to fresh rhizomes. 15 The yields of Z. officinale rhizome essential oil from different geographical locations of Nepal are summarized in Table 1.
Collection and Hydrodistillation Details for Zingiber officinale Rhizome Essential Oils from Nepal.

Chemical Composition of Essential Oils
The GC-MS analysis of Z. officinale rhizome essential oils revealed α-zingiberene (8.6%-24.1%), camphene (7.2%-12.8%), β-phellandrene (3.8%-10.1%), neral (0.6%-11.8%), geranial (1.0%-17.4%), ar-curcumene (3.0%-10.3%), and β-sesquiphellandrene (3.7%-9.7%)
to be the major components. The relative percentages of the main components of the Z. officinale rhizome from 12 geographical locations of Nepal and total essential oil compositions are listed in Table 2 and in Supplementary Table S1, respectively. A wide variation in Z. officinale rhizome essential oil chemical composition has been observed in the literature related to topological and geographical location as well as different factors such as maturity of rhizomes, harvesting time, genetic variation, and extraction procedures.8,14,20‐24
The Main Components of 12 Zingiber officinale Rhizome Essential Oils from Nepala.

Average concentration > 1.3%.
In this study, the major constituent of essential oils sample was citral (neral and geranial) except α-zingiberene in G6. Fresh Z. officinale rhizome essential oil with high citral content has been reported. 22 The highest concentration of α-zingiberene was found in sample G6 (24.1%) followed by G8 (20.2%), then G10 (15.8%), G7 (14.7%), and G3 (14.4%). Ekundayo et al reported that the essential oil from fresh Z officinale rhizome had less α-zingiberene than that obtained from dry rhizome. 25 In this study, fresh Z. officinale rhizome was used and that may be reason for the relatively low content of α-zingiberene.
The predominant compound class in G2 was the monoterpene hydrocarbons (32.2%), whereas G1, G4, G5, G9, G11, and G12 were rich in oxygenated monoterpenoids (34.6%-41.9%), and G3, G6, G7, G8, and G10 showed an abundance of sesquiterpene hydrocarbons (34.6%-62.4%). In these Z. officinale rhizome samples, monoterpene hydrocarbons and oxygenated monoterpenoids such as camphene, neral, and geranial were detected in significant concentrations consistent with fresh Z. officinale rhizome.
To provide insight into the variations, a hierarchical cluster analysis (HCA) has been carried out using the main components of Z. officinale rhizome essential oils from this work as well as compositions from other Himalayan locations, Sikkim,26‐28 Himachal Pradesh,29,30 Uttarakhand,21,30 as well as a previous report from Nepal 20 (Figure 1).

Dendrogram based on hierarchical cluster analysis (HCA) of Himalayan Zingiber officinale rhizome essential oils.
The cluster analysis shows two clearly defined clusters. Cluster A is made up mostly of samples from Sikkim and Nepal, and can be classified as a citral/α-zingiberene chemotype. Cluster B, populated by samples from Himachal Pradesh and Uttarakhand, can be classified as a geraniol-rich chemotype. Pandotra and co-workers described two clusters of Z. officinale rhizome essential oils from northwestern Himalayas (Himachal Pradesh and Uttarakhand). 30 The two clusters defined by Pandotra et al are indicated as BI and BII as shown in Figure 1. Cluster BI can best be described as a cluster without a dominating component. Cluster BII, on the other hand, is a cluster rich in geraniol, geranyl acetate, and α-zingiberene. To place these essential oil compositions into perspective, a commercial Z. officinale rhizome essential oil (doTERRA International) was included in the cluster analysis for comparison. The samples most similar in composition to the doTERRA commercial sample are sample G6 (this work) and a Gorubathane cultivar from Sikkim 27 (subcluster AII). These essential oils were especially rich in α-zingiberene (26.4%, 24.1%, and 32.2% for doTERRA, G6, and Gorubathane, respectively), with lower concentrations of neral and geranial (Tables 2 and 3). Subcluster AI, on the other hand, is rich in α-zingiberene as well as citral (neral and geranial).
Comparison of Major Components of Himalayan Zingiber officinale Rhizome Essential Oils.

t = trace (< 0.05%).
Labeled as β-farnesene.
Labeled as β-farnesol.
Labeled as α-funebrene.
Labeled as sesquisabinene.
According to ISO 16928:2014, ginger essential oil from China should be composed of α-zingiberene (29%-40%), β-sesquiphellandrene (10%-14%), ar-curcumene (5%-11%), camphene (4.5%-10%), and β-bisabolene (2.5%-9%), while ginger essential oil from India should contain α-zingiberene (35%-40%), β-sesquiphellandrene (11.5%-13.5%), ar-curcumene (6.5%-9%), camphene (5%-8%), and β-bisabolene (2.5%-5.5%), and the essential oil from West Africa should have α-zingiberene (23%-45%), β-sesquiphellandrene (8%-17%), ar-curcumene (3%-11%), camphene (0.2%-12%), and β-bisabolene (3%-7%). In terms of α-zingiberene and β-sesquiphellandrene content, samples G6 and G8 had the highest percentages, but both were lower than the ISO standards. The ar-curcumene concentrations in the Nepal samples are comparable to the ISO standard for West African ginger essential oil. Camphene concentrations in the Nepal samples fall within the parameters for China ginger essential oil, but are somewhat high compared to the standard for India, while β-bisabolene concentrations are somewhat low compared to the ISO standards. The ISO standards for neral are 0.0% to 0.5%, 0.1% to 0.4%, and 0.2% to 2.0% for China, India, and West Africa ginger oils, respectively, while geranial ISO standards are 0.0% to 0.6%, 0.1% to 0.6%, and 0.5% to 3.5%, respectively. Thus, the neral and geranial concentrations in the ginger essential oils from Nepal are substantially higher than the ISO standards.
Enantiomeric Composition of Essential Oils
The enantiomeric distributions of 21 chiral terpenoids were determined in essential oil samples G2–G6. The relative percentages of the levorotatory (–) and dextrorotatory ( + ) enantiomers in the selected Z officinale rhizome essential oils are listed in Table 4. The majority of chiral compounds are in dextrorotatory form, including α-pinene, camphene, β-pinene, α-phellandrene, δ-3-carene, limonene, β-phellandrene, citronellal, camphor, terpinen-4-ol, borneol, α-terpineol, citronellol, germacrene D, β-bisabolene, and α-copaene. On the other hand, the levorotatory enantiomer predominated for bornyl acetate, β-elemene, and (E)-nerolidol, while the enantiomeric distribution varied for limonene, linalool, and δ-cadinene depending upon their geographical location. The presence of these identified chiral compounds in Z officinale rhizome essential oil and their enantiomeric distributions may serve as a benchmark for authentication or adulteration for consumer safety as well as quality for export.
Enantiomeric Distributions of Chiral Terpenoids of Selected Zingiber officinale Rhizome Essential Oils.

Antibacterial and Antifungal Activities
Previous studies have shown essential oils of Z. officinale rhizome to have demonstrated a broad range of antimicrobial activities against different pathogens.31‐34 This present study showed that the assayed Z. officinale rhizome essential oils had different degrees of microbial inhibitory activity. The minimum inhibitory concentrations (MICs) of selected Z. officinale rhizome essential oils against a panel of bacteria and fungi are shown in Tables 5 and 6, respectively. Samples G3 and G5 were effective against Pseudomonas aeruginosa with an MIC value of 78.1 μg/mL. The other Z. officinale rhizome essential oil samples showed no notable activities against the bacterial strains, and essential oils demonstrated weaker antibacterial activities than those of the positive control, gentamicin (MIC < 19.5 μg/mL).
Minimum Inhibitory Concentrations (MICs) of Zingiber officinale Rhizome Essential Oils Against Tested Bacterial Strains.

Minimum Inhibitory Concentrations (MICs) of Zingiber officinale Rhizome Essential Oils Against Tested Fungal Strains.

Z. officinale rhizome essential oil samples G3 and G5 showed effective activity against Aspergillus niger (MIC = 39.1 μg/mL) while G2 and G6 sample showed notable activity (MIC = 78.1 μg/mL). Similarly, G3 and G5 were active against Aspergillus fumigatus and Trichophyton mentagrophytes (MIC = 78.1 μg/mL) while G2 showed activity against Aspergillus fumigatus. However, the Z. officinale rhizome essential oils demonstrated weaker antifungal activities than those of the positive control, amphotericin B (MIC < 19.5 μg/mL).
Material and Methods
Plant Material and Isolation of Essential Oils
Fresh mature Z. officinale rhizomes were collected in September/October 2019 (ie, during the flowering stage) from 12 different geographical locations in Nepal based on the major production site (Figure 2); G1–G12 represents samples from different collection sites. The collected sample mass was around 250 g from each site and 200 g of each rhizome was used for the hydrodistillation. After collection, each of the rhizomes was cut into smaller pieces and rhizomes and water in 1:2 ratios were hydrodistilled for 6 h using a Clevenger-type apparatus. The essential oils were dried with anhydrous sodium sulfate and stored in bottles at 5 °C until analysis. Z. officinale rhizome essential oil yields from different geographical locations, GPS coordinates with altitude are summarized in Table 1.
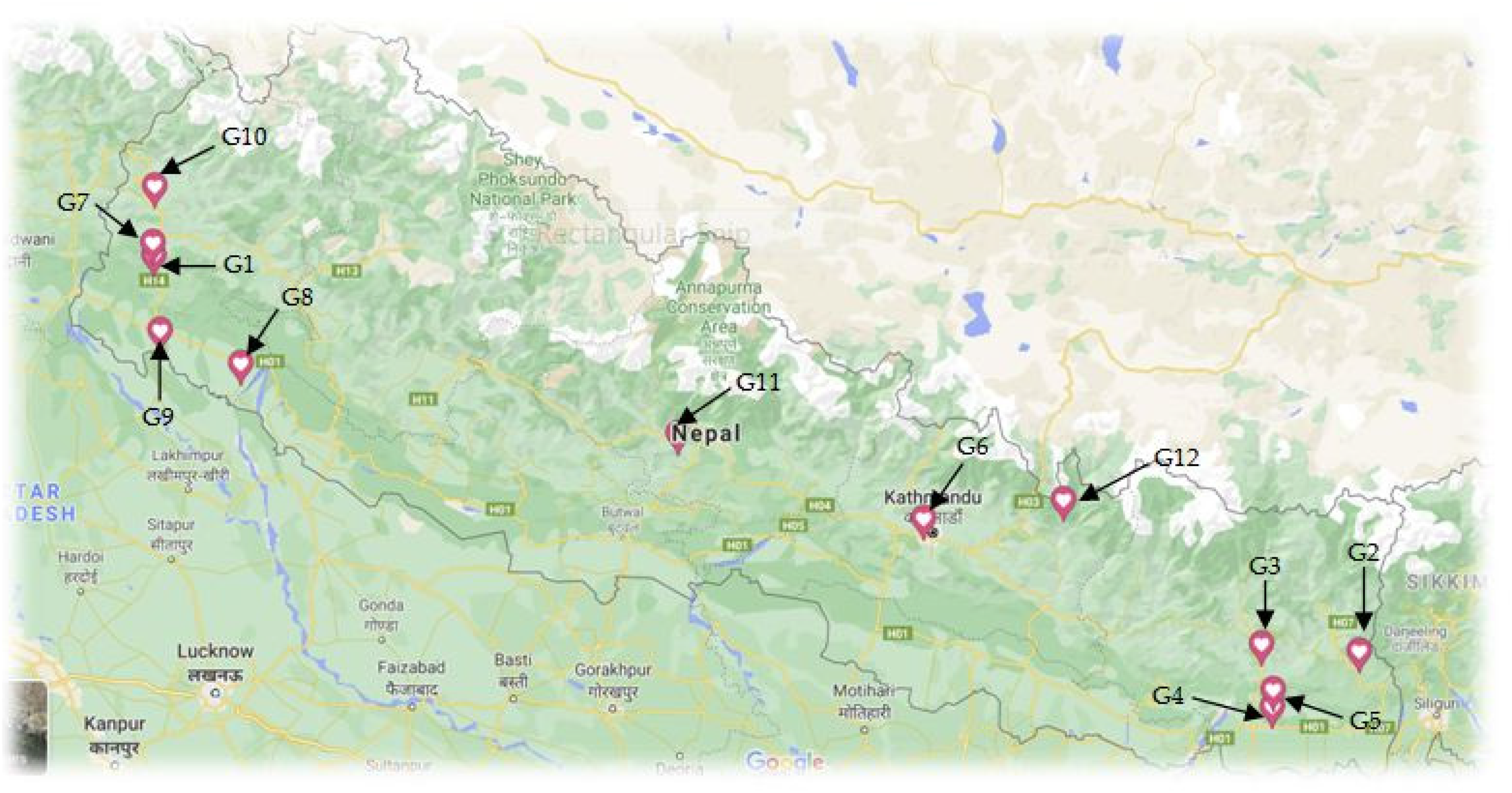
Collection sites of Zingiber officinale rhizomes (G1–G12).
Chemical Composition by GC-MS
Analysis of the chemical constituents in the Z. officinale rhizome essential oils was carried out using a Shimadzu GCMS-QP2010 Ultra under the following conditions: mass selective detector (MSD), operated in the electron impact (EI) mode (electron energy = 70 eV), with scan range = 40 to 400 m/z, and scan rate = 3.0 scans/sec. The GC column was a ZB-5MS fused silica capillary with a (5% phenyl)-polydimethylsiloxane stationary phase, a film thickness of 0.25 μm, a length of 60 m, and an internal diameter of 0.25 mm. The carrier gas was helium (80 psi) with a column head pressure of 552 kPa and a flow rate of 1.37 mL/min. The injector temperature was 260 °C, and the detector temperature was 280 °C. The column temperature was set at 50 °C for 2 min and then increased at 2 °C/min to the temperature of 260 °C. For each essential oil sample, 1:10 v/v solution in dichloromethane (DCM) was prepared, and 0.3 μL was injected using a split ratio of 1:30. Identification of the individual components of the essential oils was determined by comparison of the retention indices and comparison of the mass spectral fragmentation patterns (over 80% similarity match) with those found in the MS databases using the LabSolutions GCMS solution software version 4.45 (Shimadzu Scientific Instruments).35,36 The component percentages were determined from peak areas without standardization.
Enantiomeric Analysis by Chiral Gas Chromatography–Mass Spectrometry (CGC-MS)
Shimadzu GCMS-QP2010S with EI mode (70 eV) and B-Dex 325 chiral capillary GC column was used to perform enantiomeric analysis of Z. officinale rhizome essential oils. Scans in the 40 to 400 m/z range at a scan rate of 3.0 scan/sec. The column temperature was set at 50 °C, at first increased by 1.5 °C/min to 120 °C and then 2 °C/min to 200 °C. The final temperature of the column was 200 °C and was kept constant. The carrier gas was helium with a constant flow rate of 1.8 mL/min. For each essential oil sample, 3% w/v solution in DCM was prepared, and 0.1 μL was injected using a split ratio of 1:45.35‐37 The enantiomer percentages were determined from peak area. Comparison of retention times and mass spectral fragmentation patterns with authentic samples obtained from Sigma-Aldrich (Milwaukee, WI, USA) was used to identify the enantiomers.
Antibacterial Activity Screening
Seven microorganisms were used to evaluate the antibacterial activities of some selected Z. officinale rhizome essential oils: Five Gram-positive bacteria, Bacillus cereus (ATCC-14579), Staphylococcus epidermidis (ATCC-12228), Propionibacterium acnes (ATCC-11827), Staphylococcus aureus (ATCC-29213), and Streptococcus pyogenes (ATCC-19615), and two Gram-negative bacteria, Serratia marcescens (ATCC-14756) and P aeruginosa (ATCC-27853), using the microbroth dilution technique. Tryptic soy agar medium was used to culture all tested bacterial strains. A 5000 μg/mL solution of each essential oil was prepared in dimethyl sulfoxide (DMSO) and twofold dilution in 100 μL of cation-adjusted Mueller–Hinton broth (CAMHB) (Sigma-Aldrich) was added to the top well of a 96-well microdilution plate. The prepared stock solution of essential oils was then serially twofold diluted in fresh CAMHB to obtain final concentrations of 2500, 1250, 625, 312.5, 156.3, 78.1, 39.1, and 19.5 μg/mL. The freshly harvested bacteria with approximately 1.5 × 10 8 CFU/mL final concentration were added to each well of 96-well microdilution plates and were incubated at 37 °C for 24 h. Gentamicin (Sigma-Aldrich) and DMSO were used as positive and negative controls, respectively.35,38
Antifungal Activity Screening
Seven fungal strains were used: A niger (ATCC-16888), Candida albicans (ATCC-18804), Microsporum canis (ATCC-11621), Trichophyton mentagrophytes (ATCC-18748), Aspergillus fumigatus (ATCC-96918), Microsporum gypseum (ATCC-24102), and Trichophyton rubrum (ATCC-28188). All tested fungi were cultured on yeast nitrogen base growth medium (Sigma-Aldrich). Stock solutions (5000 μg/mL) of some selected Z. officinale rhizome essential oils were prepared in DMSO and diluted as above. The freshly harvested fungi with approximately 7.5 × 107 CFU/mL final concentrations were added to each well of 96-well microdilution plates and were incubated at 35 °C for 24 h. DMSO and amphotericin B (Sigma-Aldrich) were negative and positive antifungal control, respectively.35,39
HCA
HCA was carried out based on the percentages of the major essential oil components from this work and from the published literature. Chemical compositions were treated as operational taxonomic units (OTUs) using XLSTAT Premium, version 2018.1.1 (Addinsoft, Paris, France). Dissimilarity was determined using Euclidean distance, and clustering was defined using Ward’s method.
Conclusions
This present work surveys the chemical compositions of Z. officinale rhizome essential oils from several locations across Nepal to identify potential sites for quality essential oil for commercialization. As far as we are aware, this is the first report that includes chemical composition, enantiomeric distribution, and antimicrobial activity of Z. officinale rhizome essential oils and showed variation in chemical compositions; the enantiomeric distributions of chiral terpenoids were comparable. The hydrodistillation yields for fresh rhizomes were relatively low ranging from 0.28% to 0.34%. The predominant constituents of Z. officinale essential oil were monoterpene hydrocarbons, oxygenated monoterpenoids, and sesquiterpene hydrocarbons, depending upon their geographic location. The major components in the Z. officinale essential oils were α-zingiberene, camphene, β-phellandrene, neral, geranial, ar-curcumene, and β-sesquiphellandrene. In these Z. officinale samples, camphene, neral, and geranial were detected in significant concentration, which signifies a fresh Z. officinale. Samples G6, G8, G10, G7, and G3 had comparatively high content of α-zingiberene. On the other hand, samples G4, G5, G9, G11, and G12 showed comparatively high content of citral. Based on the content of α-zingiberene and citral (neral and geranial) in respective samples collected from their respective locales might represent ideal Z. officinale rhizome production zones in Nepal. At the same time, G3 and G5 possessed good bacterial activity against P. aeruginosa (MIC = 78.1 μg/mL) and excellent antifungal activity against A. niger (MIC = 39.1 μg/mL). After Z. officinale rhizome production, improvements in harvesting, processing, and extraction procedures may result in higher quality essential oils. The research will surely be useful to choose the right locations for investment and encouragement for farmers and entrepreneurs as well as authentication or adulteration detection of Z. officinale rhizome essential oil for export. Besides these the rhizome essential oil has been used in aromatherapy, to treat nausea, and to alleviate muscular aches and joint discomforts.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221080322 - Supplemental material for Quality Assessment of Zingiber officinale Roscoe Essential Oil from Nepal
Supplemental material, sj-docx-1-npx-10.1177_1934578X221080322 for Quality Assessment of Zingiber officinale Roscoe Essential Oil from Nepal by Darbin Kumar Poudel, Sabita Dangol, Anil Rokaya, Salina Maharjan, Pawan Kumar Ojha, Janaki Rana, Sumitra Dahal, Sujan Timsina and Noura S. Dosoky, Prabodh Satyal, William N. Setzer in Natural Product Communications
Footnotes
Acknowledgments
The authors would like to thank the APRC and ARC for GC-MS and Chiral GC-MS analysis. We acknowledge Rakesh Satyal, Sunita Satyal, and Ambika Satyal for their constructive suggestions and support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Ethical Approval
Ethical approval is not applicable for this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Author Contributions
Conceptualization: P.S. and D.K.P.; methodology: D.K.P., N.S.D., P.S., and W.N.S.; validation: P.S. and W.N.S.; formal analysis: D.K.P., A.R., S.M., and S.D.; investigation: D.K.P., A.R., S.M., SD, N.S.D., and W.N.S.; writing—original draft preparation, D.K.P. and S.M.; writing—review and editing, D.K.P., P.S., and W.N.S.; supervision, P.S.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
