Abstract
A new polyketide, paraconthone A (
Introduction
Marine-derived endophytic fungi refer to harmless parasitic fungi on the internal organs of plants and animals in a diverse marine environment without causing any adverse reactions. Host provided nutrients for endophytes, and endophytes produced bioactive substances giving the host advantage in survival competition.1,2 Symbionts coexist with symptomless fish, sponges, and soft corals that grow in a relatively harsh marine environment, characterized by high salinity, scarce nutrients, and high osmotic and hydraulic pressures, provided many environment-specific microorganisms, which could coevolve with their hosts by undergoing rapid and dynamic change of their genomes. 3 Thus, marine-derived endophytic fungi are considered an important treasure trove of unique complex structural and diverse bioactive metabolites. 2
The genus Paraconiothyrium was discovered as a new genus by Verkley et al in 2004 and 27 species of Paraconiothyrium have been reported to date. 4 However, limited secondary metabolites, including sesquiterpenes, 5 diterpenes, 6 polyketides 7 and aromatic compounds, 8 were isolated from the species in genus Paraconiothyrium. Most of them showed a wide range of biological activities, such as anti-inflammatory, antifungal, cytotoxic, phytotoxic, neuroprotective activities, and inhibiting the liver X receptor and HIV-1.9-11
Previously, we mainly focused on the bioactive metabolites from marine-derived endophytic fungi. In our search for novel fungi-derived metabolites contributing to the development of drugs and the discovery of new pesticides, we systematically study the fermentation extract from strain Paraconiothyrium sp., which was originally isolated from the fiddler crab in Zhoushan island and was identified by tis complete ITS gene sequences. The current study on the chemical constituents of strain Paraconiothyrium sp. Results in the isolation of one new naphthalenone, named paraconthone A (
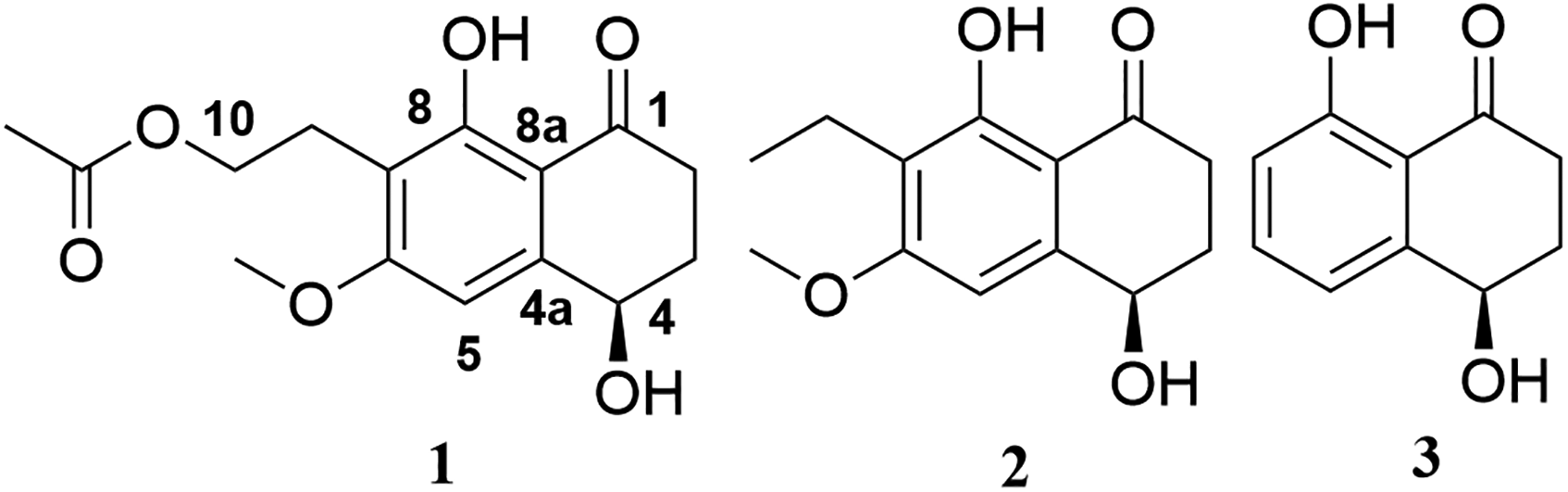
The structures of compounds
Results and Discussion
The Paraconiothyrium sp. Extract was initially chromatographed on a DAD-HPLC column and then further purified by C18 reversed-phase HPLC to yield a new 3,4-dihydro-2H-naphthalen-1-one compound
Paraconthone A (
1H NMR (600 MHz) and 13C NMR (150 MHz) Data of 1 in CDCl3.

the carbon type was assigned by HSQC spectrum.
The planar structure of

HMBC correlations for compound
Furthermore, the absolute configuration of

Experimental and calculated ECD spectra (4R-
Compounds
Experimental
General Experimental Procedures
NMR spectra were acquired on Bruker AVIII-600 NMR spectrometers using tetramethylsilane (TMS) as an internal standard. UV and ECD spectra were recorded on a Shimadzu UV-1800 spectrophotometer and chirascan circular dichroism spectrometers, respectively. The High-Resolution Electrospray Ionization Mass Spectrometry (HR-ESI-MS) was obtained with an Agilent 6529B Q-TOF instrument. The Optical rotation was measured on a JASCO P-1020 polarimeter in MeOH at 25 °C. Preparative high-performance liquid chromatography (Pre-HPLC) was performed by utilizing a Shimadzu LC-20 system equipped with a Shim-pack RP-C18 column (10 μm, 1.5 cm × 25 cm), which was recorded by a binary channel UV detector at 210 and 254 nm with a flow rate at 10 mL/min at room temperature. The Column chromatography (CC) was used with silica gel and ODS on a SepaBen Flash machine.
Fungal Material
The endophytic fungus was isolated from a sample of crab (Chiromantes haematocheir), which collected from the intertidal zone of Zhoushan, Zhejiang, China, in July 2018. The strain was identified as Paraconiothyrium sp. using ITS rDNA (accession number SUB10819600 ITS OL966906) sequence analysis by RuiDi (Shanghai, China) and its DNA sequence using BLAST was compared to the GenBank data. The fungal strain was deposited in the culture collection of the State Key Laboratory of Bioreactor Engineering laboratory, East China University of Science and Technology.
Fermentation, Extraction and Isolation
The fungus Paraconiothyrium sp. was incubated on potato dextrose agar (PDA) medium at 28 °C for approximately 2 to 3 days. Afterward, the agar plugs were cut into three pieces and transferred into 500 mL-Erlenmeyer flasks, containing 200 mL of potato dextrose broth (PDB). The flasks were cultured for two days at 28 °C on a rotary shaker at 180 rpm for inoculation. Then the seed cultures were added to the 20 × 1 L flasks containing 200 mL malt extract medium (1000 mL deionized water contained 20 g malt extract, 20 g sucrose, and 1 g peptone), previously sterilized at 121 °C for 25 min. All flasks were incubated at 28 °C on a rotary shaker at 180 rpm for three days, and then they were transformed into the static culture for fourteen days at 28 °C.
The culture was filtered to give fermentation broth and mycelia, and the mycelia (170 g, wet weight) immersed in methanol (10 L) and then extracted with ultrasound, repeated three times to get a crude extract (5.0 g). The mycelia extract fractionated on silica gel with a stepped gradient elution with petroleum ether-EtOAc (10:1 to 1:1) to give four fractions (A-D) based on the results of TLC analysis. The fraction A (2.8 g) was sequentially fractionated by an ODS column eluting with MeOH-H2O (70% to 95%) to give six fractions (a-f). The subfraction b (130 mg) was sequentially loaded onto silica gel CC (dichloromethane-MeOH, 50:1) and preparative HPLC (MeCN-H2O, 55:45, 10.0 mL/min) to yield compound
Spectroscopic Data
Paraconthone A (
ECD Calculations
The detailed procedures are detailed in the Supporting Information.
Anti–Inflammatory Assays
The human monocytic cell line, THP-1 (Cell Bank of China Science Academy, Shanghai, China) and P. acnes (ATCC6919, Xiangfu biotech, Shanghai, China), were used for the anti-inflammatory assay. THP-1 cells were cultured in RPMI1640 medium with 10% fetal bovine serum (FBS, Gibco, NY, USA) in a humidified incubator (37 °C, 5% CO2). P. acnes bacteria were incubated in Cooked Meat Medium, containing cooked beef granules (Rishui biotechnology, Qingdao, China) in an anaerobic environment. The THP-1 cells were stimulated by the P. acnes, harvested at the exponential phase. The viability of THP-1 cells was evaluated by the MTT assay, specifically, seeding the THP-1 cells in 96-well plates at a density of 2 × 105 cells/well and treated with serially diluted compounds for 36 h (37 °C, 5% CO2). After that, adding 20 µL MTT regent (5 mg/mL, Genetimes Technology Inc., Shanghai, China) to each well and incubating the samples at 37 °C for 4 h. Removing the supernatant, the formazan crystals were fully solubilized in DMSO (150 µL), and the absorbance was measured at 570 nm and 630 nm wavelength. 15 Tretinoin was used as a positive anti-inflammatory control. All the experiments were performed in three independent replicates.
Microorganisms Culture and Anti-Microbial Activities Assay
Minimum Inhibitory Concentration (MIC) assays were used to assess antimicrobial activities of all isolated compounds against two agricultural pathogenic bacteria (Xanthomonas oryzae pv. oryzae and Xanthomonas oryzae pv. oryzicola) and four fish pathogens, Edwardsiella tarda, Vibrio anguillarum, Aeromonas hydrophilia, and Vibrio parahaemolyticus. Chloramphenicol was used as a positive antibacterial control and ketoconazole was used as a positive antifungal control. All the experiments were performed in three independent replicates. 15
Cell Culture and Cell Viability Assay
Four kinds of cell lines including A549, H460, MDA-MB-231 and BT549 were cultured in RPMI-1640 medium (10% FBS, 1% P/S). Cells were incubated at 37 °C with 5% CO2 in a humidified atmosphere. 16 5-Fu was used as a positive cytotoxicity control. All the experiments were performed in three independent replicates.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221075986 - Supplemental material for A New Polyketide from Marine-Derived Paraconiothyrium sp
Supplemental material, sj-docx-1-npx-10.1177_1934578X221075986 for A New Polyketide from Marine-Derived Paraconiothyrium sp by Xing Wei, Yi Ding and Faliang An in Natural Product Communications
Footnotes
Ethical Approval
Ethical Approval is not applicable for this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supporting Information
Experimental procedures, computational details, biological activity determination procedure, and original spectra of compound
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant number 41876189).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
