Abstract
Viral infection imposes a major threat to human health. To develop new potent antiviral agents, Hederagenin (
Introduction
HIV and hepatitis C infections continue to be a major threat to human health worldwide. According to the World Health Organization, there were an estimated 37.7 million people living with HIV at the end of 2020, and hepatitis C killed about 290 000 people in 2019.1,2 Although antiviral therapy has significantly reduced the death rate for both HIV and hepatitis C, drug resistance has emerged to be a drawback for the therapy and new classes of antiviral drugs are needed to combat the drug-resistant strains. 3 A key strategy in developing new drugs is to identify and modify bioactive compounds from natural resources.
The flower buds of Lonicera fulvotomentosa Hsu et S. C. Cheng, a widely used traditional Chinese medicine, have been shown to possess various biological effects, such as antiviral, antimicrobial, and liver-protection activities.4–7 The major active ingredients have been identified to be pentacyclic triterpenes and phenolic compounds, such as aglycones, fulvotomentoside A, chlorogenic acid, and caffeoylquinic acid derivatives. Previous studies have shown that some triterpenes, such as hemiesters of ursolic acid, 3-oxo-tirucalla-7,24-dien-21-oic acid, and 16β-hydroxy-2,3-seco-lup-20(29)-en-2,3-dioic acid, exhibit potent inhibitory activities towards HIV-1 protease (HIV PR).8–12
As a naturally occurring derivative of oleanolic acid, hederagenin (

Numbering of carbon atoms in hederagenin.
Material and Methods
Instruments and Chemicals
An Autopol-1 polarimeter (Rudolph Research Analytical), Bruker Vector-22 FTIR spectrometer (Bruker BioSpin Corporation), Bruker DRX 600 spectrometer (Bruker BioSpin Corporation), VG Auto Spec-3000 spectrometer (Agilent Technologies), and Micro TOF-QII mass spectrometer equipped with an ESI interface (Bruker Daltoniks GmbH) were used to analyze related parameters. Silica gel, octadecylsilane, and silica gel GF254 were used to purify the compounds. All reagents were obtained from Aladdin Chemistry Co. Ltd (Shanghai, P.R. China).
Isolation of HE
Enzyme Assay Kits
Inhibitory activities of the acylated derivatives of
Synthesis
Synthesis of ethyl (3β)-3,23-dihydroxyolean-12-en-28-oate (
Synthesis of Other Compounds
The other compounds were synthesized according to the corresponding methods outlined below.
Synthesis of (3β,23)-di-O-disuccinyl-hederagenin (1a )
(3β,23)-di-O-diglutaryl-hederagenin (1b )
Following the procedure described for
(3β,23)-di-O-disuccinyl-hederagenin ethyl ester (2a )
(3β,23)-di-O-(3′,3′-dimethylsuccinyl-hederagenin ethyl ester [2b ]
Following the procedure described for
(3β,23)-di-O-diglutaryl-hederagenin ethyl ester (2c )
Following the procedure described for
23-t-Butyldimethylsilyloxy-3β-hydroxyl-hederagenin ethyl ester (3a )
23-t-Butyldimethylsilyloxy-3β-O-succinyl-hederagenin ethyl ester (3b )
Following the procedure described for
23-t-Butyldimethylsilyloxy-3β-O-(3’, 3’-dimethylsuccinyl)-hederagenin ethyl ester (3c )
Following the procedure described for
23-t-Butyldimethylsilyloxy-3β-O-glutaryl-hederagenin ethyl ester (3d )
Following the procedure described for
23-Hydroxyl-3β-O-3′,3′-dimethylsuccinyl-hederagenin ethyl ester (4a )
23-Hydroxyl −3β-O-glutaryl-hederagenin ethyl ester (4b )
Following the procedure described for
23-t-Butyldimethylsilyloxy-3-oxo-hederagenin ethyl ester (5a )
PCC (103.0 mg, 0.48 mmol) and
23-Hydroxyl-3-oxo-hederagenin ethyl ester (5b )
Hydrochloric acid (10%, 2 mL) was added dropwise to a solution of
3-Oxo-23-O-succinyl-hederagenin ethyl ester (5c )
Following the procedure described for
3-Oxo-23-O-(3′,3′-dimethylsuccinyl)-hederagenin ethyl ester (5d )
Following the procedure described for
3-Oxo-23-O-glutaryl-hederagenin ethyl ester (5e )
Following the procedure described for
Molecular Docking
To gain insight into how
Results
Preparation of HE Derivatives
Since 3,28-disubstitutions of oleanolic acid can significantly increase the antiviral activity against HIV-1 protease,
8
we decided to investigate the inhibitory activities of

Synthesis of hederagenin derivatives with different C3-acylation and/or C23-acylation. Reagents and conditions: (a) K2CO3, C2H5I, DMF, rt, 12 h; (b) acid anhydride, DMAP, pyridine, reflux, 16 h; (c) TBSCl, DMAP, CH2Cl2, pyridine, 35 °C, 5 h; (d) acetone, 10% HCl, rt, 22 h; (e) PCC, CH2Cl2, rt, 6 h.
Inhibitory Activities of HE Derivatives Against HIV-1 and HCV NS3/4A PRs
Inhibitory activities of
Inhibitory Activities of Acylated Derivatives of Hederagenin Against HIV-1 and HCV NS3/4A Proteases.

HPLC method test.

We further decided to investigate the effect of 23-substitution on the antiviral activity of
Next, we evaluated how the COOH moiety in the 23-substituted functional groups affects the antiviral activity. A series of 23-TBS derivatives of
Finally, we synthesized a series of 3-oxo-23-acyl derivatives of
Molecular Docking and Interaction
Molecular docking showed that (3β,23)-di-O-diglutaryl-hederagenin (
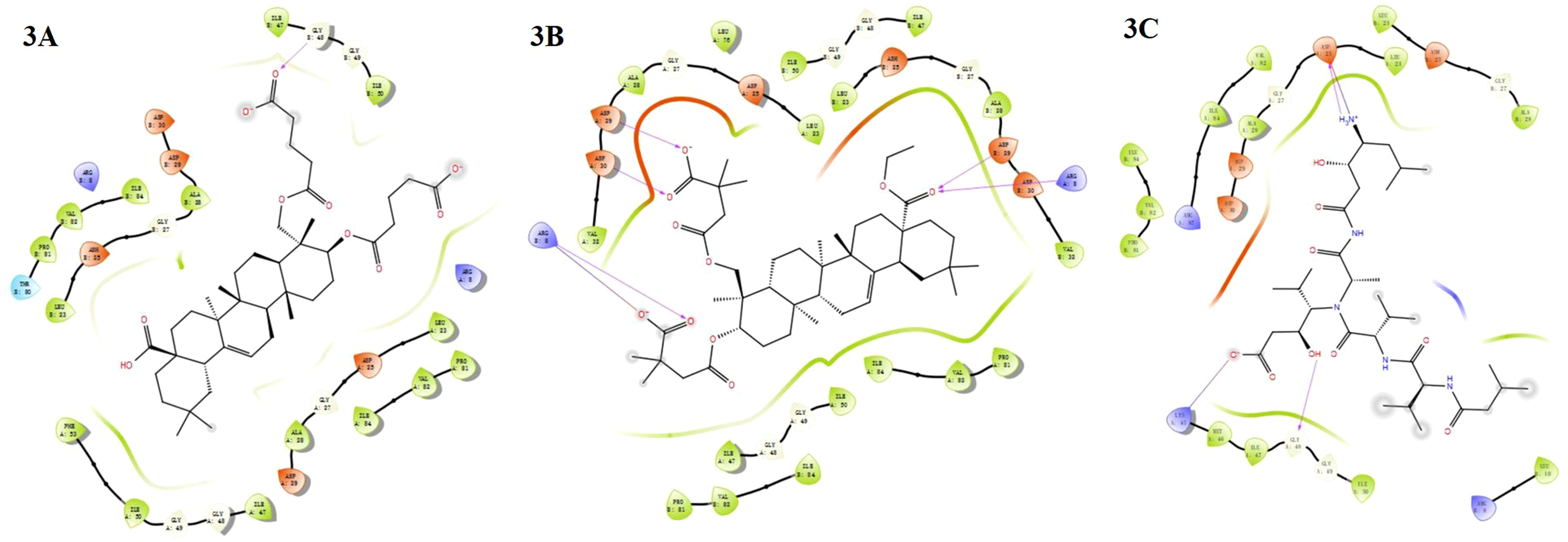
2D Ligand interaction plot highlighting compounds
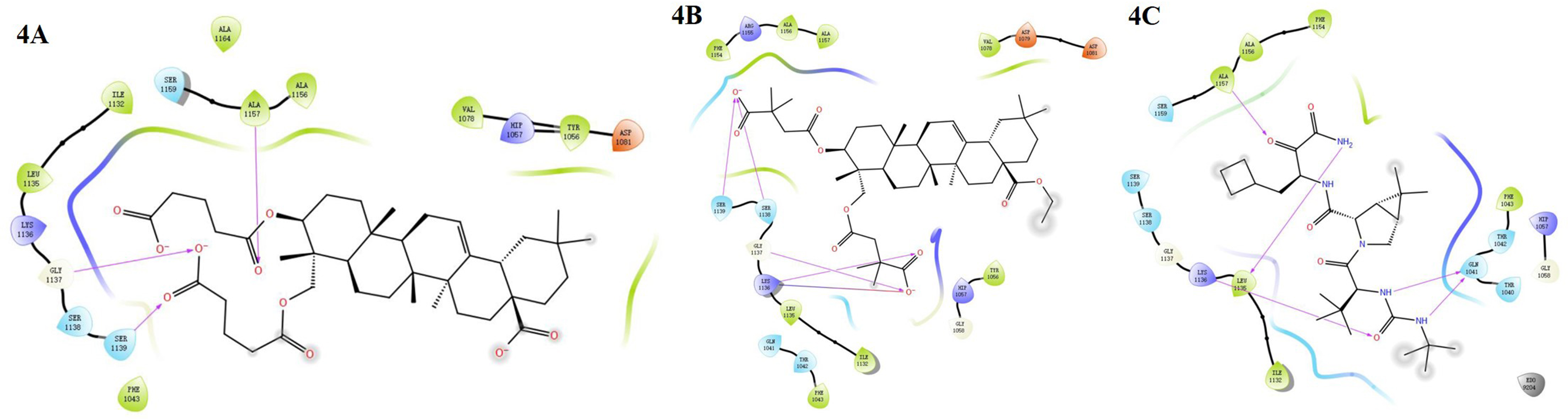
2D ligand interaction plot highlighting compounds
Conclusion
In this study, we synthesized 16 3-acyl and 23-acyl derivatives of either
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the grants from the Association of Natural Science Foundation of China [grant no. 81760758] and Research Center of Molecular Bioactivity from Traditional Chinese Medicine and Ethnomedicine [grant no. 3411-4110000520364].
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

