Abstract
Phloridzin is a naturally occurring dihydrochalcone with various therapeutic properties. However, its low aqueous solubility and poor enzyme inhibitory capacity have limited its application in functional foods and medicines. Inspections of the properties of natural polymeric flavonoids suggest that these limitations could be mitigated by the polymerization of phloridzin, although to date, no relevant studies have been conducted. Here, oxidative polymerization was used to prepare polymeric phloridzin using laccase as the catalyst, and its structure, antioxidant capacity and α-glucosidase inhibitory activity were characterized. The results showed that laccase catalyzed polymerization via oxidative generation of phenolic radicals in the B ring of phloridzin to achieve the polymerization. The 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt (ABTS) radical scavenging capacity of the polymer (IC50, 0.48 mg/mL) was inferior to that of phloridzin (IC50, 0.20 mg/mL), but the α-glucosidase inhibitory activity of the polymer (IC50, 0.12 mg/mL) was significantly higher than that of phloridzin (IC50, 0.21 mg/mL). These effects could be attributed to the reduction of available phenolic groups and binding of the polymer to the enzyme, respectively.
Keywords
Introduction
Phloridzin is a natural dihydrochalcone with various pharmacological effects, such as antioxidant, anti-inflammatory, anti-viral, antihypertensive, antihyperglycemic and anti-tumor activities.1-3 The compound is found in many plants, particularly Malus species, being an active ingredient in herbal medicines.4,5 However, its low aqueous solubility and poor enzyme inhibitory capacity have limited its application in functional foods and medicines. Natural flavonoid polymers, such as theaflavins, proanthocyanidins and tannins, have higher aqueous solubility and biological activities than flavonoid monomers.6-8 This suggested that polymerization could improve the enzyme inhibitory capacity of phloridzin. Chemical and enzymatic methods can be used for the polymerization of flavonoids. Geng et al 9 and Kim et al 10 synthesized catechin-glyoxylic acid and catechol-aldehyde polycondensates, respectively, in aqueous ethanol. Compared with catechin, the polycondensates showed increased α-glucosidase and tyrosinase inhibitory activities. However, the chemical polymerization method has some limitations, such as poor regional selectivity, the formation of toxic residues, low product yields, and environmental pollution. Laccase is a multi-copper oxidase, which was first found in lacquer from the tree Toxicodendron vernicifluum. 11 It can catalyze the oxidative polymerization of phenols, polyamines and lignins.12,13 Because the main product of laccase-mediated reaction is water, it is a green synthesis enzyme and has attracted the attention of researchers. In this study, oxidative polymerization was used to prepare polymeric phloridzin using laccase as the catalyst. The structure of the polymer was confirmed by ultraviolet-visible (UV), Fourier transform infrared (FT-IR) and nuclear magnetic resonance (1H and 13C NMR) spectroscopy. Its antioxidant and α-glucosidase inhibitory activities were also determined.
Results and Discussion
In the presence of oxygen, laccase can catalyze the oxidative generation of free radicals in the phenolic moiety, thus initiating their polymerization. Here, phloridzin was polymerized by laccase and its properties were systematically characterized. Compared with phloridzin (2.84 mg/mL), the aqueous solubility of the polymer was much improved (18.52 mg/mL). The UV spectra of flavonoids typically exhibit absorption bands at 300 to 550 nm (band I) and 240 to 285 nm (band II); band I is mainly related to the cinnamyl system of the B ring, while band II is related to the benzoyl system of the A ring. Since phloridzin is a dihydrochalcone with a benzoyl system, its UV spectrum shows a band II absorption maximum at 285 nm. The maximum absorption peak of the polymer also occurred at 285 nm, indicating that the skeleton structure of the parent flavonoid was retained; the reduced intensity of the absorption peak could be attributed to polymerization.
The characteristic FT-IR spectral peaks of polyphenols (3384, 2921, 1627, 1455, 1206, 1172 and 1073 cm−1) could also be found in the FT-IR spectrum of the polymer although, compared with phloridzin, the peaks of the polymer were broader and smoother. Furthermore, the fingerprint region corresponding to the benzene ring (932 and 831 cm−1) of the polymer was significantly different from that of phloridzin (975 and 896 cm−1), suggesting that the substitution pattern of rings A and B of phloridzin had changed after polymerization.
The 1H and 13C NMR spectral data of phloridzin and its polymer are given in Table 1. After polymerization, the chemical shifts of the 1H NMR peaks corresponding to H-2, H-3, H-5, and H-6 on the B ring (Figure 1) shifted downfield, while those of the peaks for H-3′ and H-5′ on ring A remained unchanged. The intensities of the 13C NMR peaks corresponding to C-2, C-3, C-4, C-5, and C-6 on ring B also decreased sharply and shifted to the high-field region. However, the chemical shifts for the remaining carbon peaks did not shift significantly. Khlupova et al also found similar changes in the NMR spectra during a study of the laccase-mediated biotransformation of dihydroquercetin. 14 It could be concluded that laccase catalyzed the oxidation of the phenolic hydroxyl groups on ring B to initiate the polymerization via ether bond linkages.

Chemical structure of phloridzin.
1H- and 13C-NMR δ Values for of Phloridzin and its Polymer in DMSO-D6.

The ABTS radical scavenging assay is the widely used method for the antioxidant assessment of natural products. It is a spectrophotometric technique based on quenching of the stable colored radical of ABTS, and the characteristic absorbance peak at 734 nm decreases with the scavenging activity of antioxidants. Figure 2 shows that the ABTS radical scavenging activities increased with increasing concentrations of phloridzin and its polymer. The performance of phloridzin (IC50, 0.20 mg/mL) was superior to that of its polymer (IC50, 0.48 mg/mL). This might be due to the loss of phenolic hydroxyl groups on ring B during polymerization, resulting in a decrease in antioxidant activity.

ABTS radical scavenging activities of phloridzin and its polymer.
Diabetes mellitus is a metabolic disorder characterized by hyperglycemia and can cause many health complications. Estimates of the global cost of diabetes have shown that it may account for 2 to 3% of the total health care budget in all countries. Studies have demonstrated that the use of bioactive α-glucosidase inhibitors is an effective means of hyperglycemia treatment. However, the clinical α-glucosidase inhibitors currently in use (eg, acarbose, voglibose and miglitol) are associated with significant side effects that affect many patients. Consequently, the screening and development of alternative safe and effective α-glucosidase inhibitors is a priority.15,16 The α-glucosidase inhibitory activities of phloridzin and its oxidative polymer are shown in Figure 3. While both samples inhibited α-glucosidase in a concentration-dependent manner, the oxidative polymer possessed a higher inhibitory capacity (IC50, 0.12 mg/mL) than phloridzin (IC50, 0.21 mg/mL). The enhanced performance of the phloridzin polymer may be due to improved binding with the enzyme.
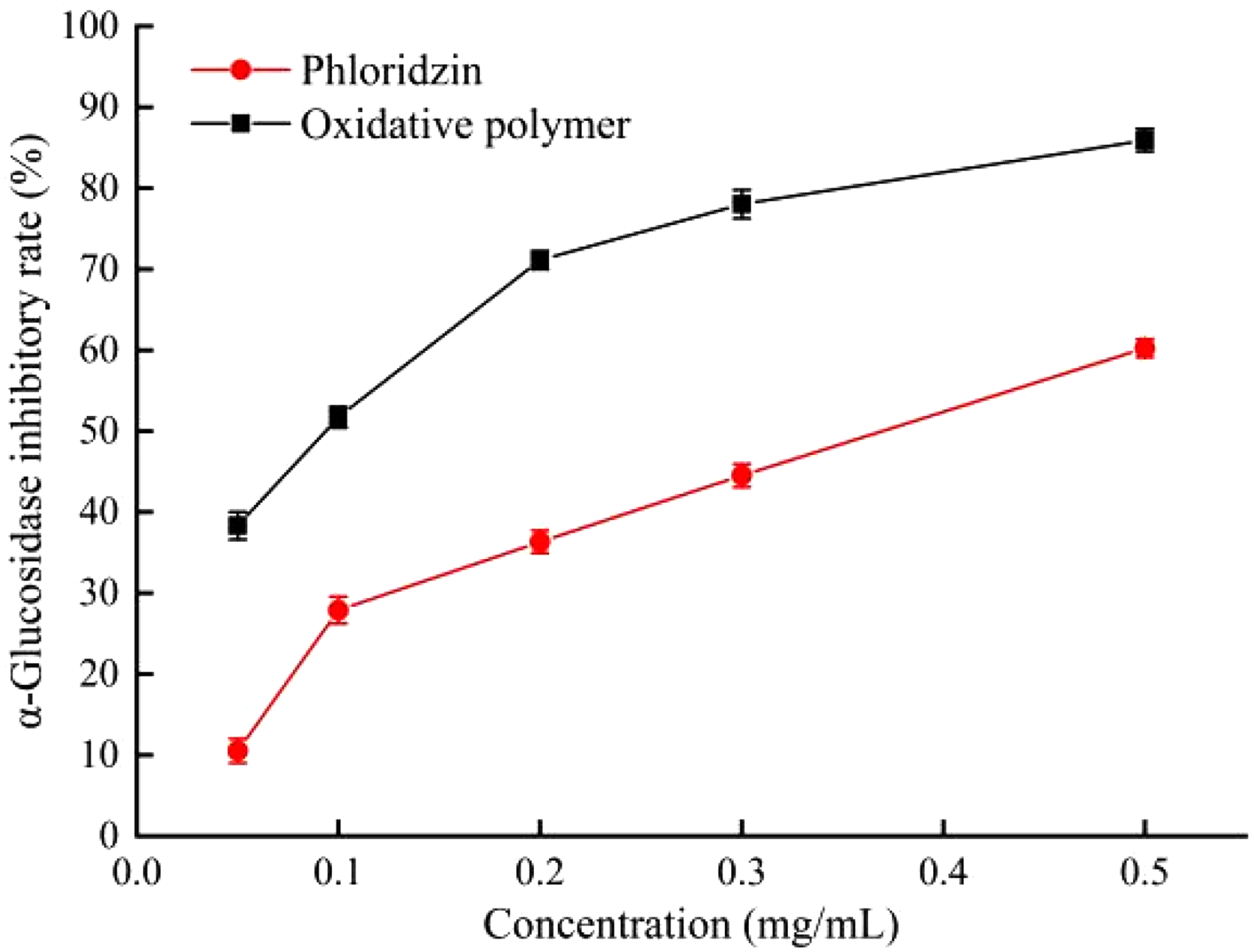
α-Glucosidase inhibitory activities of phloridzin and its polymer.
Conclusions
In this study, polymeric phloridzin was successfully prepared by oxidative polymerization using laccase as the catalyst. Characterization using FT-IR and NMR spectroscopy indicated that the laccase catalyzed polymerization via oxidation of phenolic hydroxyl groups in the B ring of phloridzin. Compared with phloridzin, the ABTS radical scavenging capacity of the polymer was inferior, but its α-glucosidase inhibitory activity was significantly higher.
Materials and Methods
Chemicals
Phloridzin was from Aladdin (Shanghai, China), and laccase, ABTS, α-glucosidase, and p-nitrophenyl-α-D-glucopyranoside (pNP-G) from Sigma-Aldrich (St. Louis, MO, USA).
Oxidative Polymerization of Phloridzin
Phloridzin and laccase were dissolved in phosphate buffer (0.1 mM, pH 5.8). Phloridzin solution (100 mL of 0.75 mg/mL) was mixed with laccase (15 mL of 10 U/mL). The obtained solution was kept at 25 °C for 48 h, dialyzed for 24 h, and freeze-dried to yield the oxidative polymer.
Structural Characterization
The UV spectra of phloridzin and its oxidative polymer were measured in methanol over 220 to 400 nm using a TU-1810PC UV spectrophotometer (Beijing Persee General Instrument Co., Ltd, Beijing, China). FT-IR spectra (400-4000 cm−1) were obtained from samples prepared by the KBr pellet method on a TENSOR 27 FT-IR spectrophotometer (Bruker, Germany). NMR spectra were obtained from samples in DMSO-D6 at 25 °C on an AVANCE 600 MHz NMR spectrometer (Bruker, Germany).
ABTS Radical Scavenging Assay
ABTS radical scavenging activity was measured according to the procedure of Takano, Nakamura and Tabata.
17
Briefly, sample solution (1 mL of varying concentrations) was mixed with ABTS test solution (2 mL) and maintained at 25 °C for 10 min. The absorbance of samples and the control (deionized water) were recorded at 734 nm and the ABTS scavenging rate was calculated from equation (1):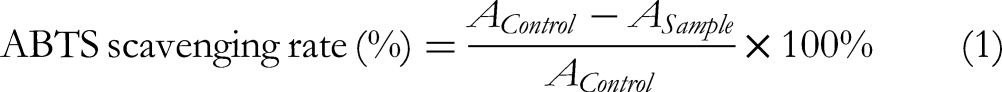
α-Glucosidase Inhibitory Assay
α-Glucosidase inhibitory activity was determined by the method of He et al
18
α-Glucosidase solution (1 mL of 0.2 U/mL) and sample solution (1 mL of varying concentrations) were mixed and maintained at 37 °C for 10 min. pNP-G solution (1 mL of 1 mM) was added and the mixture incubated for 20 min at 37 °C. Finally, the reaction was quenched by the addition of ethanol (1 mL). The absorbance of samples and the control (phosphate buffered saline) were measured at 410 nm and the α-glucosidase inhibitory rate was obtained from equation (2):
Statistical Analysis
All results of three replicates were expressed as mean ± SD from three replicates measurements. Statistical comparisons were analyzed based on the Tukey method with a confidence level of 95% using IBM SPSS Statistics for Windows, Version 22.0 (IBM Corp., Armonk, NY).
Footnotes
Acknowledgments
The authors are very thankful to all authors whose work has been cited in this paper.
Author Contributions
Haoyu Zhou: Conceptualization, Methodology, Validation, Data curation, Writing-original draft preparation, Writing-review and editing, Visualization, Funding acquisition; Changzhong Liu: Software, Formal analysis, Resources, Writing-review and editing, Funding acquisition; Sheng Geng: Conceptualization, Methodology, Investigation, Writing-original draft preparation, Supervision, Project administration.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Key Scientific and Technological Project in Henan Province of China (No. 202102110135) and Zhongyuan high level talents special support plan (No. 204200510010). Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
