Abstract
Non-alcoholic fatty liver disease (NAFLD), characterized by seatosis and adipose storage in liver parenchyma cells, is closely related to insulin resistance (IR). Therefore, improvement of IR is one of the important therapeutic measures to treat NAFLD. Insulin sensitizers include thiazolidinediones-induced glucose uptake and lipid synthesis, which may increase weight. Thus, searching for drugs without induction of lipid synthesis is requisite. Adipose tissue is the main storage site for excess energy in the form of triglycerides, which provide up to 70% of the free fatty acids for synthesis of triglycerides in the liver. 1 Under adipose tissue impairment, excess fatty acids are generated, accompanied by ectopic excessive lipid deposition, systemic IR and inflammation. In NAFLD, the number of fatty acids transported from adipose tissue to the triglyceride pool in the liver is more than that of fatty acids generated from de novo synthesis of lipid in the liver. 2 Lipid mobilization is strongly inhibited by insulin under normal physiological conditions. However, with the expansion of fat storage in the liver, free fatty acids are increased and the insulin signal is ameliorated, and then IR is accelerated. Adipose tissue is becoming a kind of mediator in nonalcoholic fatty liver disease and metabolic disease, 3 and a vital target organ for insulin, which stimulates glucose uptake and inhibits lipid lipolysis. IR is closely correlated with liver damage in NAFLD. 4,5 Thus, it is an efficient way to recover the sensitivity of adipose tissue to insulin for alleviating lipid toxicity of the liver.
Liver kinase B1 (LKB1) is involved in inhibition of lipid synthesis and promotion of glucose uptake via phosphorylating Thr-172 of AMPK, which inactivates acetyl-CoA carboxylase via regulating 3-hydroxy-3-methyl glutaryl coenzyme A reductase. 6 A recent study has suggested that AMPK activation could improve metabolic imbalances caused by type Ⅱ diabetes. 7 ROS could activate the JNK pathway via various pathways, the activation of which further phosphorylates serine 307 of IRS-1 to inhibit the transduction of the insulin signal. 8 Phosphorylation of IRS-1 at the serine 307 site is the main mechanism in IR. Besides, insulin receptor substrate (IRS) could be phosphorylated by insulin receptor tyrosine kinase to mediate the transmission of the insulin signal. Frequently, p-IRS binds to the SH2 domain of PI3K regulatory subunit p85, which recruits and activates PI3K. Then, PI3K mediates the activation of AKT, which could regulate glycogen synthesis, gluconeogenesis, and glucose absorption through phosphorylating the corresponding substrates. Thus, ROS/JNK/IRS/PI3K/AKT affects the conversion of glucose to glycogen and protein, and absorption.
Jian-Gan-Xiao-Zhi decoction (JGXZ) is a Traditional Chinese medicine (TCM) that is applied to patients with NAFLD. Clinical research has demonstrated that JGXZ has obvious effects on improving clinical symptoms and blood lipid level in NAFLD patients. 9,10 JGXZ is made up of 12 medicinal herbs; Salvia miltiorrhiza, Curcuma zedoaria, Crataegus pinnatifida, Astragalus membranaceus, Citrus reticulata, Paeoniae Radix Rubra, Curcuma longa, Chrysanthemum, Lotus Leaf, Glycyrrhiza uralensis, Alismatis Rhizoma, and Panax notoginseng root and rhizome. Previous animal experiment has indicated that JGXZ decreases IR and affects AMPK/JNK levels in a non-alcoholic fatty liver rat model. 11 Although TCMs, which are made up of many kinds of substances, are widely used in China, the therapeutic outcomes of patients are not always effective. The unsatisfactory effects of some substances, the uncertainty of the active ingredients, and the poor pharmacokinetic properties greatly restrain the popularization of TCM. Besides, taking an overdose of TCM for a long time might cause certain toxicity reactions, which limits the clinical effects of TCM to some extent. 12
Liquid chromatography-tandem mass spectrometry (LC–MS/MS) has been developed for qualitative and quantitative analysis of components in TCM, including some components with lower content or which are not easily separated. 13,14 Therefore, this technique is important to determine the concentration of main components in serum after TCM administration for further analyzing active components and studying the specific mechanism. This study aimed to investigate the potential mechanism of JGXZ in adipocytes and provide theoretical support for the clinical application of JGXZ. In addition, the concentration of the main components in serum after TCM administration was determined by LC–MS/MS.
Materials and Methods
Preparation of Serum Containing Jian-Gan-Xiao-Zhi (JGXZ) Decoction
Five to 6 week old, Sprague-Dawley (SD) rats, each weighing 180‐200 g, were purchased from Chongqing Shidong Biotechnology Co., Ltd. (China). Jian-Gan-Xiao-Zhi-Fang (JGXZ) consisted of Danshen 15 g, Notoginseng powder 6 g, Zedoary rhizome 15 g, Hawthorn 20 g, Astragalus 20 g, Qingpi 10 g, Red peony root 20 g, Turmeric 12 g Alisma 15 g, Chrysanthemum 15 g, Lotus leaf 15 g, and Liquorice 6 g. SD rats were randomly divided into seven groups (10 in each group, half male and half female), namely, JGXZ group (JGXZ-low dose: 5.43 g/kg, JGXZ-medium dose:10.86 g/kg, JGXZ-high dose: 21.73 g/kg), Metformin group (0.2 mg/kg) and normal saline control group (5 ml 0.9% NaCl). The different doses of JGXZ were calculated according to the table of conversion of equivalent doses of human and animal drugs. 15
The rats were administered orally two times for 3 days. One hours after the last dose, the blood was collected from the abdominal aorta and centrifuged at 3000 r/min for 15 minutes to collect serum. This was inactivated at 56℃ for 30 minutes and filtered to eliminate bacteria using a 0.22 µm microfiltration membrane. HPLC detected the main constituents of JGXZ, including curcumin, notoginseng, astragaloside IV, paeoniflorin, and tanshinone. After passing inspection, the serum was stored at −80℃ until use. The study complied with the National Institutes of Health Regulations, and was approved by the Institutional Animal Care and Use Committee at The First Affiliated Hospital of Yunnan University of Chinese Medicine.
Cells
3T3-L1 preadipocytes were purchased from the Kunming Institute of Animals, Chinese Academy of Sciences. The cells were cultured in DMEM high sugar medium containing 10% fetal bovine serum, and 1% penicillin and streptomycin at 37℃, in 5% CO2. After cell adherence, the medium was replaced. Cells in the logarithmic phase were inoculated on the culture plate, and induced for differentiation 48 hours after cell contact inhibition. Medium containing 0.5 mmol/L IBXM, 1.0 μmol/L DEX, and 10 µg/mL INS was used to replace the original medium, and the cells were cultured for a further 48 hours.
Then, 10 µg/mL INS was added to this medium for incubation 48 hours. The medium was replaced with DMEM to continue the culture of the cells. After 7 d, oil red O was used to stain the fat in cells and determine whether cells were differentiated and mature (Sigma, America). The cells were used for further study once more than 90% cells presented a “round” lipid droplet. The mature adipocytes were divided into control and model groups. The control group was cultured in DMEM medium. The model group was cultured in a medium containing 1 μmol/L DEX to establish the IR adipocyte model. IR adipocytes were cultured in DMEM medium containing 10% JGXE, Rosiglitazone or normal saline (NS) drug-containing serum. After 48 hours of either JGXZ or Rosiglitazone treatment, a glucose content kit (Yan Jin, Shanghai, China) was used to detect glucose levels in the medium for evaluating insulin sensitivity of the cells. The absorbances of the blank, standard, and measured tubes were recorded at 505 nm as A1, A2, and A3, respectively. The glucose contents were calculated by the formula: glucose contents (µmol/mg prot) = (C standard ×V1)×(A3-A1)/(A2-A1)/(V1 ×Cpr)=0.5 × (A3-A1)/(A2-A1)/Cpr (V1 is sample volume, Cpr is protein concentration, C is standard substance concentration).
CCK8 Assay
IR cells were seeded into a 96-well plate (1*105/ml). The serum containing JGXZ was added according to the different groups. Following incubation for 12, 48, or 72 hours, the cell supernatant was abandoned and 10 µL CCK8 solution was added to each well (Beyotime Biotechnology, Shanghai, China). The absorbance at 450 nm was detected.
TC and TG Contents
After either JGXZ or Rosiglitazone treatment, the cells were washed with PBS twice and treated by ultrasound in an ice bath. Then, the prepared cell homogenate was used to detect TC and TG levels using the appropriate kits, according to the manufacturers’ protocols (NanJing JianCheng, China).
Detection of ROS
A cell suspension was prepared at a concentration of 5*104 cells/well. The cells were seeded into a 6-well plate and differentiation was induced. The cell suspension was prepared through digestion with pancreatin for 48 hours after either JGXZ or Rosiglitazone treatment. Then, the cells were centrifuged at 1000 r/min for 5 minutes. After using PBS to wash the cells twice, the cell precipitate was collected by centrifugation. ROS Assay Stain Solution (eBioscience, America) (100 ml) was added to each tube, and the blank group without dye was set as the control. The green fluorescence intensity of DCFH-DA was detected by flow cytometry after incubation at 37℃ for 60 minutes.
Western Blot
To cells collected in an EP tube, protein lysate was added. After centrifuging for 10 minutes at 12 000 r/min at 4℃ for 15 minutes, the supernatant was collected and the protein concentration detected by a BCA method. The target proteins were separated by SDS-PAGE electrophoresis. Blots were blocked by skimmed milk powder solution at 37℃ for 2 hours. Afterwards, the blots were incubated with the primary antibodies at 4℃ overnight. Next, the blots were washed and incubated with secondary antibody. The bands were colored with ECL solution for analysis.
LC-MS/MS Method for Analyzing the Components of JGXZ
The nine main components of JGXZ were determined by LC-MS/MS (Triple Quad™ 5500, Applied Biosystem Sciex, USA). Chromatographic separation was performed on a Phenomenex Luna C18 column (150 mm × 2.0 mm, 3 µm), eluted with a gradient program, as shown in Table 1. The running time for each sample was 8 minutes and the column temperature was kept at 40℃; 0‐4 min belonged to the negative ion detection mode. Analytes were detected with the following massspectrometer source settings. ESI source; ion spray voltage: −4500V; temperature: 500℃; collision gas (N2) pressure: 8 psi; nebulizer gas (N2) pressure: 50 psi; heater gas (N2) pressure: 50 psi; curtain gas (N2) pressure: 35 psi; inlet potential (EP): −10 V; the ion parameters used for quantitative analysis were respectively: notoginsenoside R1 m/z 977.3→931.4, DP: −134, CE: −42; ginsenoside Rg1 m/z 845.3 DP: −166.3 CE: −44.3; paeoniflorin m/z 525.1→449.2 DP: −63.5 CE: −21.1; Internal standard naringin m/z 579.2→271 DP: −214 CE: −44. The results of the ion scan are shown in Supplemental Figure S1; 4-8 min belonged to the positive ion detection mode. Analytes were detected with the following mass spectrometer source settings. ESI source; ion spray voltage: 5500 V; temperature: 500℃; collision gas (N2) pressure: 8 psi; nebulizer gas (N2) pressure: 50 psi; heater gas (N2) pressure: 35 psi; curtain gas (N2) pressure: 35 psi; inlet potential (EP): 10 V; the ion parameters used for quantitative analysis were respectively: ginsenoside Rb1 m/z 1131.5→365.3, declustering potential (DP):219.8, collision energy (CE): 76.9 astragaloside IV m/z 785.6→143.1 DP: 123.2 CE: 16.8; curcumin m/z 369.2→285.2, DP: 87, CE: 21.9; entanshinone m/z 297.2→251.2 DP: 168 CE: 32.2; tanshinone m/z 277.1→249.1 DP: 191.5 CE: 28.9; tanshinone A m/z 295.2→277.2 DP: 174.7 CE: 26.4; Internal standard, cannabidiol m/z 315.2→193.1 DP: 90.6 CE: 28.8. The results of the ion scan are shown in Supplemental Figure S2.
Gradient Program Used for Chromatographic Separation.

Quantitative Ranges and Determination of Nine Compounds in Serum
SD rats (n = 6, half male and half female) were randomly divided into blank (n = 5) and JGXZ (n = 1, marked as 3) groups. Blood was collected from the eye socket and centrifuged at 3000 r/min for 15 minutes. The serum from the blank serum group (100 ml) was placed into a blank centrifuge tube. After vortexing for 30 s, a mixture of methanol-acetonitrile (1:1) (200 µL) was added to the tube. Following further vortexing for 1 minutes, the mixture was centrifuged for 5 minutes at 10 000 r/min and the supernatant (100 ml) was collected, of which 5 µL was used for sample injection. A typical chromatograph is shown in Supplemental Figure S1. The mixture solution of the reference substances (10 µL, 1000 ng/mL) was added to 90 µL of blank serum. Then, 5 μL was injected in the same way as above. A dose equivalent to 32 g/kg of JGXZ was given to rats by gavage twice a day with an interval of 12 hours for three consecutive days. Then, the blood was collected through the abdominal aorta 1 hours after the last JGX administration. The blood was stored at 4 ℃ overnight. Afterwards, the serum was collected through centrifugation at 3000 r/min for 15 minutes and inactivated at 56 ℃ for 30 minutes, then filtered and sterilized by passage through a microporous membrane of 0.22 µm. The serum (100 ml) was placed in a blank centrifuge tube, followed by the addition of the mixed internal standard solution of 10 µL (containing naringin 10 000 ng/mL and cannabidiol 1000 ng/mL). Similarly, a sample injection of 5 µL was obtained through the same way. The quantitative ranges of the 9 compounds in serum were as follows: notoginsenoside R1: 0.5 ~ 50 ng/mL; ginsenoside Rg1: 1 ~ 100 ng/mL; paeoniflorin: 5 ~ 500 ng/mL; ginsenoside Rb1: 10 ~ 1000 ng/mL; astragaloside IV : 0.1 ~ 10 ng/mL; curcumin: 1 ~ 100 ng/mL; cryptotanshinone: 1 ~ 100 ng/mL; tanshinoneⅠ: 0.2 ~ 20 ng/mL; tanshinone Ⅱ A: 2 ~ 200 ng/mL. The dose equivalent to 32 g/kg of JGXZ was given to rats (n = 12, half male and female) by gavage twice a day with an interval of 12 hours for three consecutive days. Then, the blood was collected through the abdominal aorta 1 hour after the last JGXT administration. Since No. 5 rat died after the last administration, its blood was not collected. The concentration of the nine main compounds in the serum containing JGXZ was determined (Table 2).
Concentrations of 9 Main Components of JGXZ in Rat Serum Detected by LC-MS/MS. (N = 12, No. 5 Rat Died).

Statistical Analysis
The experimental data were analyzed using Graph Pad software. P < 0.05 was considered statistically significant.
Results
3T3-L1 IR Adipocyte Model Was Established
The 3T3-L1 preadipocytes were long and fusiform and presented no lipid droplet (LD). After cell differentiation was induced, cells became larger and rounder with LDs appearing in the cytoplasm; these small LDs were the reason for the accumulation of lipid. With the extension of time in cell culture, the small LDs merged into larger LDs. The nucleus and organelles of the mature adipocytes were squeezed to the side of cells by larger LDs. Besides, the cells were round. On the seventh day, oil red 0 staining indicated that large amounts of LDs were stained red in the cytoplasm, which formed a “ring” structure around the nucleus. 95% cells differentiated into mature adipocytes according to random cell counting results (Figure 1(A)). Then, we evaluated the effects of JGXZ on adipocyte viabilities through CCK8 assay. Compared with the control group, cell viabilities showed obvious decreases at 12 hours, 48 hours, and 72 hours (Figure 1(B)). Compared with the model group, JGXZ at different doses markedly enhanced adipocyte viabilities. In addition, cell viabilities presented evident increases after JGXZ administration for 48 hours.

(A) Oil red staining showing preadipocytes differentiation. (B) CCK8 assay analyzed activities of adipocytes treated by different concentration of JGXZ through CCK8 assay analysis. *P < 0.05, ***P < 0.001.
JGXZ Markedly Reduced Lipid Deposition in IR Adipocytes
LD is the organelle closely related to lipid storage, metabolism and secretion, and it consists of a single layer of phospholipid and a hydrophobic core composed of neutral lipids. The hydrophobic core mainly includes TG and cholesterol esters. The results showed that JGXZ significantly lowered the LD levels (Figure 2(A)). The glucose contents in the model group were significantly reduced compared with the control group, implying that cells became insulin-resistant (Figure 2(B)). Furthermore, TG and TC were notably decreased by JGXZ treatment (Figure 2(C–D)). Thus, JGXZ significantly reduced lipid deposition in IR adipocytes. In addition, high-dose JGXZ had similar effects to Rosiglitazone in decreasing lipid deposition.
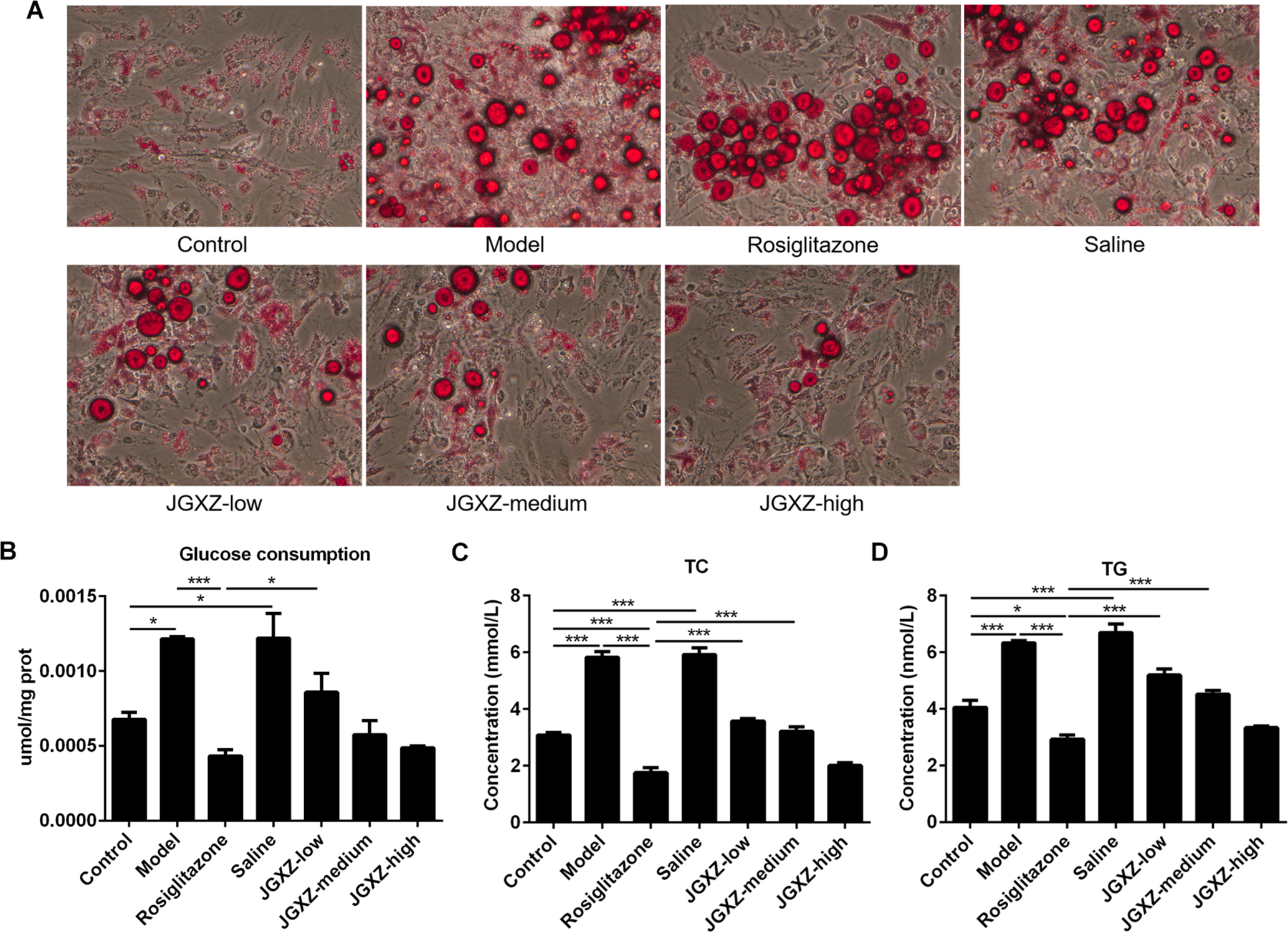
(A) The effects of serum containing drug on lipid accumulation for IR adipocytes. (B) The effects of serum containing drug on glucose consumption for IR adipocytes. (C) The effects of serum containing drug on TC for IR adipocytes. (D) The effects of serum containing drug on TG for IR adipocytes. *P < 0.05, ***P < 0.001.
JGXZ Affected AMPK and ROS/JNK/IRS/PI3k/AKT Levels
After treatment, LKB1 and AMPK were significantly increased by JGXZ and Rosiglitazone, indicating that JGXZ affected lipid deposition and IR partly via LKB1/AMPK. JGXZ at low concentration significantly increased ROS production in IR adipocytes, the effect of which was similar to that of Rosiglitazone treatment (Figure 3(A–B)). ROS levels gradually recovered to the normal with the increase in JGXZ concentration compared with the control group. In addition, the effects of JGXZ with a high concentration on AMPK/JNK/IRS/PI3K/AKT levels were similar to that of Rosiglitazone treatment in IR adipocytes (Figure 3(C)). Moreover, the phosphorylation levels of the Ser-473 and Thr-308 residues were both obviously increased after JGXZ treatment, which could be attributed to the complexity of the JGXZ components. Therefore, the results indicated that JGXZ reduced lipid deposition and IR partly through the ROS/AMPK/JNK pathway.
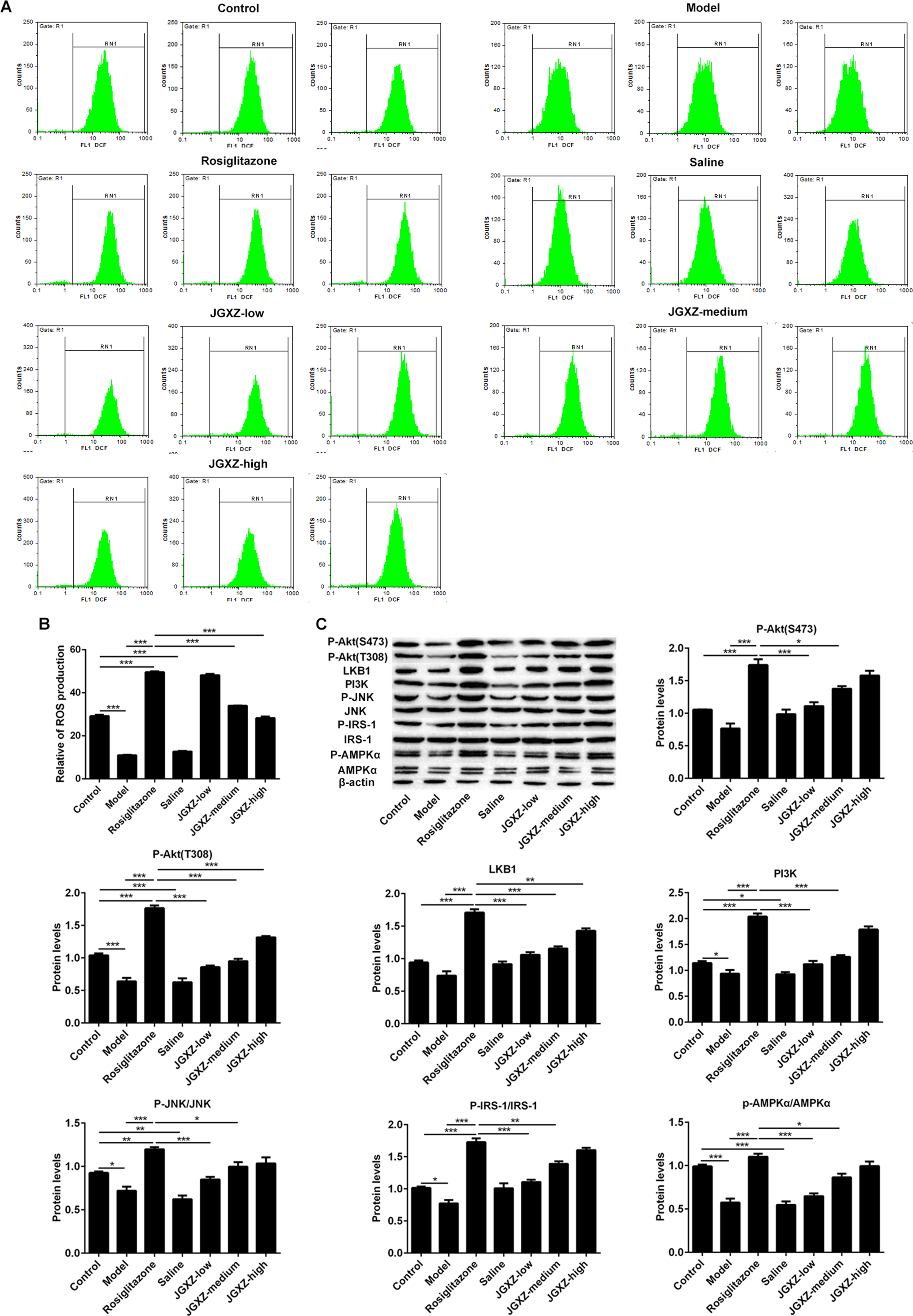
(A-B) ROS levels were detected by flow cytometry after adipocytes were treated with serum containing drug. (C) The effects of serum containing drug on LKB1/AMPK and JNK/IRS/PI3k/AKT levels for IR adipocytes. *P < 0.05, **P < 0.01, ***P < 0.001.
Content of Main Contents in JGXZ in Rat Serum
The LC–MS/MS method was used to determine and quantify nine main ingredients of JGXZ in rat serum. After JGXZ administration for three consecutive days, the concentrations of R1, Rg1, paeoniflorin, Rb1, astragaloside IV, and tanshinone were quantified by LC–MS/MS, as shown in Table 2. Paeoniflorin and Rb1 tended to show high concentrations relative to the other components of JGXZ.
Chromatograms of Nine Analytes
The chromatograms of nine analytes are shown in Supplemental Figure S1 and S2. The serum of the blank group was used to perform chromatographic analysis under anion detection, as exhibited in Supplemental Figure S1(A). The nine analytes and JGXZ are separately presented in Supplemental Figure S1(B and C). The blank serum in the blank group was used to perform chromatographic analysis under cationic mode, as shown in Supplemental Figure S2(A). The chromatograms of serum containing the nine analytes and JGXZ are separately shown in Supplemental Figure S2(B and C).
Content of Nine Components in JGXZ
Since No. 5 rat died after the last administration, its blood was not collected. The concentration of the nine main compounds in serum containing JGXZ was determined (Table 2). Of the nine components in serum containing JGXZ in rats, Rb was the most abundant. However, the concentrations of curcumin, cryptotanshinone and tanshinone were not significant, as detected by LC-MS/MS.
Discussion
Although numerous studies have demonstrated that ROS in mitochondria could not contribute to IR in adipocytes, 16 a study reported that ROS inhibitors could reduce IR caused by chronic insulin treatment. 17 In our study, a high dose of JGXZ significantly reduced ROS levels, accompanied by decreased lipid disposition in adipocytes, the effects of which were reversible using Rosiglitazone. This indicated that except for decreasing IR and increasing glucose uptake, JGXZ could also decrease lipid synthesis, possibly by decreasing ROS levels. We speculated that the complexity of JGXZ components was the main reason. A previous study has suggested that ROS, the main reason of IR in adipocytes, promotes lipid deposition in the liver. 18 Furthermore, ROS has been proved to be closely related to decreased lipid accumulation and ROS suppression in mitochondria-meditated preadipocytes differentiation. 19 -21
ROS is reported to partake in regulating the insulin signal pathway and inducing JNK activation. 17 However, with the increasing concentration of JGXZ, ROS presented a reversal trend with p-JNK levels in our study, which suggested that the JNK pathway was also activated by other molecules apart from ROS. ROS is not only involved in promoting AMPK phosphorylation, but also participates in the activation of AMPK. 22,23 Redox status can modulate AMPK effects and antioxidation-reduced lipid accumulation via activating AMPK in adipocytes. 24 However, the trend of AMPK was different from that of ROS after JGXZ treatment. Thus, JGXZ activated AMPK possibly not merely through ROS mediation. More and more studies showed that AMPK activation could decrease lipid accumulation in adipocytes. 19,25 JGXZ reduced lipid accumulation, possibly partly via the LKB1/AMPK pathway. Compelling evidence has suggested that AMPK activation could phosphorylate ACC, inhibit fatty acid synthesis and promote fatty acid oxidation. 26,27 Besides, insulin signaling was significantly increased in the IRS/AKT pathway by JGXZ, indicating that JGXZ recovered the sensitivity of adipocytes to insulin. In our study, JGXZ could significantly reduce DEX-induced IR in adipocytes and increase glucose uptake, possibly through AMPK, ROS and the JNK/IRS/PI3K/AKT pathway.
To evaluate blood concentration of JGXZ in rats, a LC-MS/MS method was used. It was observed that the concentrations of R1, Rg1, paeoniflorin, Rb1, astragaloside IV and tanshinone in rat serum could be determined by LC-MS/MS, implying that they could play important roles in IR adipocytes induced by DEX. Some research has demonstrated that some active components of JGXZ have obvious effects on decreasing lipid levels in NFALD. A study demonstrated that Rg1 and Rb1 were involved in improving lipid levels and ameliorating inflammation in NAFLD mice. 28 Paeoniflorin has obvious effects on suppressing lipid ectopic deposition, enhancing insulin sensitivities and decreasing oxidation in NAFLD rats. 29,30 Astragaloside has obvious anti-lipid accumulation effects through regulating the AMPK signal in NAFLD cell models. 30,31 Taken together, JGXZ possibly exerted significant effects on decreasing glucose and lipid deposition mainly through these components. The results are essential for JGXZ use in the clinic and further study of the effects of these components in NAFLD.
Conclusions
JGXZ decreased glucose and lipid deposition by regulating the ROS/LKB1/AMPK and JNK/IRS/PI3K/AKT pathways. R1, Rg1, paeoniflorin, Rb1, astragaloside IV and tanshinone were detected by LC-MS/MS. JGXZ plays a pivotal role in NAFLD, and thus it is important for us to understand the mechanism of JGXZ in the treatment of this disease.
Supplemental Material
Figure S1 - Supplemental material for The Mechanism of Jian-Gan-Xiao-Zhi Decoction in Insulin Resistant Adipocytes and Its Component Analysis
Supplemental material, Figure S1, for The Mechanism of Jian-Gan-Xiao-Zhi Decoction in Insulin Resistant Adipocytes and Its Component Analysis by Fang Fang, Wei-Bo Wen, Xue-Hua Xie, Ling Yang, Xu Zhang and Jie Zhao in Natural Product Communications
Supplemental Material
Figure S2 - Supplemental material for The Mechanism of Jian-Gan-Xiao-Zhi Decoction in Insulin Resistant Adipocytes and Its Component Analysis
Supplemental material, Figure S2, for The Mechanism of Jian-Gan-Xiao-Zhi Decoction in Insulin Resistant Adipocytes and Its Component Analysis by Fang Fang, Wei-Bo Wen, Xue-Hua Xie, Ling Yang, Xu Zhang and Jie Zhao in Natural Product Communications
Footnotes
Statement of Human and Animal Rights
All of the experimental procedures involving animals were conducted in accordance with the institutional animal care guidelines of The First Affiliated Hospital of Yunnan University of Chinese Medicine, China and approved by the Administration Committee of Experimental Animals, Yunnan Province, China.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (The Study of Jiangan Xiaozhi Recipe Improving Non-alcoholic Fatty Liver Disease by Regulating Insulin Resistance on Basis of AMPK/ROS/JNK Signaling Pathway, 81560772).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
