Abstract
A new flavone glycoside, lumnitzerone (
α-Glucosidase is an intestinal enzyme that breaks down α-1,4 linked polysaccharides to α-glucose, which leads to high blood sugar levels. The development of an α-glucosidase inhibitor derived from a natural product could be an important contribution to diabetes prevention.
Lumnitzera littorea (family Combretaceae) grows in Can Gio mangrove forest in Vietnam. There have been 2 reports about the phytochemistry of this species. The antimicrobial activities of n-hexane, ethyl acetate, and methanol extracts of the leaves were evaluated against 6 human pathogenic microbes and the first extract was the most active. 1 Recently, we reported the isolation of gallic acid and naringenin. 2 Here, we present the chemical constituents as well as the α-glucosidase inhibitory activity of extracts and compounds isolated from L. littorea leaves.
The ethyl acetate extract of L. littorea leaves yielded one new and 9 known flavonoids. The new flavonoid glycoside (
The known compounds were identified from spectroscopic analysis and comparison with literature data as quercetin (
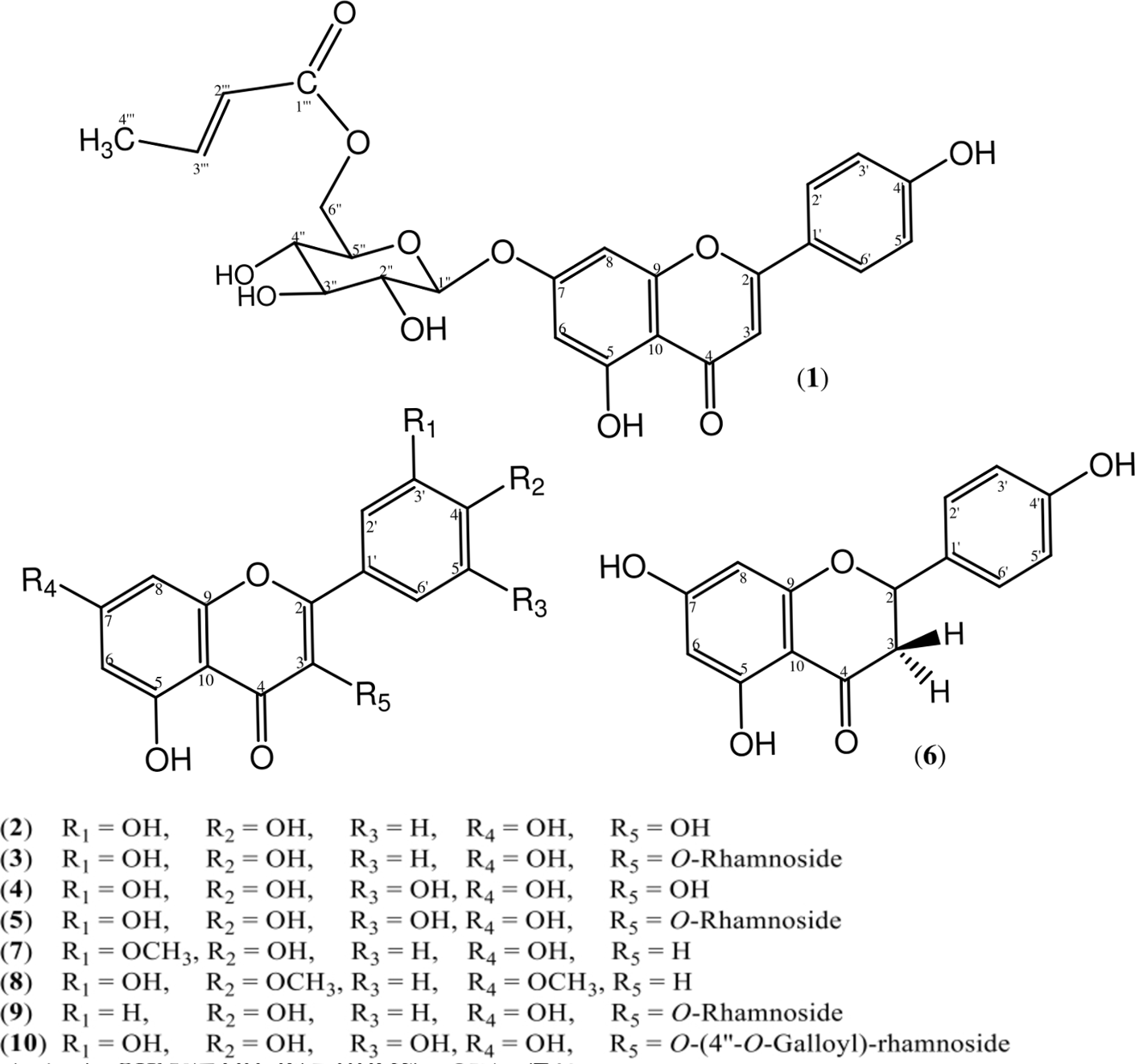
Structure of compounds isolated from Lumnitzera littorea.
The in vitro α-glucosidase inhibitory activity was evaluated of the extracts and purified compounds (Tables 1 and 2). All the extracts and most of the isolated compounds exhibited better activities than the positive control acarbose. The flavonoid-type structure, the position, and the number of hydroxy groups are determining factors for α-glucosidase inhibition. The A-ring 7-OH and the B-ring 4′-OH groups play an important role in the inhibitory effect. This observation was proved by the most potent inhibitors such as naringenin (
In Vitro α-Glucosidase Inhibitory Activity of Isolated Compounds From Lumnitzera littorea.
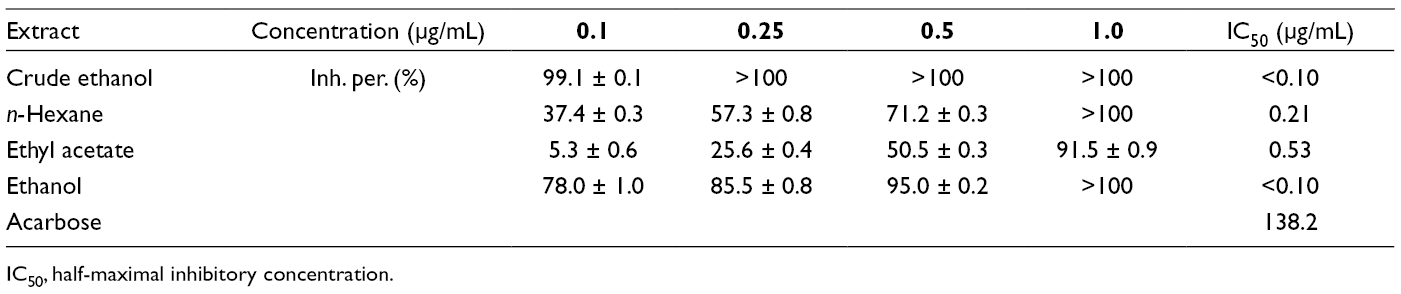
IC50, half-maximal inhibitory concentration.
In Vitro α-Glucosidase Inhibitory Activity of Isolated Compounds From Lumnitzera littorea.
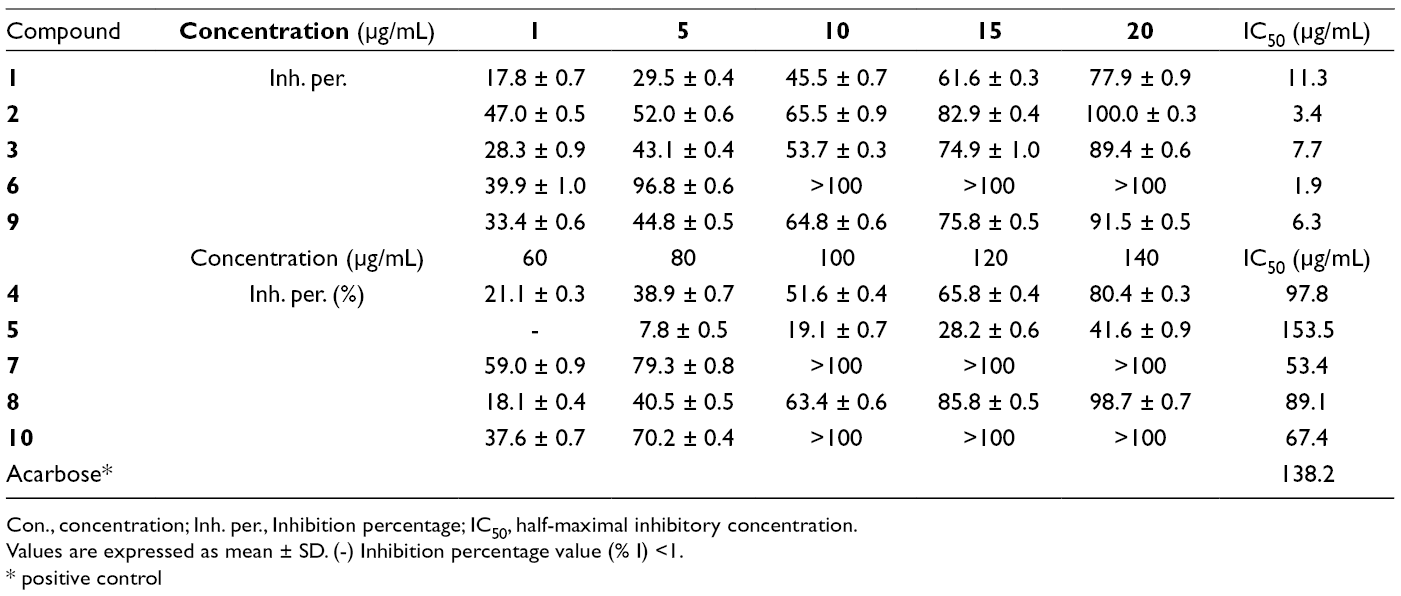
Con., concentration; Inh. per., Inhibition percentage; IC50, half-maximal inhibitory concentration.
Values are expressed as mean ± SD. (-) Inhibition percentage value (% I) <1.
* positive control
Careful analysis of the chemical shifts, multiplicities, and coupling constant magnitudes in the 1H NMR spectrum, along with the 1H-1H COSY and 13C NMR spectra of
Nuclear Magnetic Resonance Data of Compound 1.

HMBC, heteronuclear multiple bond correlation; DMSO, dimethyl sulfoxide.
(
The multiplicity of the protons H3-4′′′, H-3′′′, and H-2′′′ and the 1H-1H COSY spectrum confirmed their contiguous arrangement. The heteronuclear multiple bond correlation (HMBC) experiments showed cross-peaks (Figure 2) of the propenyl protons at δ 5.83 (H-2′′′) and 6.85 (H-3′′′), as well as the methylene protons at δ 4.41 (H-6′′a) and 4.08 (H-6′′b) to the carbonyl carbon C-1′′′ (δ 165.3) suggesting that the butenoyloxy group was attached at C-6″ of the D-glucose. The attachment of this sugar to C-7 in the apigenin nucleus was confirmed by the HMBC cross-peak of the anomeric proton at δ 5.13 (1H, d, 7.5 Hz) to carbon C-7 (δ 162.6). Accordingly,

Key COSY and HMBC correlations of compound 1.
Experimental
General Experimental Procedure
The NMR spectra were recorded on a Bruker Avance III spectrometer at 500 MHz for 1H NMR and 125 MHz for 13C NMR spectra. HR-ESI-MS were obtained on a Shimadzu +IDA time-of-flight MS. TLC was performed on silica gel 60 F254 (Merck, Darmstadt, Germany). Gravity column chromatography was performed on silica gel 60 (0.040-0.063 mm, Merck) and Sephadex LH-20 (GE Healthcare Bio-Science AB, Uppsala, Sweden). α-Glucosidase (EC 3.2.1.20) from Saccharomyces cerevisiae (750 UN) and p-nitrophenyl-α-D-glucopyranoside were purchased from Sigma Chemical Co. (St Louis, MO, USA). Acarbose and dimethyl sulfoxide were obtained from Merck. Other chemicals were of the highest grade available.
Plant Material
Leaves of Lumnitzera littorea (Jack) Voigt (Combretaceae) were collected at Can Gio mangrove forest, Ho Chi Minh City, Viet Nam in August of 2014. The scientific name of the plant was authenticated by Dr Pham Van Ngot, Faculty of Biology, Ho Chi Minh City University of Pedagogy. A voucher specimen (No US-B012) was deposited in the herbarium of the Department of Organic Chemistry, University of Science.
Extraction and Isolation
The fresh leaves were washed under running tap water to remove all sand partitcles and epiphytes and then were dried and ground into fine powder. The powder (15 000 g) was exhaustively extracted with ethanol at room temperature by maceration. After filtration the ethanol solution was evaporated to dryness under reduced pressure to yield a crude ethanol residue (1000 g). This was applied to a silica gel solid phase column and eluted consecutively with n-hexane, ethyl acetate, and finally with ethanol. After evaporation under reduced pressure, 3 extracts were obtained, n-hexane (100 g), ethyl acetate (250 g), and ethanol (550 g). The ethyl acetate extract (250 g) was fractionated by silica gel column chromatography using a mixture of EtOAc−MeOH (99:1 to 0:100) to yield 8 fractions (EA1-EA8). These were then continuously separated using silica gel and Sephadex LH-20 and eluted with appropriate solvent systems of EtOAc−MeOH to give 10 compounds. As a result, fraction EA2 afforded
Bioactivity Assay
The α-glucosidase inhibitory activity was determined according to the method of Apostolidis et al 12 The inhibitory activity was calculated using the following equation:
% Inhibition= (A control− A sample) / A control
The IC50 values were determined from plots of percent inhibition vs log inhibitor concentration and calculated by non-linear regression analysis from the mean inhibitory values.
Lumnitzerone (1)
Light yellow solid.
1H, 13C NMR and HMBC (DMSO-d 6): Table 3.
HR-ESI-MS: m/z [M+H]+ calculated for C25H24O11+H, 501.1397; found: 501.1398.
Acid Hydrolysis of 1
Compound
Supplemental Material
Supporting information - Supplemental material for A New Flavone Glycoside From Lumnitzera littorea with In Vitro α-Glucosidase Inhibitory Activity
Supplemental material, Supporting information, for A New Flavone Glycoside From Lumnitzera littorea with In Vitro α-Glucosidase Inhibitory Activity by Nguyen T. L. Thuy, Pham T. Thuy, Bui T. Tung, Huynh T. Loc, Truong T. T. Dang, Le L. Ngoc, Nguyen X. Duc, Le T. Dung, Poul E. Hansen, and Nguyen K. P. Phung in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is funded by HoChiMinh City Open University under the grant number E2017.2.11.1.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
