Abstract
The antimicrobial activity and the underlying action mechanisms of dihydrotanshinone I against Staphylococcus aureus, methicillin-resistant Staphylococcus aureus, extended-spectrum beta-lactamases Staphylococcus aureus were investigated with Kleihauer-Betke (K-B) test. The antibacterial mechanisms of dihydrotanshinone I were investigated by monitoring the changes in electric conductivity, concentration of AKP, protein content, and patterns of protein electrophoretic bands in sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE). The antibacterial rings showed that antimicrobial activity of dihydrotanshinone I at 18 mM was stronger to Staphylococcus aureus than to methicillin-resistant Staphylococcus aureus and extended-spectrum beta-lactamases Staphylococcus aureus. The minimum inhibitory concentration (MIC) and IC50 values showed that dihydrotanshinone I had the strongest inhibitory activity against S. aureus (MIC = 280 µM, IC50 = 874 ± 0.01 µM, respectively). Dihydrotanshinone I could increase the electric conductivity, concentration of alkaline phosphatase (AKP) and protein content. The patterns of protein bands in SDS-PAGE were changed obviously. Dihydrotanshinone I also significantly inhibited S. aureus, methicillin-resistant S. aureus, and extended-spectrum beta-lactamases S. aureus, indicating that dihydrotanshinone I can damage the structures of cell wall and cell membrane to increase permeability of cell membrane and release of cell components. Dihydrotanshinone I could influence the synthesis of bacterial protein, destroy the protein, or reject the anabolism or expression of the protein, and finally lead to the loss of normal physiological function of bacteria.
Staphylococcus aureus (SA) is a common human infection bacterium, which easily causes various diseases. It mostly exists in human skin, especially in nasopharynx. 1 When the host’s immune function is low, SA can invade the body and cause respiratory tract infection. 2 In the severe cases, septicemia may occur. The first case of methicillin-resistant Staphylococcus aureus (MRSA) infection was reported in 1961. 3 MRSA infection increased rapidly and spread all over the world. Due to the change of drug resistance of multidrug-resistant strains, antibacterial resistance has become an important problem in clinical treatment. At the present, the main drug-resistant bacteria include MRSA, penicillin-resistant Streptococcus pneumoniae (PRSP), vancomycin-resistant Enterococcus (VRE), and vancomycin-resistant Staphylococcus aureus (VRSA). In particular, MRSA has a wide range of distribution, rapid transmission and high mortality. 4 According to the China bacterial resistance monitoring network in 2018, the proportion of SA in clinical distribution was 9.0%, which was the fourth largest clinical infection strain. 5 In August 2016, China put forward the national action plan to curb bacterial drug resistance (2016, 2020), aiming to formulate the comprehensive management strategies and measures at the national level, and to strengthen the management of antibacterial drugs and curb bacterial resistance. 6 In addition to bacteria, there are also viruses that seriously threaten human health, for example COVID-19. 7 -9 COVID-19 is mainly spread by direct transmission, aerosol transmission, and contact transmission. Unlike a bacterial infection, but viral infection makes a big difference. 10 -12
Dihydrotanshinone I is one of the main liposoluble components of tanshinone. It possesses good activity and shows many pharmacological effects. 13,14 Research showed that dihydrotanshinone I could reduce the levels of total cholesterol (TC) and triglyceride (TG) in serum and liver. 15 It was reported to inhibit the growth of K562 cells and lymphocyte leukemia cells (P388) in vitro in a time- and dose-dependent maner. 16 Dihydrotanshinone I was identified as one of the main antitumor active ingredients of Salvia miltiorrhiza Bge, which inhibited the growth of tumors and enhanced the radiosensitivity in vivo. 17,18 In addition, dihydrotanshinone I had a competitively inhibitory effect on protein tyrosine phosphatase 1B, enhancing the sensitivity of insulin action. It also had a therapeutic effect on type II diabetes. 19 It has been reported in the literatures that the total ketone of S. miltiorrhiza has strong antibacterial activity against Gram-positive cocci, especially S. aureus (SA). Further studies confirmed that cryptotanshinone and dihydrotanshinone I had the strongest antimicrobial activity in total tanshinone in vitro. 20 Luo et al. reported that dihydrotanshinone I had different inhibitory effects on mycobacterium 607, human tuberculosis bacillus H37RV, and mycobacterium ulcerans. 21
Dihydrotanshinone I has good clinical prospects as an antimicrobial agent. However, there is no report on the antibacterial mechanism of dihydrotanshinone I. In this paper, dihydrotanshinone I was investigated to elucidate the antibacterial mechanism by detecting the changes in cell wall and cell membrane electrical conductivity, alkaline phosphatase (AKP), extracellular proteins, and changes in pattern of protein expression/synthesis revealed via sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE).
Material and Methods
Material
Staphlococcus aureus 25923 was purchased from Shanghai Tiancheng Bio-information and Technology Co., Ltd., (Shanghai, P. R. China). MRSA and Extended spectrum beta-lactamase-SA (ESBLs-SA) were provided by Huaihe Hospital (Kaifeng, Henan, P. R. China), and identified by VITEK-AMS (Automated Microbic System). The coincidence rate was 99%. The SAs were activated and inoculated into Broth Agar Medium.
Dihydrotanshinone I (purity HPLC >98%) was purchased from Sichuan Vickie Biotechnology Co., Ltd (Sichuan, P. R. China). Alkaline phosphatase assay kit and Coomassie Brilliant Blue Protein assay kit were purchased from Nanjing Jiancheng Bioengineering Company (Nanjing, Jiangsu, P. R. China).
Bacterial Culture
The 3 S. aureuss trains were activated and inoculated into broth agar medium at 37℃ for 24 hours in a thermostat, and then bacterial concentration was diluted with sterile broth agar to 106 CFU/mL.
The Antibacterial Activity in Vitro
Determination of Bacteriostasis Circle, Minimum Inhibit Concentration (MIC) and 50% inhibiting concentration (IC50).
According to literature, 22,23 the K-B method was used to determine antibacterial activity. The half inhibitory concentration (IC50) was determined by liquid culture method.
The Antibacterial Mechanism of Dihydrotanshinone I in Vitro
The effect on the bacterial cell wall 24 . Dihydrotanshinone I solutions at MIC and 3 × MIC were added to bacterial suspension. Sterile water was used as the negative control group. All the groups were cultured in incubator at 37 ℃ and time sampling method was adopted. Then the testing objects were centrifuged at 3500 rpm for 10 minutes, the content of alkaline phosphatase (AKP) in supernatant was measured by the AKP kit assay with time.
The effect on cell membrane 25 . Dihydrotanshinone I solutions at MIC and 3 × MIC were mixed with bacterial suspension. Sterile water was used as the negative control group. The bacterial suspension was cultured at 37 ℃ and sampled regularly to determine the conductivity.
Determination of Soluble Protein
Dihydrotanshinone I solutions at MIC and 3 × MIC were mixed with bacterial suspension. Sterile water was used as the negative control group. The bacterial suspension was cultured at 37 ℃ and sampled regularly to determine. Coomassie Brilliant Blue Color Reagent was added and let it standing for 10 minutes. OD value at 595 nm (OD595) was determined.
SDS-PAGE Analysis of Bacterial Proteins 26
The dihydrotanshinone I solution at MIC was added to logarithmic growth period bacterial suspension and the mixed suspension was cultivated at 37 ℃. Sterile water was used as the negative control. After the samples were diluted and adjusted into the same OD value, the precipitations were obtained by centrifuge, and then 80 µL of sterile water and 20 µL of 5 × SDS sample buffer precipitation were added, mixed thoroughly and boiled in a water bath at 100 °C for 5 minutes, the solution was centrifuged again and supernatant on standby was collected and used as protein extracts.
Results
Diameter of Antibacterial Rings, MIC and IC50 of Dihydrotanshinone I
The inhibitory zone of dihydrotanshinone I to SA (12 mm) at 18 mM were larger than those of the MRSA (10 mm) and ESBLs-SA (10 mm). MIC showed the best activity to SA, MIC = 280 µM. In liquid culture method, the order of activities for three bacterial strains was as follows: SA (IC50 = 874 ± 0.01 µM)>ESBLS SA (IC50 = 1.0x103 ± 0.01 µM)>MRSA (IC50 = 1.9x103 ± 0.03 µM). (Table 1)
Diameter of Antibacterial Rings, MIC, and IC50 of Dihydrotanshinone I Against SA, MRSA, and ESBLS-SA.
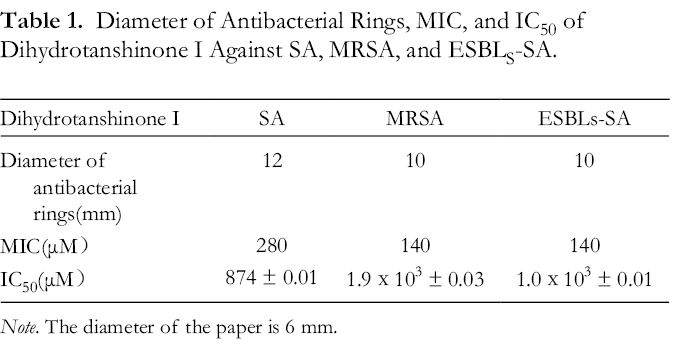
Note. The diameter of the paper is 6 mm.
Effects of Dihydrotanshinone I on Cell Walls of Three Staphylococcus Aureus Strains
Alkaline phosphatase (AKP) exists between cell wall and cell membrane. When the cell wall is intact, AKP can not be detected outside the cell wall. But when the cell wall is destroyed, AKP will leak out of the cell wall. Therefore, the change of AKP can indirectly reflect the effect of dihydrotanshinone I on the cell wall permeability. 27 -29
Dihydrotanshinone I could significantly increase the extracellular AKP contents of SA, MRSA, and ESBLS-SA (Figure 1). The obvious change was seen from 1 to 2 hours, and was proportional to the concentration of dihydrotanshinone I. That is, the higher the concentration was, the higher the detected AKP content. At the same concentration, the effect of dihydrotanshinone I on AKP of ESBLS-SA was significantly weaker than those of SA and MRSA. It may be due to the reason that the resistance of ESBLs-SA on dihydrotanshinone I is greater than those of SA and MRSA. In Figure 1, dihydrotanshinone I can destroy the integrity of cell walls of SA, MRSA, ESBLs-SA, and increase the content of extracellular AKP. The higher the Dihydrotanshinone I concentration was, the stronger the destructive force of the cell wall.

The activity of AKP extra cellular against SA, MRSA, ESBLs-SA.
Effect of Dihydrotanshinone I on Cell Membrane Permeability
When the microorganism is in an unfavorable environment or is poisoned by the drug, it tends to cause the decrease in the stability and fluidity of the biofilm. At this time, a large number of electrolytes in the cell will leak out, such as K+. The change of conductivity of bacterial fluid reflects the change of permeability of bacterial cell membranes. The loss of these ions can lead to the blockage of many metabolic pathways and the influence of the activities of many enzymes. On the other hand, the regulation of osmotic pressure inside and outside cells is also affected by the fluidity and semi-permeability of cell membranes. The loss of the regulatory function of the osmotic pressure will damage the cell’s imbibition and rupture or even death. 30,31
The conductivity of the bacterial suspension added with dihydrotanshinone I at 0 hours was higher than that of the control group. It is suggested that the charged component of dihydrotanshinone I could increase the conductivity or destroy bacteria rapidly. With the prolongation of time, dihydrotanshinone I could gradually increase the conductivity of the bacterial suspension. It was higher than that of the control group, and the higher the concentration was, the greater the conductivity was. Its action duration on three kinds of S. aureus was concentrated in 60 minutes (Figure 2). It indicated that dihydrotanshinone I could destroy the cell membranes of SA, MRSA and ESBLS-SA. It affected the structure and composition of cells, increased the permeability of cell membranes, and continuously exuded electrolytes, leading to the increased conductivity. 32

The change of electrical conductivity against SA, MRSA, and ESBLs-SA.
Effects of Dihydrotanshinone I on the Contents of Extracellular Proteins
The concentrations of soluble proteins were increased with increasing the concentrations of dihydrotanshinone I (Figure 3). Protein yield was higher in bacterial solution treated with dihydrotanshinone I at 3 × MIC than at 1 × MIC. It indicated that dihydrotanshinone I could damage to bacteria and lead to exclusion of soluble proteins, which could, in turn, result in offsetting parts of the proteins consumed by bacteria. After 1 hours of treatment of SA and ESBLs-SA and 2.5 hours of MRSA with dihydrotanshinone I, the protein contents in bacterial fluid were decreased gradually. It may be due to the reason that after being inhibited, the bacterial somatic cells could stimulate their own self-repair mechanism, resulting in a decrease in protein leakage or self-repair for a period of time. A small number of bacteria can resume their viability, continue to survive and reproduce under suitable conditions, and the protein in the bacterial fluid will be consumed by the growing bacterial cells. However, the amount of exuded protein is not adequate to offset the protein consumption in the bacterial solution.

The change of the soluble protein concentration against SA, MRSA, and ESBLs-SA.
Changes in Pattern of Bacterial Proteins Revealed by SDS-PAGE Analysis
In Figure 4(A), six bands in patterns of SA were not changed significantly at 3 hours. The obvious changes of R-S1 (43 KDa) and R-S2 (43 KDa-34 KDa) bands were observed at 6 hours and 9 hours, which were shallower than those of the control. It indicated that dihydrotanshinone I inhibited the normal metabolism of SA protein to some extent. It was inferred that dihydrotanshinone I hindered the protein expression or the normal synthesis of proteins.

SDS-PAGE patterns of SA, MRSA, and ESBLs-SA affected by treatment with dihydrotanshinone I (A: Lanes1, 3, and 5, the normal electrophoretic bands of SA for 3 hours, 6 hours, and 9 hours; Lanes 2, 4, and 6: the electrophoretic bands of SA affected by treatment with dihydrotanshinone I for 3 hours, 6 hours, and 9 hours; B: Lanes 1, 3, and 5: the normal electrophoretic bands of MRSA treated with dihydrotanshinone I for 3 hours, 6 hours, and 9 hours; Lanes 2, 4, and 6: the electrophoretic bands of MRSA affected by treatment with dihydrotanshinone I for 3 hours, 6 hours, and 9 hours and C: Lanes 1, 3, and 5: the normal electrophoretic bands of ESBLs-SA treated with dihydrotanshinone I for 3 hours, 6 hours, and 9 hours; Lanes 2, 4, and 6: the electrophoretic bands of ESBLs-SA affected by treatment with dihydrotanshinone I for 3 hours, 6 hours, and 9 hours.
As shown in Figure 4(B), the densities of six bands in pattern of MRSA became shallower or absent at 3 hours, 6 hours, and 9 hours as compared with those of the control. The changes of R-M2 (72 KDa-55 KDa), R-M3 (55 KDa-43 KDa), and R-M4 (43 KDa) bands were more obvious at 6 hours and 9 hours than at 3 hours, and their densities were shallower than those of the corresponding ones in the controls. It is inferred that dihydrotanshinone I could affect the normal metabolism of MRSA protein, hinder the synthesis and expression of MRSA protein or the normal protein synthesis of MRSA, and decrease the total expression level of MRSA protein. The density of R-M5 (34 KDa-26 KDa) band was deepened at 3 hours as compared with the corresponding one in the control, suggesting that MRSA may stimulate its own self-repair mechanism and accelerate the synthesis of such proteins after being stimulated with dihydrotanshinone I.
In Figure 4(C), six bands in pattern of ESBLs-SA showed that the bands of R-E1 (130 KDa), R-E2 (95 KDa-72 KDa), R-E3 (55 KDa), and R-E4 (34 KDa-26 KDa) were changed obviously. R-E1 (130 KDa) bands were absent or shallower as compared with those of the control at 3 hours, 6 hours, and 9 hours. It was inferred that dihydrotanshinone I affected and hindered the expression and synthesis of proteins in ESBLs-SA, and reduced the synthesis of such proteins. The densities of bands of R-E3 (72 KDa-55 KDa) and R-E4 (34 KDa-26 KDa) became deeper than those of corresponding bands in the control at 3 hours and 6 hours. It may be due to the reason that ESBLs-SA stimulated the self-healing mechanism of life within the first 6 hours. This leads to an increasing synthesis of these proteins to meet the needs of bacterial self-repair. Compared with the control group, ESBLs-SA in the control group did not change in 9 hours. It was inferred that with the prolonging the time, ESBLs-SA in the control group grew and multiplied continuously under suitable conditions. After a period of self-repairing, the unaffected bacterial cells continued to grow and multiply, and the synthesis of such proteins increased correspondingly.
Discussion
The basis of antibacterial research of natural products needs to provide scientific basis through the study of antibacterial mechanism. Due to the complexity of the active components of traditional Chinese medicine, the relatively fewer researches on antibacterial mechanism have been conducted and reported, especially those on the antibacterial mechanism of the active components separated by activity tracking are much fewer. 33 In our group’s previous studies, He et al. isolated chelerythrine from the root of Toddalia asiatica; Ma et al. isolated hydroquinone from Ainsliaea bonatii Beauv and found that it could significantly change the morphology of SA. 24,25 Li et al. found that punicalagin affected the cell wall integrity and cell membrane permeability of SA, and inhibited the bacterial protein synthesis to a certain extent. 34 Qin et al. found that rhein inhibited the growth and reproduction of bacteria by damaging their cell membrane. 35 Using isobaric tags for relative and absolute quantitation (iTRAQ) technology, Wang et al. found that after treatment with juglone, RpmJ, RpIN, and RpmG2 proteins related to the ribosomal structure of SA were down-regulated, suggesting that juglone destroyed the ribosomal structure of bacteria and prevented the normal protein synthesis. 36 Qiu and other studies demonstrated that baicalin could directly act on α-hemolysin itself. 37 Huang et al. found that flavonol kaempferol could bind PriA and inhibited bacterial reproduction. 38 Together, these studies indicate that the naturally-derived active components are capable of inhibiting bacterial protein synthesis to certain extend.
In this study, we studied the antimicrobial activity and underlying action mechanism of dihydrotanshinone I. The inhibition circle showed that dihydrotanshinone I at 18 Mm caused more stronger inhibition on SA than on MRSA and ESBLS-SA. The MIC value and IC50 value showed that its inhibitory activity on SA with MIC of 280 µM and IC50 of 874 ± 0.01 µM was the strongest. The results also showed that the conductivity, AKP, and soluble protein content of the 3 staphylococcus aureus were increased after the action of dihydrotanshinone I. SDS-PAGE electrophoresis showed that the densities of protein bands were also changed significantly. Based on these results, the underlying action mechanism of dihydrotanshinone I can be proposed as follows (Figure 5): dihydrotanshinone I inhibits bacterial growth and multiply, possible via destroying the structure of bacterial cell wall and cell membrane, leading to the increased permeability of cell membrane, and in turn making the cellular contents to be leaked out. At the same time, it can be speculated that dihydrotanshinone I has a certain effect on the synthesis of bacterial protein, reduce the protein in bacteria, and affect and hinder the expression of intracellular proteins. Ultimately, the normal physiological functions of bacteria are damaged or lost.
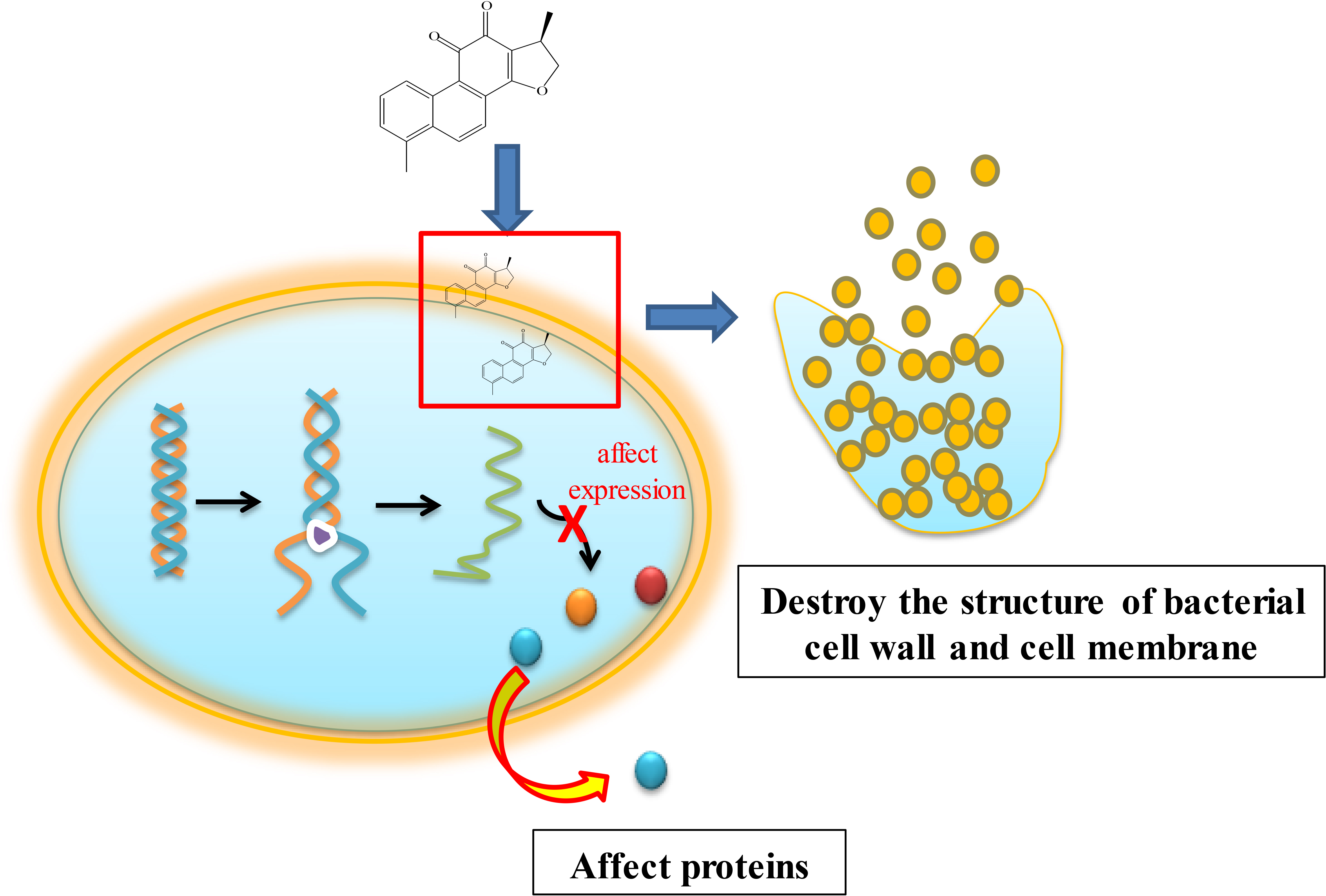
Proposed model illustrating the antimicrobial activity and possibly underlying action mechanisms of dihydrotanshinone I.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Science and Technology Development Program of Henan Province (212102310354), Key Scientific Research Projects of Higher Education Institutions in Henan Province (21B360001), College Students’ Innovative Entrepreneurial Training of Henan University Minsheng College (MSCXSY2019036, MSCXSY2020078), and College Students’ Innovative Entrepreneurial Training of Henan Province (S202013501009, 201910475107).
