Abstract
Ziziphus joazeiro Mart., popularly known as “juazeiro”, is a tree widely found in the northeast of Brazil. It is commonly used as an anti-inflammatory, antibacterial, antifungal, and analgesic agent. The stem extract exhibited, beside cytotoxic properties, substantial activity against the Gram-negative bacterium Allivibrio fischeri. UHPLC-ESI-Orbitrap-HR-MS analysis of the alkaloidal fraction of the crude methanolic stem extract of this species enabled the detection and putative identification of sixteen cyclopeptide alkaloids (CPAs), including four possibly new structures. According to the MS2 fragmentation analysis, from the sixteen identified CPAs, three possess a type-Ia1, one a type-Ia2, and twelve a type-Ib cyclopeptide alkaloid core. The structures of paliurine-C and -D were supported by NMR data.
Introduction
Cyclopeptide alkaloids (CPAs) are macrocyclic compounds (13-15 members), defined as heterocyclopeptides due to the presence of a styrylaminine moiety. 1 They are commonly found in the Rhamnaceae family, especially in Ziziphus species, but are also present in other families like Asteraceae, Celastraceae, Euphorbiaceae, and Rubiaceae. According to their ring characteristics, CPAs are divided into subtypes. These compounds have been reported as being active on the central nervous system, e.g. as an analgesic, in addition to anti-inflammatory, antibacterial, antimycobacterial, antifungal, antiplasmodial, and immunostimulant properties. This class of secondary metabolites is found in almost all plant parts, like roots, stems, seeds, fruits, and flowers, but most commonly in the bark derived from roots or stems.2,3
Ziziphus joazeiro Mart., popularly known as “juazeiro”, is a tree that often occurs in the northeast of Brazil (Caatinga biome). Its leaves and stem-bark are used in folk medicine against fever, bacterial and fungal infections, chronic bronchitis, and gingivitis, while its stem is used to treat fungal mouth infection, flu and tooth decay.4-6 Phytochemical investigations led to the isolation and identification of several compounds, especially triterpenes (lupane-, ceanothane- and dammarane-type), flavonoids and phenolic derivatives.5-9 Despite the high number of phytochemical studies with Z. joazeiro, there are only few studies describing the isolation and structural elucidation of cyclopeptide alkaloids from this species (nummularine-K, zoazerine and nummularine-M).10,11 Triggered by the widespread use of Z. joazeiro and following up on our earlier analytical and synthetic work on such ansa-type cyclopeptide alkaloids from Rhamnaceae and Ziziphus spp. in general, we take a deeper look on such compounds for this species.9,12-15
Results and Discussion
To characterize cyclopeptide alkaloids, the alkaloidal fraction of the crude methanolic extract of stems from Z. joazeiro was analyzed by UHPLC-ESI-Orbitrap-HR-MS. It is noteworthy that in most cases, a high amount of botanical material is necessary to obtain feasible amounts of isolated compounds to evaluate its biological activities.16,17
The positive ion UHPLC-ESI-HR-MS base peak chromatogram (Figure 1) shows approximately twenty peaks. Among them, sixteen cyclopeptide alkaloids (CPA) could be detected and putatively identified 18 based on their elemental composition and fragmentation behavior. The detailed results are summarized in Table 1.

UHPLC-ESI-HR-MS base peak chromatogram of crude alkaloidal fraction of Ziziphus joazeiro (positive ion mode, full MS m/z 100-2000, retention time window 7.7-10.7 min). Compounds were numbered in order of increasing retention time.
Ia and Ib-Type Cyclopeptide Alkaloids Characterized in the Crude Alkaloid Fraction From the Stem of Ziziphus joazeiro.

Same as compound number (Figure 2).
Of non ionized compound.
The fragmentation patterns of compounds

Ia and Ib-type cyclopeptide alkaloids annotated in the crude alkaloid fraction of Ziziphus joazeiro.
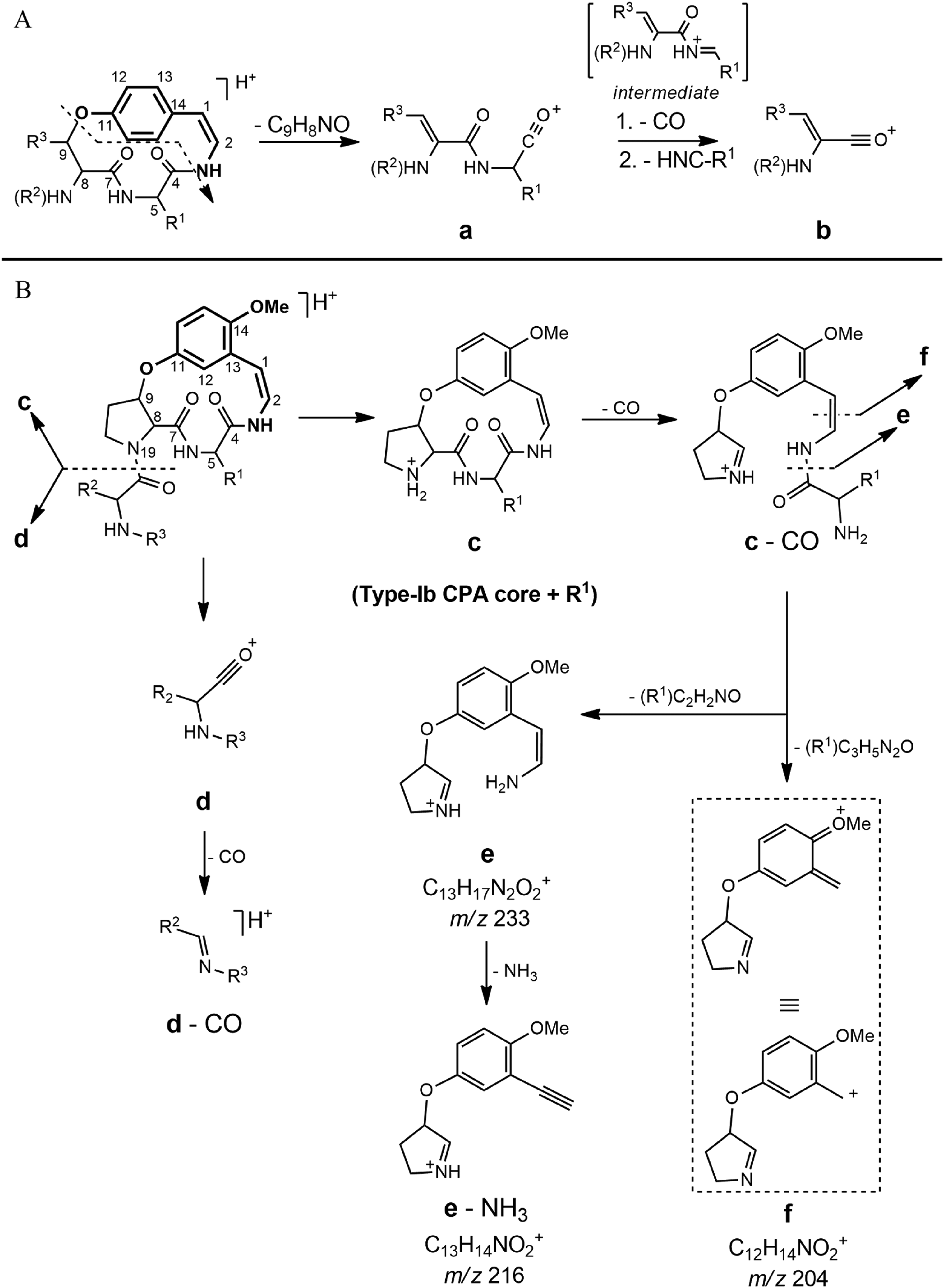
Proposed fragmentation mechanism of type-Ia (A) and type-Ib (B) cyclopeptide alkaloids. The characteristic styrylaminine moiety is highlighted in bold in the core structures.
Type-Ib CPA fragmentation (Figure 3B) is initiated at the side chain attached to the proline moiety by a [1,3] sigmatropic rearrangement of hydrogen to N-19, which generates the protonated ion species
Alkaloids
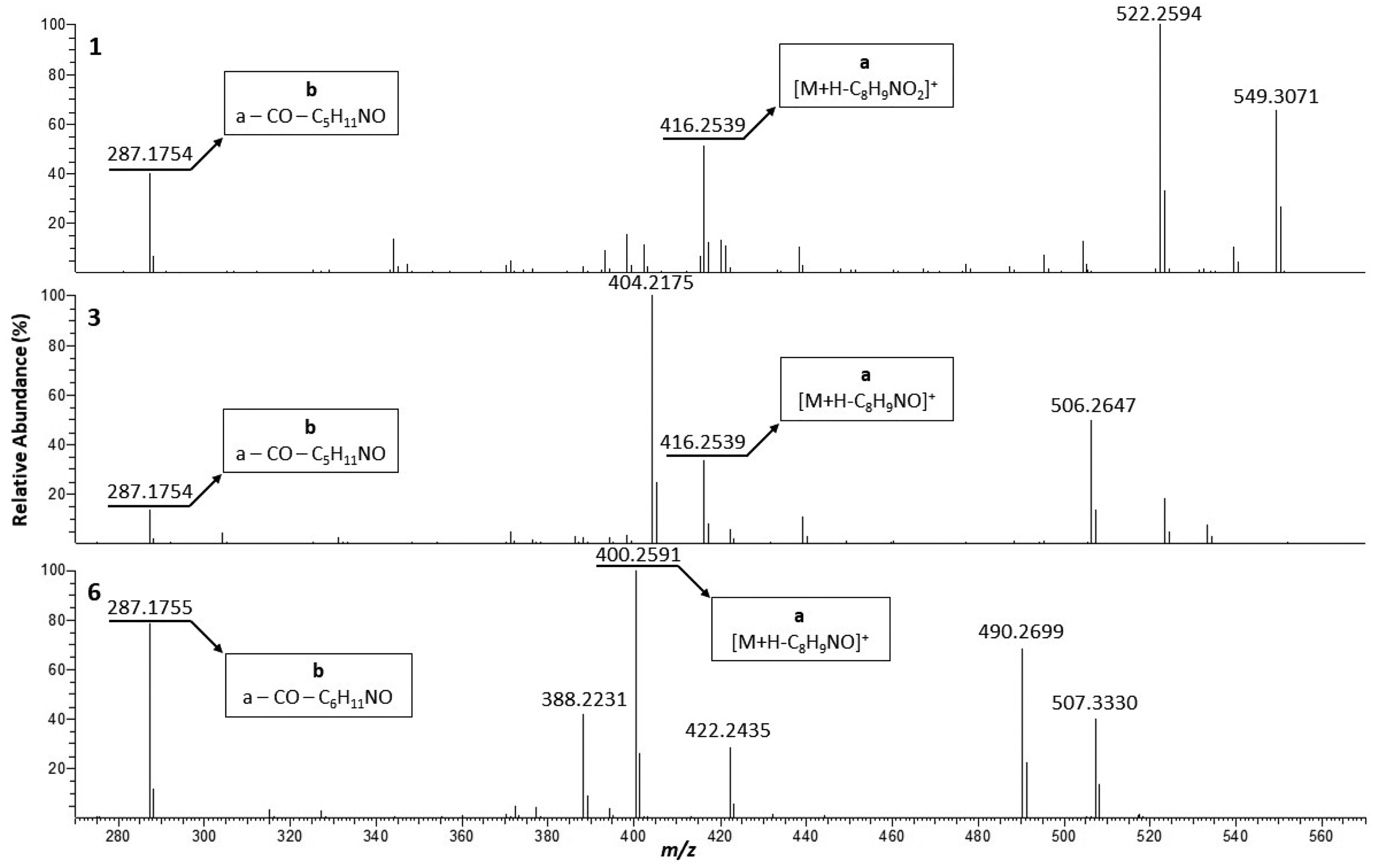
Comparison of the positive ion ESI-HR-MS2 spectra of type-Ia1 CPAs 1, 3 and 6 [m/z range 270 to 570].
For alkaloid
Alkaloid
The twelve remaining alkaloids possess a type-Ib CPA core, characterized by a meta-substituted aromatic ring (from the styrylaminine moiety) and with a proline residual chain attached (Figure 2). The characterization is based on the high number of fragment ions arising in the MS2 spectra, which are typical for this type of CPA (Figure 3B). In particular, the fragments
Alkaloid
Alkaloid
Alkaloid
Compound
Alkaloid
Alkaloid
For alkaloid
Alkaloids
Alkaloids
Alkaloids
Alkaloid
The extract of stems from Z. joazeiro exhibited significant activity (at 500 μg/mL) against the Gram-negative bacterium Allivibrio fischeri but influenced the growth of the Gram-positive test organism Bacillus subtilis only to a minor extent (Table 2). Furthermore, substantial cytotoxic activity (at 50 μg/mL) was detected when using the human cancer cell lines HT-29 and PC-3. Antibacterial activity is frequently reported for CPAs37-39 and is in accordance with the traditional application of this species. The observed anticancer effects might be attributed to the characteristic triterpenoids reported for Z. joazeiro.40,41
Antibacterial and Cytotoxic Activity of Crude Methanolic Extract From the Stem of Z. joazeiro.

100 μM chloramphenicol.
125 μg/mL digitonin.
Conclusions
Sixteen cyclopeptide alkaloids were detected and characterized in the crude alkaloid fraction from the stems of Ziziphus joazeiro by means of UHPLC coupled to high-resolution mass spectrometry. The putative identification of several cyclopeptide alkaloids was possible, while others escaped exact constitutional analysis due to the presence of residues of isomeric amino acid side chains (esp. Leu/Ile). The present method demonstrates an effective way to determine CPAs in alkaloidal extracts, and can be easily reproduced as required, e.g., for phytopharmaceutical quality control and adulteration analyses. Collision-induced fragmentation of type-Ia and -Ib CPA show many similarities in their fragment ions within the classes and some key fragments which can be used to identify related compounds in alkaloid rich extracts. This can guide the way to identify new cyclopeptides with yet to be determined configurations.
Experimental
Plant Material
Stems of Ziziphus joazeiro Mart. (Rhamnaceae) were collected on the campus of the Federal Rural University of Rio de Janeiro (Seropédica – RJ, Brazil, GPS coordinates: −22.762988 −43.695338). The species was identified by Prof. MSc. Marilena de Menezes Silva Conte from the Botany Department of the Federal Rural University of Rio de Janeiro and a voucher specimen was deposited in the Herbarium of this University under register number: RBR 39319.
Extraction Procedure and Sample Preparation
Stems of Z. joazeiro (1.72 kg) were dried, milled and macerated with methanol (3 × 2 L). After removal of the solvent under reduced pressure, 118 g of crude extract was obtained. The extract was suspended in H2O (1 L) and acidified with HCl (2 M) to pH 1 to 2. The solution was then extracted with EtOAc (3 × 300 mL). The acidified residue was basified with ammonia solution to pH 8 to 9 and the basified solution was partitioned with CHCl3 (3 × 300 mL). The organic layer was concentrated under reduced pressure affording 523 mg of the crude alkaloidal fraction. Ten mg of the crude alkaloidal fraction was solubilized in 1 mL of methanol (spectroscopic grade, ChromasolvTM, Honeywell Riedel-de Haën™, Germany) and filtered through a Chromabond C18 SPE cartridge (Macherey-Nagel, Germany), eluting with 3 × 1 mL of methanol.
UHPLC-ESI-Orbitrap-HR-MS Analysis
The positive ion high resolution ESI mass spectra were obtained from an Orbitrap Elite mass spectrometer (Thermofisher Scientific, Bremen, Germany) equipped with a HESI electrospray ion source (spray voltage 4.5 kV; source heater temperature: 300 °C; capillary temperature 300 °C; FTMS resolution 30.000). Nitrogen was used as the sheath and auxiliary gas. The MS system was coupled with an ultra-high-performance liquid chromatography (UHPLC) system (Dionex UltiMate 3000, Thermofisher Scientific), equipped with a RP-C18 column (1.9 μm; 50 × 2.1 mm; Hypersil Gold; Thermofisher Scientific; column temperature: 40 °C), and a photodiode array detector (PDA, Thermofisher Scientific). For UHPLC a gradient system was used, starting from H2O:CH3CN 95:5 (each of them containing 0.1% formic acid, isocratic for 2 min) raised to 5:95 within 15 min and then held on 5:98 for a further 3 min; flow rate 400 μL/min; injection volume 2 μL. The wavelength range of the PDA measurements was 190 to 600 nm. The CID mass spectra (buffer gas: helium, FTMS resolution 15.000) were recorded in data dependent acquisition mode using a normalized collision energy of 45%. The instrument was externally calibrated by the Pierce® LTQ Velos ESI positive ion calibration solution (product number 88323, Thermo Fisher Scientific, Rockford, IL, 61105 USA). The data were evaluated with Xcalibur software 2.2 SP1 (Thermo Fisher Scientific).
Isolation and HPLC Separation of Major CPAs
An aliquot of the crude alkaloidal fraction (474 mg) was fractionated by column chromatography on Sephadex LH20, eluting with MeOH/CHCl3 (9:1), followed by CC of 73 mg on silica gel eluting with CHCl3/MeOH (9:1). The separation was monitored by TLC (silica gel, CHCl3/MeOH [9:1]) and fractions containing Dragendorff positive spots (Rf 0.63) were combined. Final purification was performed on an Agilent 1260 HPLC system (quaternary pump, autosampler, column oven, DAD) equipped with a fully endcapped RP C18 column (5 μm, 150 × 4.6 mm YMS, ODS-A). Due to the low amount of compounds, the separation was performed on an analytical scale applying an isocratic solvent mixture of 20% acetonitrile/80% H2O containing 0.1% formic acid, flow rate 1 mL/min, detection at 280 nm) to give compounds
NMR Analysis
1D NMR spectra were obtained with an Agilent DD2 400 NMR spectrometer at 399.915 and 100.569 MHz for 1H and 13C, respectively. 2D NMR spectra were recorded on an Agilent 600 NMR spectrometer (599.831 MHz). TMS was used as an internal standard for 1H NMR analysis.
Biological Activities
The crude methanol extract of Z. joazeiro stems (50 and 500 μg/mL) was tested against Gram-negative (Aliivibrio fischeri) and Gram-positive (Bacillus subtilis) bacteria, as well as against the human tumor cell lines PC-3 (prostate cancer) and HT 29 (colon adenocarcinoma), as reported previously. 42
Supplemental Material
sj-docx-1-npx-10.1177_1934578X211054955 - Supplemental material for UHPLC-ESI-Orbitrap-HR-MS Analysis of Cyclopeptide Alkaloids From Ziziphus joazeiro
Supplemental material, sj-docx-1-npx-10.1177_1934578X211054955 for UHPLC-ESI-Orbitrap-HR-MS Analysis of Cyclopeptide Alkaloids From Ziziphus joazeiro by Carlos Henrique Corrêa dos Santos, Mário Geraldo de Carvalho, Annegret Laub, Katrin Franke and Ludger Wessjohann in Natural Product Communications
Footnotes
Acknowledgments
The authors are very grateful to the CAPES Foundation for a scholarship granted to Carlos Henrique Corrêa dos Santos (PDSE/88881.133796/2016-01), CNPq, FAPERJ and the Leibniz Association for financial support. We thank Andrea Porzel for NMR measurements, Anja Ehrlich for HPLC analysis, and Anke Dettmer and Martina Lerbs for antibacterial and cytotoxic assays, respectively.
Supplemental Material
Supplemental material for this article is available online, and all data (NMR and MS) will be made available upon request.
Author Contributions
CHCS conceived the study, performed experimental work, wrote the manuscript draft. AL performed MS measurements. KF contributed to data curation and manuscript writing. All authors contributed to evaluation of MS fragmentation patterns. MGC and LW were involved in supervision, guidance of the work and raising the funding. All authors took part in editing and correcting the first draft and read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (grant number PDSE/88881.133796/2016-01).
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
