Abstract
In continuation of our study on Cotinus coggygria from Serbia, 10 known flavonoids (
Introduction
The genus Cotinus (family Anacardiaceae) comprises 8 species, namely Cotinus coggygria Scop. (syn. Rhus cotinus L.), Cotinus obovatus Raf., Cotinus americana Nutt., Cotinus carranzae Rzed. and Calderon, Cotinus chiangii (D. A. Young) Rzed. and Calderón, Cotinus kanaka (R. N. De) D. Chandra, Cotinus nanus W. W. Sm., and Cotinus szechuanensis Pénzes.
1
C. coggygria, a shrubby tree, also known as the European smoketree, is native to a large area from southern Europe to northern China, inhabiting mainly dry slopes, rocky and gravely areas in forest clearings, and south-facing limestone soils. In Serbia, C. coggygria is widely distributed, especially on limestone and sedimentary rocks and in the forests of black hornbeam and black pine. In folk medicine of various countries, as well as in Serbia, this plant is used for the treatment of different health problems. Extracts of the various parts (leaves, flowers, heartwood, and bark) and essential oils obtained from leaves of C. coggygria showed a broad spectrum of pharmacological activities, including antioxidant, anticancer, antigenotoxic, antimicrobial, antiviral, hepatoprotective, and antiinflammatory.
2
Flavonoids are the most important and abundant group of biologically active constituents of Cotinus species, followed by phenolic acids, sterols, and other secondary metabolites. Previously reported compounds isolated from the wood of C coggygria are sulfuretin, fustin, 3-O-methyl-2,3-trans-fustin, 3-O-galloyl-2,3-trans-fustin, fisetin, garbanzol, eriodyctiol, butein, butin, taxifolin, quercetin, catechin, liquiritigenin, isoliquiritigenin, disulfuretin, sulfurein, gallic acid, methyl gallate, pentagalloyl glucose, β-resorcylic acid, gallic acid, gallic acid methyl ester, 3-O-β-sitosterol glucoside, fisetinidol-(4α→8)-(+)-catechin, and epifisetinidol-(4β→8)-(+)-catechin, a dimer of butin.
5
In continuation of our examination of C. coggygria from Serbia4,9 and searching for compounds with a possible antigenotoxic activity,
10
we now report the evaluation of the DNA protective activity of the flavonoids
The CBMN assay used in this study is a technique that is widely recognized as one of the most reliable and successful assays for genotoxic studies. The recording of the frequency of MN, commonly used as a cytogenetic biomarker (and MN expression in peripheral blood lymphocytes) is well established as a method for monitoring chromosome damage in human populations. The method originally proposed by Countryman and Heddle 14 was subsequently improved with the development of CBMN assay, which has been extensively used to evaluate chromosome damage in human populations. 15 This technique, a couple of years ago endorsed by the Organisation for Economic Co-operation and Development, is considered one of the most robust methods for assessing cytotoxicity and genotoxicity. 18 The application of CBMN assay for antioxidant evaluation of heterocyclic compounds, including flavonoids (until 2011), is reviewed elsewhere. 13
An important effect of flavonoids is the scavenging of oxygen-derived free radicals. This has been the subject of several studies in the past few years, and important structure–activity relationships of the antioxidant activity have been established. 19 Antioxidants protect against molecular damage in living organisms from reactive oxygen species (ROS), such as DNA damage altering gene expression, cell growth, and differentiation.20,21 As oxidative DNA damage is considered to be a pathogenic event in the induction of many illnesses (eg, Alzheimer's and Parkinson's diseases, cancer, and diabetes), 22 decreasing the degree of such damage by plant polyphenols may reduce the risks.
Results and Discussion
Compounds

Flavonoids from Cotinus coggygria that were tested for DNA protective activity.

Decrease in frequency of MN (%) in comparison with the control (100%) (see Table 1) effected by flavonoids
Incidence of MN, Cytokinesis-Block Proliferation index, Distribution of MN per Cells, and Frequency of MN Measured in Cell Cultures of Human Lymphocytes Treated with Different Concentrations of the Isolated Compounds From Cotinus coggygria.
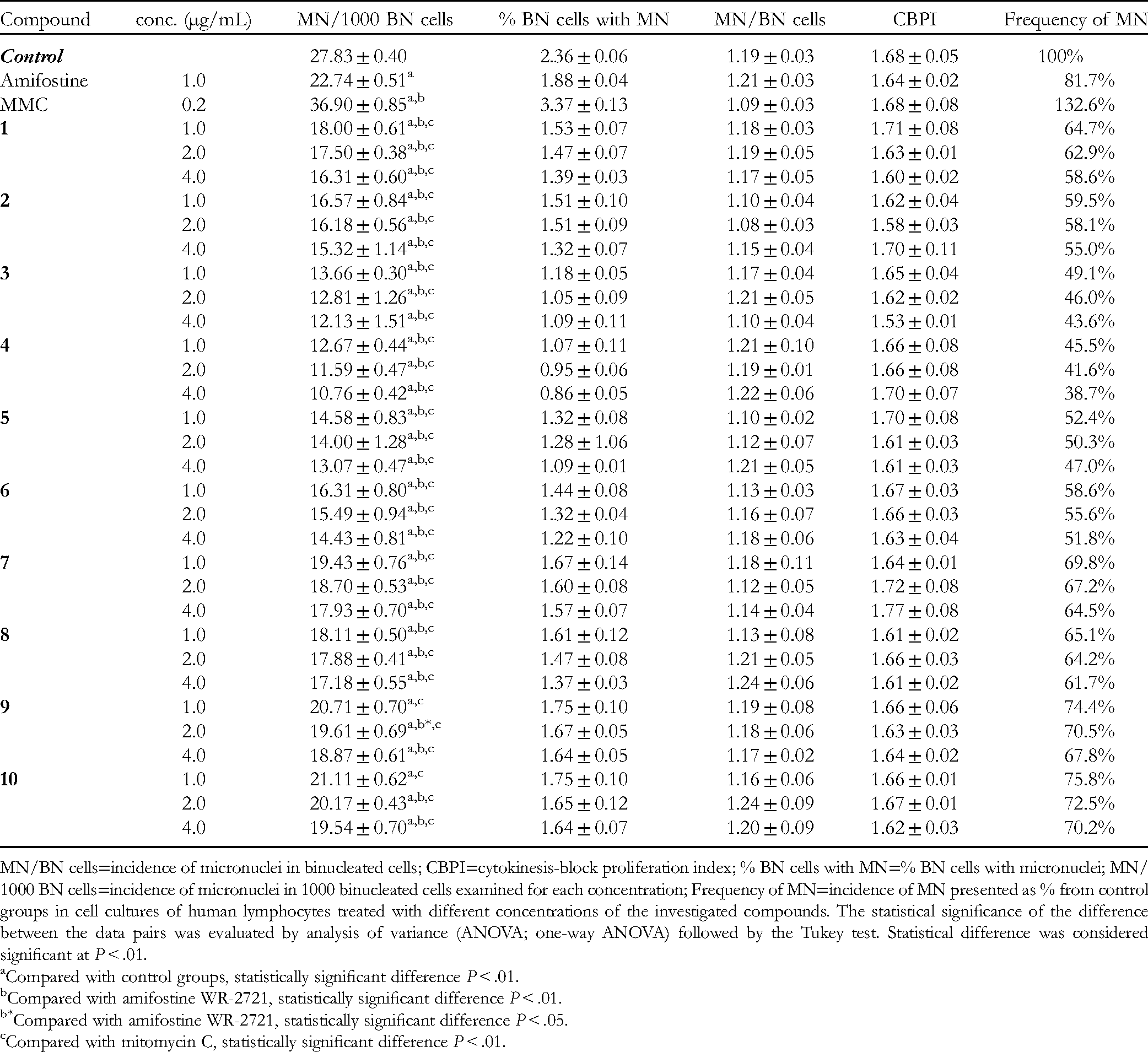
MN/BN cells=incidence of micronuclei in binucleated cells; CBPI=cytokinesis-block proliferation index; % BN cells with MN=% BN cells with micronuclei; MN/1000 BN cells=incidence of micronuclei in 1000 binucleated cells examined for each concentration; Frequency of MN=incidence of MN presented as % from control groups in cell cultures of human lymphocytes treated with different concentrations of the investigated compounds. The statistical significance of the difference between the data pairs was evaluated by analysis of variance (ANOVA; one-way ANOVA) followed by the Tukey test. Statistical difference was considered significant at P < .01.
Compared with control groups, statistically significant difference P < .01.
Compared with amifostine WR-2721, statistically significant difference P < .01.
Compared with amifostine WR-2721, statistically significant difference P < .05.
Compared with mitomycin C, statistically significant difference P < .01.
The majority of the studied compounds, occurring in many plant species, belong to the group of pharmacologically active metabolites with a wide spectrum of biological activities such as anticancer, antioxidant, antiinflammatory, and antiviral.
25
All tested flavonoids, except
The CBMN assay of
All the tested compounds (in minimal doses of 1.0 μg/mL) decreased the DNA damage of human lymphocytes in the range of 24.2% to 54.5%, more effectively than amifostine at the same concentration. Among the tested compounds, eriodictyol (
Butein (
Sulfuretin (
Cotinignan A (
Conclusion
In this study, the DNA protective activity is presented of 10 known flavonoid compounds from C. coggygria. All of the investigated compounds exhibited better activity than the radio protectant amifostine, while eriodyctiol was the best protector, decreasing the MN frequency by 54.5%. Functional groups responsible for the antioxidant activity of flavonoids, 3′,4′-dihydroxyphenyl (catechol), 5-OH, 3-OH, and 4-keto (3-keto in aurones) are supposed to influence DNA protective activity in CBMN assay as well. Further investigation, including DNA protective activity against irradiation damage, will give a wider picture of the protective activity of C. coggygria flavonoids.
Experimental
General Experimental Procedures
For CC, Merck silica gel (Si gel) (particle size 0.063-0.200 mm), methanol, methylene chloride, and chloroform were used. Preparative TLC (Prep TLC) was performed on glass plates precoated with silica gel 60 (particle size <0.063 mm) with a layer thickness of 0.75 mm. Analytical TLC was performed on aluminum plates precoated with Merck silica gel 60 F254 (0.25 mm thickness). The nuclear magnetic resonance (NMR) spectra were obtained on a Bruker Avance III 500 (500 MHz for 1H; 125 MHz for 13C), in CD3OD as a solvent. Chemical shifts (δ) were expressed in ppm and coupling constants (J) in hertz (Hz). Semipreparative reversed phased HPLC was performed on an Agilent Technologies 1100 Series HPLC-DAD, and a Zorbax Eclipse XDB C18 column (150 × 9.4 mm, 5 μm) was used.
Plant Material, Extraction, Isolation, and Identification of Compounds
The C. coggygria heartwood was collected at Deliblatska Peščara (Deliblato Sand), Vojvodina province, Serbia, in June 2018. Plant material was identified by Professor Milan Veljić, Faculty of Biology, University of Belgrade, and voucher specimen BEOU 17422 was deposited at the Herbarium of the Institute of Botany and Botanical Garden “Jevremovac”, Belgrade, Serbia.
The heartwood was air-dried and milled. Wood powder, 100.0 g, was extracted 3 times with 1 L of CH2Cl2/MeOH 2:1 for 24 h (the first hour of extraction in an ultrasonic bath) at room temperature to give 8.2 g of crude extract, which was subjected to fractionation by Si gel CC.
Isolation was performed similarly to our previous work.
9
Crude extract (8.2 g) was chromatographed on a Si gel column (750 × 45 mm), with CH2Cl2/MeOH (gradient elution, from 95/5 to 60/40) and afforded 230 fractions. The eluates were monitored by TLC, and the fractions with similar retention factor values were combined. Pure compounds were isolated from these fractions by additional Si gel CC and/or Prep TLC and semipreparative HPLC. All isolated compounds were identified using 1H and 13C NMR spectroscopy, and comparison to NMR data from our previous work.
9
Compounds
Subjects
Venous blood samples were obtained by heparinized sterile vacutainers (Becton Dickinson) from 6 healthy nonsmoking male volunteers who had not been exposed to chemicals, drugs, or other substances. A safety protocol concerning blood-borne pathogens/biohazards was adopted. The volunteers gave their permission for using their blood for the experiment. From each subject, 2 aliquots of blood, 5 mL each were obtained. The study complied with the code of ethics of the World Medical Association (Helsinki Declaration of 1964, as revised in 2002). The blood samples were obtained at the Medical Unit in accordance with current Health and Ethical regulations in Serbia, Law on Health Care (2005).
CBMN Assay
The culture lymphocytes were treated with compounds
About 2 × 106 blood lymphocytes were set up in 5 mL of RPMI-1640 medium supplemented with 15% calf serum and 2.4 μg/mL of phytohemagglutinin (Invitrogen-Gibco-BRL). One hour after the start of cell stimulation, the investigated compounds were added to the samples at a final concentration of 1.0, 2.0, or 4.0 μg/mL, respectively. The incidence of spontaneously occurring MN in control samples was scored. For MN preparation, the cytokinesis block method of Fenech was used, with some modifications. 14 Cytochalasin B (Invitrogen-Gibco-BRL), at a final concentration of 6.0 μg/mL, was added to the samples after 44 h, and the lymphocyte cultures were incubated for a further 24 h. After 72 h, the cells were washed with 0.9% NaCl (Merck, Sharp, & Dohme GMBH), collected by centrifugation, and treated with a hypotonic solution at 37 °C. The hypotonic solution consisted of 0.56% KCl + 0.9% NaCl (mixed in equal volumes). The cell suspension was prefixed in methanol/acetic acid (3:1), washed 3 times with fixative, and dropped onto a clean slide. 16 Slides were air-dried and stained with alkaline Giemsa (Sigma-Aldrich) (2%). At least 1000 binucleated (BN) cells per sample were scored, registering MN according to the criteria of Countryman and Heddle 14 and Fenech.15,16
Initially, we investigated the effects of isolated compounds on cell proliferation by determining the cytokinesis-block proliferation index (CBPI). Since micronucleus expression is dependent on cell proliferation, quantification of cell proliferation and cell death should be carried out to obtain a sound evaluation of cell kinetics and micronucleus frequencies.
The CBPI was calculated as suggested by Surralle et al,
17
that is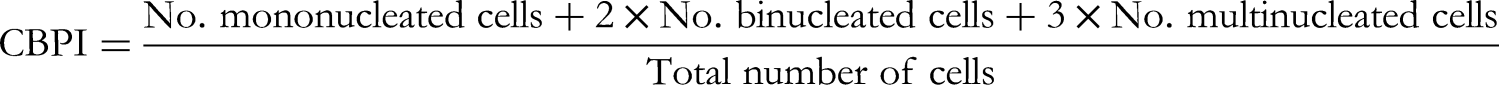
The numbers of BN cells with 1, 2, 3, or more MN were then tabulated. The data for each treatment were expressed as the frequency of MN per 1000 BN cells.
Statistical Analysis
The statistical analysis was performed using Origin software package version 7.0. The statistical significance of the difference between the data pairs was evaluated by analysis of variance (ANOVA; one-way ANOVA), followed by the Tukey test. Statistical difference was considered significant at P < .01. The index calculated is presented as the % of change comparing different groups.
Footnotes
Acknowledgments
The authors would like to thank the Ministry of Education, Science and Technological Development of the Republic of Serbia (contract numbers: 451-03-9/2021-14/200026, 451-03-9/2021-14/200168) and the Serbian Academy of Sciences and Arts (Grant No. F188).
Ethical Approval
The study complied with the code of ethics of the World Medical Association (Helsinki Declaration of 1964, as revised in 2002). The blood samples were obtained at the Medical Unit in accordance with the current health and ethical regulations in Serbia (Parliament of the Republic of Serbia. Law on Health Care. Official Gazette of the Republic of Serbia 2005; 107: 112-161)
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the Law on Health Care, Parliament of the Republic of Serbia Official Gazette of the Republic of Serbia 2005; 107: 112-161.
Statement of Informed Consent
Verbal informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Education, Science and Technological Development of the Republic of Serbia (contract numbers: 451-03-9/2021-14/200026 and 451-03-9/2021-14/200168) and the Serbian Academy of Sciences and Arts (Grant No. F188).
