Abstract
The anti-cancer activity of bannaxanthone E isolated from Garcinia mckeaniana leaves was assessed through a flow cytometric method on human lung SK-LU-1 cancer cells, including cell cycle changes and induction of cell apoptosis. Treatment with bannaxanthone E led to the suppression of cell cycle progression at the G2/M phase to 19.6% at 4 µM, and induced apoptosis via cell morphological changes, increased the fluorescence signal in caspase-3/7 activation, and accumulation of apoptotic cells in the SK-LU-1 line at 4 µM to 25.7%.
Introduction
Lung cancer or bronchogenic carcinoma, referring to tumors originating in the lung parenchyma or within bronchi, is the most commonly diagnosed cancer worldwide, at 11.4%. This type of cancer is responsible for the most cancer-related deaths, accounting for about 18% of the total cancer deaths, and this burden continues to increase. 1 In Vietnam, lung cancer is the second most frequent cancer in both genders, which is one of the current leading causes of death in males. 2 Thereby, there is a clear and urgent need for novel anticancer agents with improved efficacy for the treatment of lung cancer.
Xanthones, which are typical constituents of lichens, microorganisms and higher plants, is one of the biggest classes of oxygenated heterocyclic compounds in natural product chemistry. 3 The growing interest in this class of compounds has been associated with their pharmacological and biological properties.4–6 For example, bannaxanthone E, first isolated from the stem of the plant Garcinia xipshuanbannaensis in 2008, showed cytotoxic, antimicrobial and antimalarial activities.7–9 Interestingly, xanthone analogs are the components of some antioxidant drugs such as Vimang® (Mangifera indica) 10 and Xango® (Garcinia mangostana). 11 Dimethylxanthenone-4-acetic acid (DMXAA) underwent clinical trials as an antivascular agent. 12
G. mckeaniana is one of thirty Garcinia species recorded in Vietnam. 13 In our previous study, eleven xanthone analogs were isolated from this plant and their cytotoxic activity against different human cancer cell lines (KB, HepG2, SK-LU-1, and MCF7) have been reported.14,15 Of these compounds, bannaxanthone E (Figure 1) exhibited strong anti-proliferative activity against SK-LU-1 cells with an IC50 value of 3.92 ± 0.5 µM. Accordingly, with the aim to develop novel anticancer agents, the anticancer activity of bannaxanthone E has been further studied. Its effects on cell cycle arrest and apoptosis in the SK-LU-1 cancer cell line are reported herein.

The chemical structure of bannaxanthone E.
Results and Discussion
Cell Cycle Analysis
Cell cycle dysregulation is a cardinal feature of cancer cells. Cell cycle arrest is regarded as an effective approach for eliminating cancer cells. 16 As bannaxanthone E exhibited anti-proliferative efficacy in human lung cancer cells (SK-LU-1), 14 its effect on cell cycle progression has been assessed by monitoring the distribution of cells in populations, as described in Table 1 and Figure 2. Treatment with bannaxanthone E at concentrations of 2 µM and 4 µM caused a rise in the percentages of cells from 25.6% (control) to 32.8% and 35.7%, respectively, in the S phase, and from 7.0% (control) to 10.6% and 19.6%, respectively, in the G2/M phase. In addition, a reduced number of cells was observed in the G0/G1 phases compared with the negative control. These results suggested that bannaxanthone E possessed anti-proliferative activity through suppressing cell-cycle progression at the G2/M phase.

Effect of bannaxanthone E on cell cycle distribution in SK-LU-1 cells. Cells were treated with 2 µM and 4 µM bannaxanthone E, DMSO (control), and 2 µM ellipticine for 24 h. Cell cycle distribution was evaluated by flow cytometry Novocyte.
Percentage of Cells by Phases After Bannaxanthone E, Ellipticine, and DMSO Treatment.

Apoptotic Inductive Activities
Apoptosis, a programmed cell death, is induced by multiple factors such as cellular stress, DNA damage and immune surveillance. 17 The ability of tumor cells to elude apoptosis is a hallmark of most types of cancer. Besides that, G2/M phase arrest prevents cells from exiting mitosis and undergoes apoptosis, a feature shared by microtubule inhibiting agents such as colchicine or vincristine. 18 Since bannaxanthone E exhibited potential anti-proliferative activity and selectively suppressed cell cycle progression at different phases, its effect on apoptosis was evaluated.
Morphology
Staining cells with Hoechst stain makes the chromatin condensation and nuclear fragmentation, which are morphological hallmarks of apoptosis in the nucleus, visible by fluorescence microscopy. 19 The effect of the tested compound on the morphology of SK-LU-1 cells was observed by an Inverted Microscope Zessis. Whereas the shape of cells treated with the control had an interlaced polygonal shape, the cells treated with the test compound became rounded and shrunken. This indicated a direct interaction of the tested compound with the cytoskeleton.
As illustrated in Figure 3, treatment with bannaxanthone E caused a dose-dependent rise in the number of apoptotic cells. The photomicrograph of the apoptotic changes in SK-LU-1 cells treated with the test compound exhibited nuclei predominantly fragmented and stained more intensely because of chromatin condensation, which is one of the early events of apoptosis. The fragmented nuclei and blue fluorescence increased in a concentration-dependent manner. Budding on the plasma membrane was observed, as well as on some cells entering the late stages of apoptosis. Additionally, treatment with the test compound at the higher concentration led to growth inhibition of the cancer cells. The lower concentration of the test compound also caused a decrease in cancer cell number. Bannaxanthone E was a potent compound which induced chromatin condensation and nuclear fragmentation.
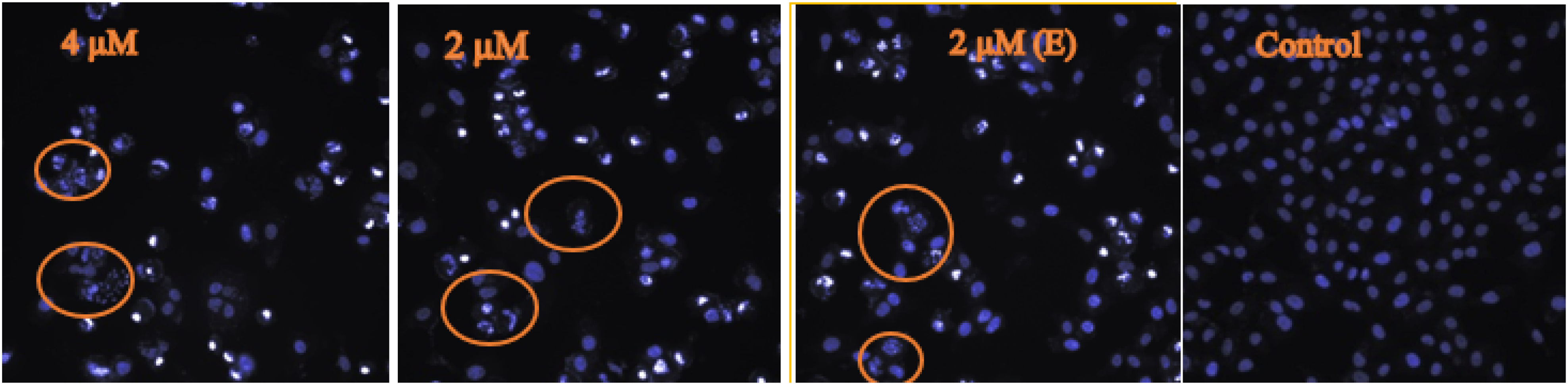
Photomicrograph of apoptotic changes in SK-LU-1 cells treated with 2 µM and 4 µM of bannaxanthone E, 2 µM of ellipticine, and DMSO (control). Cells with fragmented nuclei are enclosed within circles.
Caspase-3/7 Enzymatic Activities
Caspase-3/7 belongs to a family of protease enzymes playing essential roles in programmed cell death, and in coordinating the destruction of cellular structures such as DNA fragmentation, and degradation of cytoskeletal proteins. 20 When the cell receives some death signal, the effector caspase 3/7 is activated, resulting in the degradation of deoxyribonucleic acid (DNA). 21 The morphological modifications which occur in the apoptotic cells are also promoted by caspases. 22 Bannaxanthone E exhibited this effect on the morphology of SK-LU-1 cells, thus its effect on caspase-3/7 activity was subsequently evaluated. A significant increase in enzyme activation of cells was observed. As shown in Table 2 and Figure 4, the fluorescence signal of bannaxanthone E is higher than that of ellipticine at the same 2 µM concentration. The test compound activated caspase 3/7 in a dose-dependent manner. The results clearly showed that bannaxanthone E was able to induce apoptosis by activating the caspase pathway.

Effect of 2 µM and 4 µM bannaxanthone E, 2 µM ellipticine, and DMSO (control) on caspase-3/7 activity. The relative fluorescence units (RFU), directly proportional to the activation of caspase-3/7, was measured by a Tecan GENios Pro microplate reader.
Effect of Bannaxanthone E and Ellipticine on Caspase-3/7 Activity in SK-LU-1 Cells.

Immunophenotyping by Flow Cytometry Analysis
The translocation of phosphatidylserine to the exposed membrane surface is an early event in apoptosis. 23 Annexins, which have a very high affinity for membranes containing the negatively charged phospholipid phosphatidylserine, can be used as a marker of early apoptosis events. In order to assess the potential apoptotic effect of bannaxanthone E, a flow cytometric assay using Annexin V-FITC was performed using the SK-LU-1 cell line. The experiments were conducted for the evaluation of apoptosis in cells treated for 24 h. As indicated in Figure 5, the cells treated with bannaxanthone E at concentrations of 2 µM, and 4 µM led to an accumulation of apoptotic cells from 2.12% (control) to 11.1%, and 25.7%, respectively. In the 24 h treatment, an increase in apoptosis gave us a reason to suppose that the cells previously in early apoptosis had shifted to the cell death process. The results demonstrated the ability of bannaxanthone E to exert apoptosis in a dose-dependent manner, particularly in the early stage of apoptosis in SK-LU-1 cells.

Analyses of apoptosis induction in SK-LU-1 cells. Detection of apoptotic cells after annexin-V/PI staining by flow cytometry. Cells were treated with 2 µM and 4 µM bannaxanthone E, 2 µM ellipticine, and DMSO (control), followed by harvesting and staining with annexin-V/PI for 24 h.
Experimental
General
Hoechst 33342 (Sigma) was dissolved in DMSO at a concentration of 10 mg/mL and then stored at −20 °C in a dark place. Annexin V kits and PI/dead cell apoptosis® were from Invitrogen, and ApoONE® homogeneous caspase-3/7 kits from Promega, USA. Ellipticine was used as a reference.
Equipment for biological assays: Inverted microscope Zessis, fluorescence microscope Olympus Ex 350 nm/Em 460 nm, spectrophotometer Biotek, spectrofluorometer Tecan GENios Pro Ex 485 nm /Em 530 nm, flow cytometry Novocyte (ACEA Biosciences, San Diego, CA, USA).
Plant Material, Extraction and Isolation
The plant material, extraction and isolation are the same as described in our previous work. 14
Cell Line and Culture Conditions
The human lung cancer cell line (SK-LU-1 ATCC HTB-57TM) was purchased from the American Type Culture Collection (ATCC, Manassas, VA, USA), stored in liquid nitrogen, and cultured in DMEM medium with 7 to 10% fetal bovine serum (Gibco, USA), and 1% penicillin and streptomycin (Gibco, USA) in a humidified atmosphere with 5% CO2 at 37 °C. The cells in the log phase were preferentially used for the tests.
Cell Cycle Analysis
SK-LU-1 cells were seeded in 25 cm2 flasks (Corning, USA) at a density of 3 × 105 cells. Bannaxanthone E, at concentrations of 2 µM and 4 µM, DMSO 0.5% (negative control) and ellipticine (2 µM) were incubated with cells for 24 h. The cells were then harvested, centrifuged at 2000 rpm for 5 min, washed 3 times with PBS, and fixed with ethanol 70% for 4 h at −20 oC. The treated cells were further washed, diluted with 0.45 mL PBS, added to RnaseA (1 mg/mL), and incubated at 37 °C for 15 min. Then, 25 µL PI (1 mg/mL) was added for 30 min and diluted with 500 µL PBS. Then, samples were analyzed for apoptosis using flow cytometry Novocyte with data analyzed and reported by ACEA's Novo Express software. 24
Cell Nucleus Staining with Hoechst 33342
A suspension of SK-LU-1 cancer cells (190 mL) was grown in a 96-well plate at 37 °C for 24 h and then incubated with bannaxanthone E at concentrations of 8 µM, 4 µM, 2 µM and 1 µM. Ellipticine at 4 µM and 2 µM was used as a reference. DMSO 0.5% was used as a negative control. After 24 h of incubation, the cells were fixed with formaldehyde 4%. After 30 min, the cells were washed again with PBS and stained with Hoechst 33342 (1 µg/mL) for 10 min. Cells were observed by fluorescence microscopy at an excitation wavelength of 350 nm and an emission wavelength of 461 nm. 24 Apoptotic cells are defined as cells with either brighter nuclei (due to condensed chromatin) or nuclei that are fragmented.
Caspase-3/7 Assays
Caspase 3/7 activities were evaluated using an Apo-ONE Homogeneous Caspase-3/7 Assay kit. 25 Fifty µL of SK-LU-1 cells were grown on a 96-well plate with cell concentration of 2 × 105 cells/well. Then, 50 µL of bannaxanthone E at concentrations of 4 µM and 2 µM, and ellipticine at 2 µM were added to the wells. The mixture was incubated in a humidified atmosphere of 5% CO2 at 37 °C for 5 h. Next, 100 µL of Apo-ONE® caspase-3/7 reagent was pipetted into each well. A plate shaker was used to mix gently the contents of the wells at 350 rpm for 30 s, and then the plate was stored at room temperature for 1 to 18 h. A Tecan GENios Pro microplate reader was used to measure the relative fluorescence units (RFU) at wavelengths of Ex 385 nm/Em 530 nm. The activation of caspase-3/7, directly proportional to fluorescence, was calculated by the following formula: caspase activity = RFU value of tested sample − RFU value of the blank.
Annexin V FITC and PI Flow Cytometry Assay
The experiment was performed using an annexin V kit and PI/dead cell apoptosis® from Invitrogen. 24 Three × 105 cells were incubated with bannaxanthone E at concentrations of 2 µM and 4 µM, DMSO 0.5% (negative control) and ellipticine (2 µM) for 24 h, then collected into falcon tubes. After centrifugation, the cells were washed with PBS. The cell residue was redissolved in 100 µL binding buffer and added to 5 µL annexin V and 1 µL PI (100 µg/mL). Incubation was continued for 15 min at 37 °C; then, 400 µL binding buffer was added to the cell tubes. A total of 15,000 cells of each sample was analyzed using a Novocyte flow cytometry system to determine the proportion of apoptosis-expressing cells.
Conclusions
The effects of bannaxanthone E isolated from Garcinia mckeaniana on the cell cycle and apoptosis have been evaluated by flow cytometric analysis. The results show that bannaxanthone E has potential anti-proliferation activity due to its ability to induce G2/M phase arrest, morphological changes, activation of caspase 3/7, and an increase in the early stage apoptosis of SK-LU-1 cells. These findings illustrate the promising anticancer activity of bannaxanthone E against SK-LU-1 cancer cells, and provide a relevant basis for the development of xanthones as agents for cancer prevention and for combination therapy with anticancer drugs.
Footnotes
Acknowledgments
The authors are indebted to the Institute of Chemistry (Institute of Chemistry Project 2021).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Institute of Chemistry (grant number Project 2021).
