Abstract
Propolis is an aggregate of functional components found in plant resins and has been reported to exhibit a variety of valuable biological activities. This study investigated the inhibitory properties of propolis from Thailand toward human catechol-O-methyltransferase (COMT), a key neurotransmitter involved in Parkinson's disease and depression. Samples collected from Chanthaburi and Chiang Mai exhibited relatively high inhibitory activity against COMT. γ-Mangostin (
Introduction
Catechol-O-methyltransferase (COMT, EC 2.1.1.6) catalyzes the transfer of the methyl group from S-adenosyl-L-methionine (SAM) to one of the hydroxy groups of a catechol-bearing substrate in the presence of magnesium ions (Mg2+). 1 COMT plays a dominant role in the inactivation of endogenous catecholamine neurotransmitters, including dopamine, noradrenaline, and adrenaline 2 ; therefore, it is seen as a target for the treatment of Parkinson's disease, schizophrenia, and depression. 3 Tolcapone, entacapone, and opicapone are COMT inhibitors used to treat Parkinson's disease. 4 However, tolcapone exhibits severe hepatotoxicity, 5 and the toxicity of opicapone has not yet been evaluated in detail. 6 Therefore, COMT inhibitors with low toxicity and a good safety profile are required.
Natural sources of pharmaceuticals may offer many potentially safe COMT inhibitors because of their low toxicity. To date, various compounds that can act as COMT inhibitors, such as caffeic acid 7 and rutin, 8 have been isolated from medicinal plants. In this context, we have focused on propolis in searching for new COMT inhibitor candidates. Propolis is a natural resinous substance collected from the buds and exudates of certain trees and plants by honeybees. 9 Notably, it has been reported to display a variety of valuable biological activities and has been used as a folk medicine in many regions of the world. Therefore, propolis is a candidate in the search for new medicinal agents. We recently reported on the components of Thai propolis. For example, xanthone derivatives, including α- and γ-mangostin, are the major components found in propolis from the Chanthaburi region. This kind of propolis originates from resin on the surface of mangosteen fruits. 10 On the other hand, propolis from Chiang Mai contains prenylflavonoid derivatives. 11
Although the components of Thai propolis are potentially valuable pharmaceutical resources, they are not used effectively and are almost entirely wasted. This study investigated the COMT inhibitory activity of Thai propolis and its major compounds to further assess the pharmaceutical value and efficacy of these resources.
Results and Discussion
To identify any effective COMT inhibitors in propolis, three propolis samples were collected from different apiaries (Chanthaburi, Chiang Mai, and Phatthalung) in Thailand (Figure 1 and Table 1), and the COMT inhibitory activity of ethanol extracts from the samples were evaluated by using HPLC.
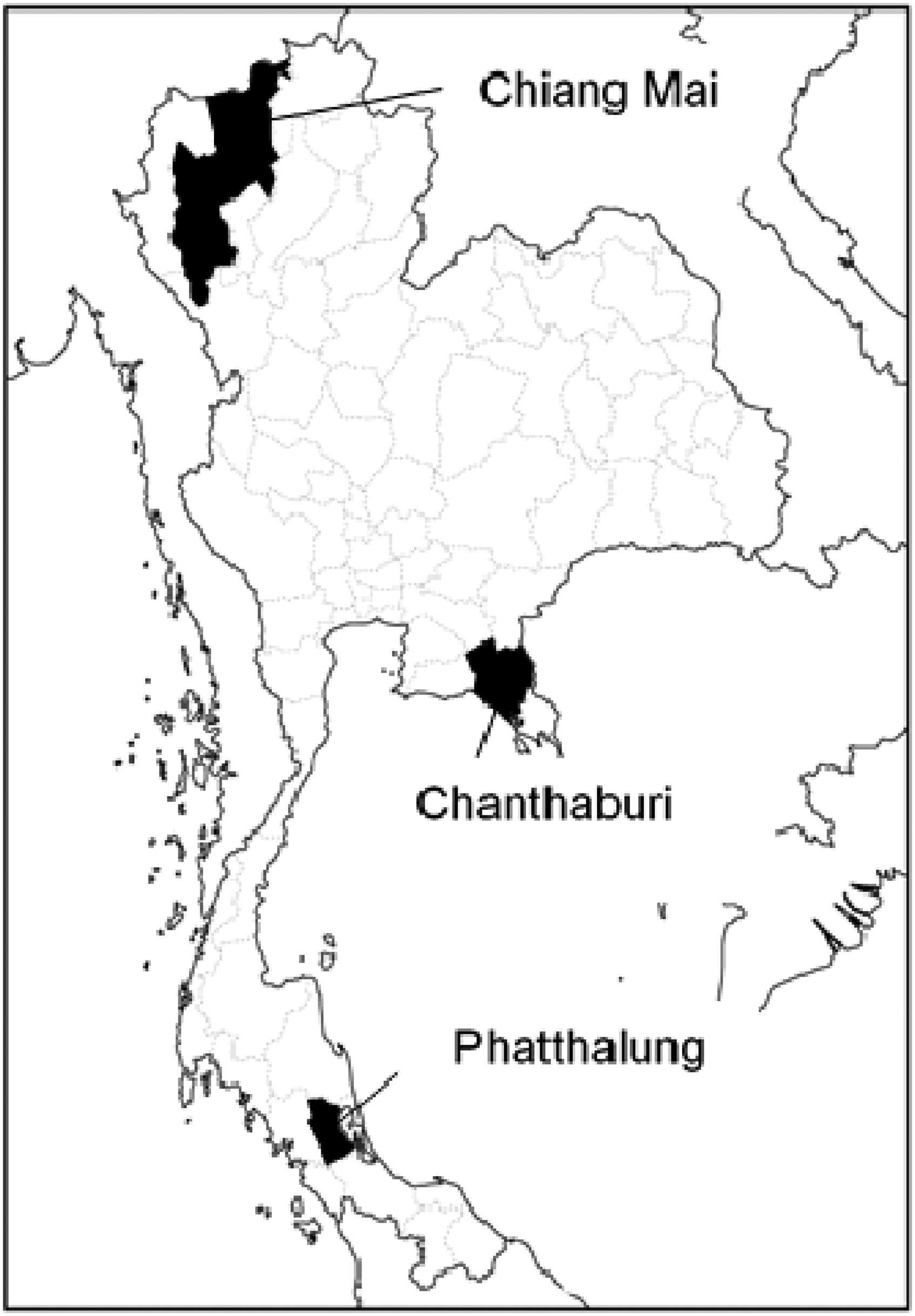
Propolis collection sites in Thailand.
Sample Information for Each Type of Propolis and the COMT Inhibitory Activity of its Ethanol Extract.

Sample concentration: 0.5 mg/mL.
The extracts obtained from Chanthaburi and Chiang Mai propolis exhibited strong COMT inhibitory activity at a concentration of 0.5 mg/mL (Table 1). By contrast, the extracts from Phatthalung propolis did not significantly inhibit COMT. To identify the COMT inhibitors in propolis, the major compounds found in the samples (Figure 2) were tested individually by using the same COMT inhibitory assay.

Major compounds in Chanthaburi (
The major compounds in Chanthaburi propolis are γ- and α-mangostins (
COMT Inhibitory Activity of Test Compounds (

Tested compounds other than
We subsequently investigated the type of inhibition exhibited by
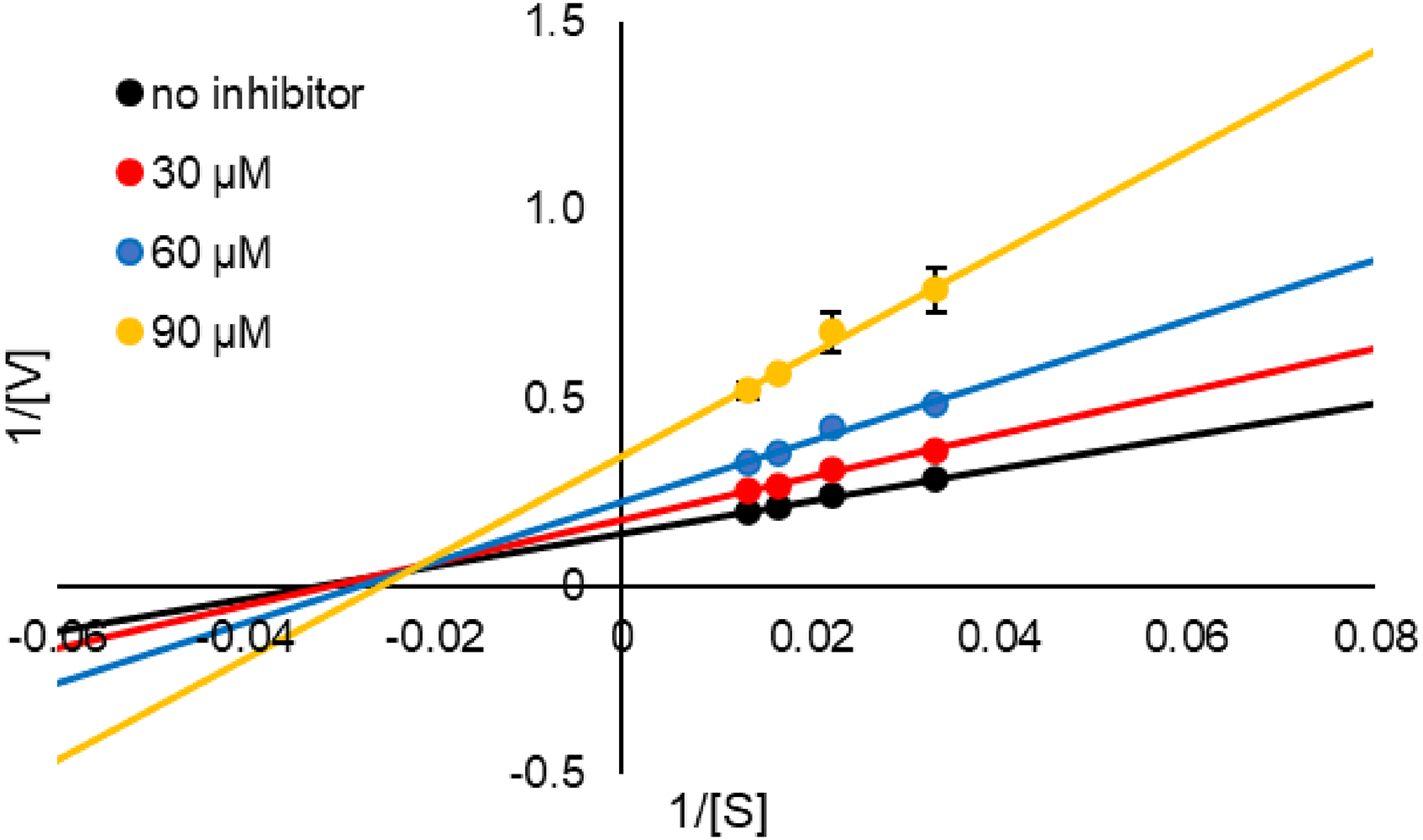
Lineweaver–Burk plot of γ-mangostin (
COMT has two substrate-binding sites: one for the methyl donor in the SAM substrate and the other for various methyl-accepting catechol substrates, such as 2-hydroxyestradiol, catechins, and dihydroxybenzoic acid. Many small-molecule inhibitors are known to bind to the methyl-acceptor pocket of COMT.
12
Compound
The amount of
Conclusions
In this study, we investigated the COMT inhibitory activity of Thai propolis and found that
Materials and Methods
Biological Materials
The Chanthaburi and Chiang Mai propolis were the same as those used in previous studies.10,11 The Phatthalung sample was collected from Ban Na Pa Kho Sub-district, Bang Kaeo District, Phatthalung Province, Thailand in May 2017. A voucher sample (no. BV20170501) of the Phatthalung propolis evaluated in this study has been deposited at the Faculty of Pharmaceutical Sciences, Burapha University, Thailand.
Tested Compounds
γ- And α-mangostins (
Expression and Purification of Human Recombinant COMT
A plasmid sample of human COMT (PMID: NP_009294.1) with C33S, C95S, and C188S mutations in the Novagen pET22b(+) vector via NdeI and HindIII, which contained a C-terminal histidine tag, was purchased from GenScript (Piscataway, NJ, USA). The plasmid was transformed into SHuffle T7. The strain was cultivated in lysogeny broth at 30 °C. When the OD600 value reached 0.6 to 0.8, 0.5 mM isopropyl-β-D-thiogalactopyranoside was added, and the cells were grown for ∼15 h at 16 °C. The cells were collected by centrifugation at 5000g for 10 min and then suspended in buffer A (100 mM Tris-HCl [pH 8.0], 300 mM NaCl, 5 mM MgCl2, 1 mM EDTA, 10% glycerol, 5 mM β-mercaptoethanol, and 10 μM phenylmethylsulfonyl fluoride). After sonication of the cells, the supernatant was collected by centrifugation at 11 000g for 30 min. The supernatant was loaded onto a 5 mL HisTrap HP column (GE Healthcare, Uppsala, Sweden) equilibrated with buffer B (100 mM Tris-HCl [pH 7.5], 100 mM NaCl, 5 mM MgCl2, and 5 mM β-mercaptoethanol) and then washed with 50 mL of buffer B. The column was washed with buffer B containing 100 mM imidazole, and the samples were then eluted with 30 mL of buffer B containing 300 mM imidazole. After concentrating the eluted samples, the samples were loaded onto a 5 mL HiTrap desalting column (GE Healthcare) to replace buffer C (50 mM Tris-HCl [pH 7.5] and 1.5 mM MgCl2). The purity of the COMT was confirmed by SDS-PAGE (Figure S1). The concentrations of the COMT samples were estimated by measuring the absorption at 280 nm with a NanoDrop spectrophotometer (Thermofisher Scientific, Waltham, MA, USA). Protein samples were stored at −30 °C.
COMT Inhibitory Assays
3′,4′-Dihydroxyacetophenone, 4′-hydroxy-3′-methoxyacetophenone, and tolcapone were purchased from Tokyo Chemical Industry Co. (Tokyo, Japan). 3′-Hydroxy-4′-methoxyacetophenone was purchased from Sigma–Aldrich. COMT inhibitory assays were performed by following a previously reported method with a slight modification. 18 The assay buffer (50 mM Tris-HCl [pH 7.5], 1.5 mM MgCl2, 20 μM 3,4-dihydroxyacetophenone, 200 μM SAM, and 0-200 μM inhibitor) was preincubated at 37 °C for 5 min. COMT samples were added to the buffer solution with a final concentration of 1 μM (total volume: 200 μL), and the reaction was started. After incubation of the mixture at 37 °C for 10 min, 3% aqueous HClO4 (50 μL) was added to terminate the reaction. In the reaction, 4′-hydroxy-3′-methoxyacetophenone and 3′-hydroxy-4′-methoxyacetophenone were produced from 3,4-dihydroxyacetophenone by COMT. To quantify the amount of both acetophenone products produced, an aliquot (20 μL) of the solution was injected onto an HPLC column under the following conditions: column, 5 μm, 2.1 × 150 mm, DAICEL CHIRALPAK® IA; flow rate, 0.4 mL/min; eluent, 0.1% trifluoroacetic acid in H2O−acetonitrile (85:15, v/v); detection, 280 nm. Standard curves for each enzyme product were plotted from the concentrations related to the integrated areas of the HPLC chromatograms. The percentage inhibition was calculated according to the following equation: Inhibition (%) = [(concentration of enzyme products in the control experiment) − (concentration of enzyme products in the sample experiment)] × 100/(concentration of enzyme products in the control experiment). For analytical HPLC, a PU-4180 RHPLC pump (Jasco, Tokyo, Japan), MD-4017 photodiode array detector (Jasco), and AS-4050 HPLC autosampler (Jasco) were used. Data were analyzed with ChromNAV software (v.2, Jasco).
Kinetic Study of γ-Mangostin (1) for COMT Inhibition
A kinetic study of
Footnotes
Acknowledgments
The authors thank Pharmaceutical Innovations of Natural Products Unit (PhInNat) for coordinating with beekeepers and Philip Hawke of the University of Shizuoka Scientific English program for his comments on the English in the manuscript.
Author Contributions
Ryo Miyata contributed toward the expression and assay of COMT, and the preparation of the manuscript. Tomoharu Motoyama contributed toward the expression of COMT. Shogo Nakano and Sohei Ito supervised the experiments and checked the descriptions in the manuscript. Kazuma Mukaide isolated the compounds from Chiang Mai propolis. Boonyadist Vongsak collected propolis samples. Shigenori Kumazawa supervised all the processes in the experiments and the manuscript preparation. All authors have read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Japan Society for the Promotion of Science (grant number KAKENHI JP18KK0165, KAKENHI JP20J23632).
Ethical Approval
This study was approved by the Committee of Genetic Recombination Experiment, the University of Shizuoka, Japan. This article does not contain any studies with animal or human subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
