Abstract
Coix (Coix lacryma-jobi Linné. var. mayuen Stapf) is, unlike many herbs used in Chinese traditional medicines, often used as a single agent. Coix seed has its specific constituents coixenolide and coixol. This review summarizes the effects of coix seed on human pathology and physiology, and the roles of coixenolide and coixol on the effects. The comprehensive literature search was conducted using PubMed, Citation Information by National Institute of Informatics of Japan (CiNii), University Hospital Medical Information Network Clinical Trials Registry (UMIN-CTR), and ClinicalTrials.gov databases. Some studies suggest that coix seed promotes the spontaneous regression of viral infections of the skin. Meanwhile, kanglite, a purified oil agent used for cancer therapy, has been implied to increase the ratio of CD4 + T cells in the peripheral blood of cancer patients undergoing treatment. These studies seem to indicate that coix seed may affect cellular immune function. However, there are limitations to the individual studies on which the above consideration is based, and thereby, it is not conclusive what specific immune or clinical indices coix seed affects. In addition, the effects of coixol on human immune function have not been clarified. Therefore, coix seed seems promising for health promotion and presents a challengeable and attractive theme for natural product chemists, pharmacologists, immunologists, and clinicians.
Introduction
Coix (Coix lacryma-jobi Linné. var. mayuen Stapf), also called adlay or pearl barley, is a grain-bearing perennial plant belonging to the grass family Poaceae. The grain is used as a source of food, drugs, and ornamentation, and the seed is used as a traditional Chinese medicine. Most traditional Chinese medical regimens are combinations of various ingredients, including from plant and animal sources. In contrast, coix seed is often used as a single source drug. It has been reported that coix seed contains coixenolide, and coixol, and it has traditionally been used to treat disorders such as cancer, as well as warts and skin pigmentation. 1
In Japan, coix seed and its water extract have been approved as ethical drugs for the treatment of verruca vulgaris and flat warts. However, coix seed is not used in countries other than Japan. This may be due in part to the fact that much of the literature on coix seed has been published in Japan and not shared internationally. In addition, although various effects have been advocated for coix seed, sound evidence on each effect is limited. This makes it impossible to set a clear clinical question to conduct a systematic review, as is the case with ginger 2 and lemon balm. 3 Therefore, we aimed to review comprehensively human trials with control groups on coix seed including literature written in Japanese for a broader audience. The findings on coixenolide and coixol were also summarized.
Methods
Review of the Effect of Coix Seed
Study Selection
A comprehensive literature search was carried out using the PubMed, Citation Information by National Institute of Informatics of Japan (CiNii), University Hospital Medical Information Network Clinical Trials Registry (UMIN-CTR), and ClinicalTrials.gov databases.
The inclusion criteria were as follows: (1) studies investigating the effect of coix seed or its extract; (2) human trials with a control group; and (3) studies written in English or Japanese. All articles were screened by two independent reviewers (Y. Suzuki and Y. Konaya) based on the title, abstract, and full text. The selected studies were confirmed as eligible for this review by the reviewers and disagreements were resolved by consensus.
Database Search
PubMed was queried on December 17, 2019. A total of 364 studies were identified using the search terms “coix lacryma-jobi,” “adlay,” and “pearl barley.” The filter “human” was applied, resulting in 108 remaining studies. Among these, six were manually selected after assessing the title, abstract, and text (Figure 1).

Flow diagram of the literature selection.
CiNii was searched on June 7, 2020. A total of 856 articles were identified using the keywords “coix,” “adlay,” “pearl barley,” and other Japanese words corresponding to coix, including “hatomugi” and “yokuinin.” Among these, six studies were manually selected after assessing the title, abstract, and text. All selected studies were written in Japanese and none were duplicates of the articles selected from PubMed. Among the studies selected from CiNii, a report by Fushiki et al 4 was a reproduction of another report by the same authors; 5 therefore, the reproduced report were excluded. The remaining five studies were selected for inclusion (Figure 1).
ClinicalTrials.gov was queried on July 19, 2020. Seven clinical trials were identified using the search words “coix,” “adlay,” and “pearl barley.” Among these, three trials were completed; however, the results were not available.
UMIN-CTR was searched on July 19, 2020. A total of eight clinical trials were identified with the search terms “coix,” “adlay,” “pearl barley,” and other Japanese words corresponding to coix, as described above. Among them, six trials were completed, but the results were also not available. However, one report was obtained by contacting the principal investigators of the study. 6
PubMed was searched again on July 19, 2020 with the search term “kanglaite.” A total of 47 studies were identified. Among them, 13 were meta-analyses.
Risk of Bias in Individual Trials
Excluding articles on kanglaite, the risk bias of the selected studies was evaluated by a reviewer (Y. Konaya) using the Cochrane risk of bias tool. 7 The other reviewer (Y. Suzuki) confirmed the results. The seven risk of bias domains assessed were: (1) random sequence generation, (2) allocation concealment, (3) blinding of participants and personnel, (4) blinding of data analysts, (5) incomplete outcome data, (6) selective reporting, and (7) other bias. The quality of the evidence was classified into three levels, low risk of bias, high risk of bias, and unclear risk of bias.
Specific Substances in Coix Seed
PubMed and CiNii were queried on August 18, 2020 using the search terms “coixenolide” or “coixol.” “Coixenbolide” had 5 entries in PubMed and 3 entries in CiNii. “Coixol” had 87 entries in PubMed and 14 entries in CiNii. Studies describing the physiological effect on mammals were selected by title, abstract, and text, and were reviewed. Substances other than coixenolide and coixol found in the selected literature were also summarized.
Results
Selected Publications
A total of 12 studies were included in this review: 6 from PubMed, 5 from CiNii, and 1 from UMIN-CTR. Due to the inconsistency of usage and subjects described, the selected literature was classified into five categories: (1) dealing with kanglaite,8,9 (2) coix seed effects on the systemic condition of diseased subjects,10,11 (3) the coix seed extract Kotaro,5,12–14 (4) original coix seed extract effects on the condition of the skin,6,15 and (5) the glycemic index of coix seed.16,17
Excluding studies on kanglaite,8,9 the quality assessment of the risk bias of the selected articles is shown in Figure 2, and the articles on comparative trials and glycemic index are summarized in Tables 1 and 2, respectively.

Risk of bias summary. Each color represents a different level of bias—red indicates high risk, green indicates low risk, and yellow indicates an unclear risk of bias.
Articles on Comparative Trials of Coix Seed.
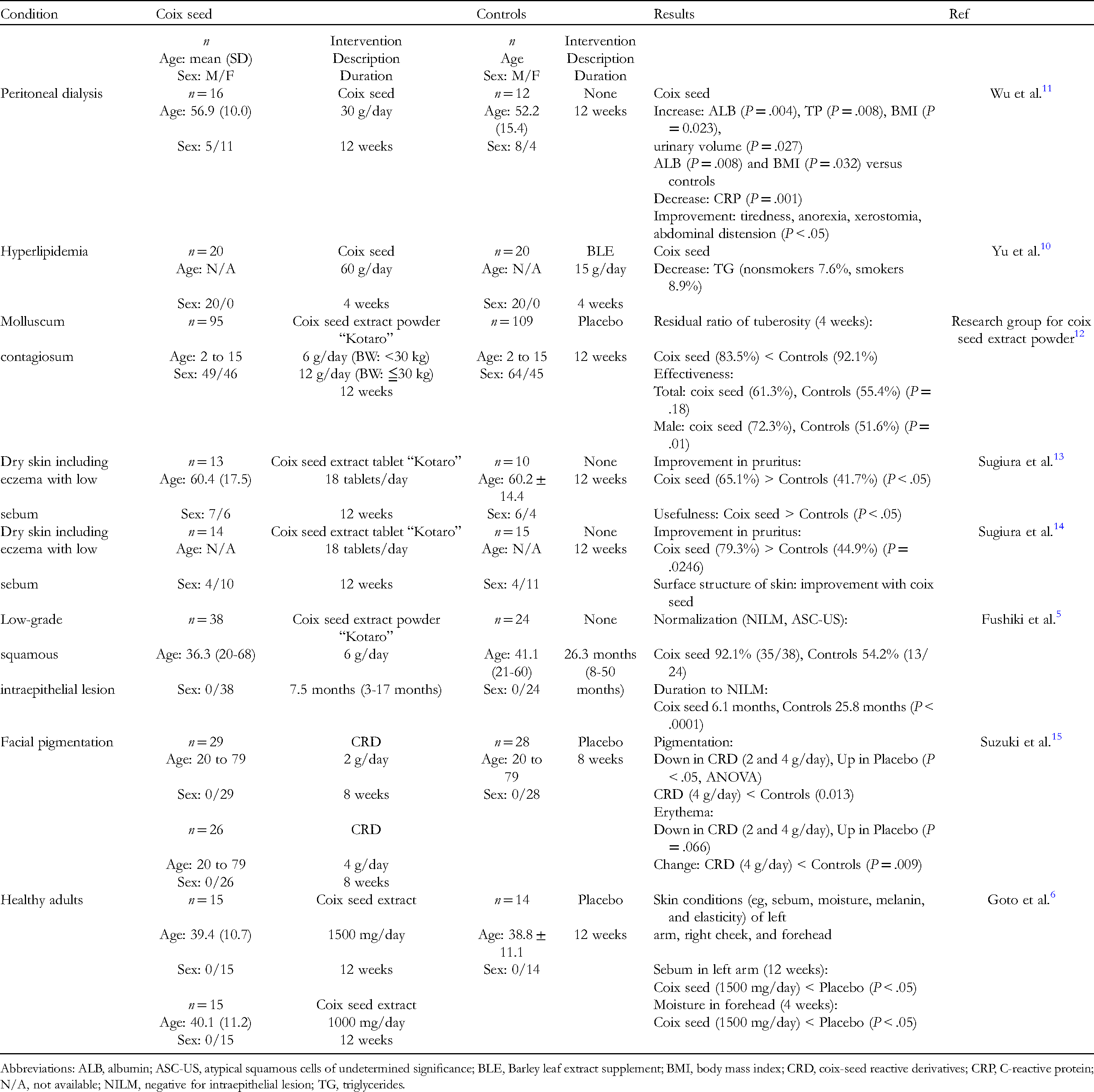
Abbreviations: ALB, albumin; ASC-US, atypical squamous cells of undetermined significance; BLE, Barley leaf extract supplement; BMI, body mass index; CRD, coix-seed reactive derivatives; CRP, C-reactive protein; N/A, not available; NILM, negative for intraepithelial lesion; TG, triglycerides.
Randomly Assigned Crossover Trials Assessing Glycemic index with a Single Administration of a Meal Containing 50 g of Carbohydrates.

Digitized from the graph with WebPlotDigitizer (https://automeris.io/WebPlotDigitizer/).
Abbreviations: BMI, body mass index; GI, glycemic index; SE, standard error; SD, standard deviation.
Kanglaite
Two reports selected from PubMed described the effect of kanglaite on cancer therapy.8,9 Kanglaite is an injectable microemulsion of purified oil extracted from coix seeds. Kanglaite has been approved as an ethical drug for the treatment of various cancers in China since 1995. However, search words related to coix failed to comprehensively identify literature concerning kanglaite. A PubMed search with the term “kanglaite” yielded 47 studies, including 13 meta-analyses. Among these meta-analyses, four evaluated the effectiveness of Chinese herbal ingestion combined with conventional cancer treatment,18–21 and the remaining nine examined the efficacy of kanglaite compared to conventional treatment on survival, drug response, disease control, quality of life, or adverse effects.22–30 Among the meta-analyses of kanglaite, four reports described an increase in the CD3 + CD4 + subset of peripheral T cells.24,27,28,30
Coix Seed Effects on Systemic Conditions in Diseased Subjects
Two reports described the effects of coix seed on systemic conditions in diseased patients. One study reported that an intake of 30 g/day for 12 weeks improved the nutritional status (eg, albumin [ALB], triglycerides [TG], and body mass index [BMI]), inflammation (eg, C-reactive protein [CRP]), and symptoms including tiredness, anorexia, xerostomia, and abdominal distension in patients receiving peritoneal dialysis. 11 The other study reported that 3 weeks of replacing part of the carbohydrate source in subjects’ diets with 60 g/day of coix seed reduced TG, whereas the control group showed no change. Plasma total cholesterol (TC) and low-density lipoprotein cholesterol (LDL-C) were significantly decreased in both the coix seed-treated and control groups, while high-density lipoprotein cholesterol (HDL-C) concentrations increased in the control group but not in the coix seed treatment group. 10
The Effects of the Coix Seed Extract Kotaro
Four studies written in Japanese described trials assessing the effect of the coix seed extract Kotaro, which is approved as an ethical drug in Japan.5,12–14 A total of 6 g of the coix seed extract powder Kotaro (Kotaro Pharmaceutical Co., Ltd) contains 2.0 g of dried water extracts obtained from 6.0 g of coix seed conforming to the Japanese Pharmacopeia and 4.0 g of excipients (eg, cornstarch and lactose monohydrate). The coix seed extract tablet Kotaro contains 2.0 g of the dried water extract in 18 tablets—equivalent to 6.0 g of coix seed or 6.0 g of the coix seed extract powder.
One publication reported a multicenter, randomized, double-blind, placebo-controlled parallel-group comparative study for molluscum contagiosum. 12 In this study, 29 institutes participated. Either the coix seed extract powder Kotaro or placebo was administered to patients aged younger than 15 years for 12 weeks. The dose was 6 g/day for patients with a body weight of 30 kg or less and 12 g/day for those with a body weight over 30 kg. The coix seed extract was orally administered two or three times a day. During the intervention, oral administration or injection of other medicines was avoided, but topical agents were allowed for the treatment of atopic disease. The condition of the patients was examined after 4, 8, and 12 weeks of intervention. Coix seed extract-treated patients showed a lower residual ratio of tuberosity (83.5%) than those treated with the placebo (92.1%) at 4 weeks. However, no difference was found at 8 and 12 weeks of intervention. The effectiveness throughout the intervention period was not different between the coix seed extract and placebo. When stratified by sex, coix seed extract showed greater effectiveness than the placebo in male subjects (P = .01).
Fushiki et al reported the effect of coix seed on the progression or regression of human papillomavirus infection, which causes cervical cancer. 5 The coix seed extract powder Kotaro (6 g/day) was administered to patients diagnosed with low-grade squamous intraepithelial lesions. The regression of the lesion to negative for intraepithelial lesions or atypical squamous cells of undetermined significance was considered to be normalized. The ratio of normalization was higher in the coix seed group (92.1%, 35 of 38) than that in the control group (54.2%, 13 of 24). The time of treatment to normalization was significantly lower in the coix seed group (6.1 months, range 2-15 months) than in the control group (25.8 months, range 8-42 months) (P < .0001).
Sugiura et al examined the effect of Kotaro on dry skin including eczema with low sebum.13,14 Eighteen tablets per day of the coix seed extract Kotaro was administered to patients for 12 weeks. During the intervention, treatment with antihistamine and topical agents was continued. The first report described the trial that was conducted from the spring to the autumn season (May 25, 2000 to October 19, 2000). 13 The coix seed extract treatment group showed a higher improvement (65.1% ± 27.7%) in pruritus than the control group (41.7% ± 20.0%; P < .05). The effectiveness was also higher in the group treated with coix seed extract (P < .05). The next report described a trial conducted from the summer to the winter season (August 30, 2001 to January 24, 2002). 14 Coix seed extract again improved pruritus compared to the controls (79.3% vs 44.9%, P = .0246). Coix seed extract also improved the surface structure of the skin. However, coix seed extract did not normalize the corneum moisture and pH or increase sebum production.
Coix Seed Extract Effects on the Condition of the Skin
The preparation of coix seed extract was reported by two independent groups, which examined its effects on the skin condition of healthy subjects.6,15 First, Suzuki et al prepared a water extract of whole coix seed, which they called coix-seed reactive derivatives (CRD). 15 A randomized controlled trial was conducted to determine the effects of CRD on facial pigmentation. Healthy female subjects with facial pigmentation were given either 2 or 4 g/day of CRD or a placebo as chewable tablets for 8 weeks. Facial pigmentation was reduced in the CRD group and increased in the placebo group (P < .05; analysis of variance [ANOVA]), and pairwise comparison showed a significant difference between the CRD (4 g/day) and placebo groups (P = .006). Erythema also decreased in the CRD group, but increased in the placebo group (P = .066; ANOVA). Pairwise comparison of the change in erythema showed a significant difference between the CRD (4 g/day) and placebo groups (P = .009).
Goto et al conducted a study to determine the effect of an extract of the coix seed endosperm. 6 A total of 660 mg of the extract was obtained from 15 g of coix seed, which conforms to the Japanese Pharmacopeia. A randomized controlled trial was conducted to determine the effect of the extract on the skin condition (eg, sebum, moisture, melanin, and elasticity) of the left arm, right cheek, and forehead. Healthy female subjects were administered either 1000 or 1500 mg/day of the extract or placebo for 12 weeks. The condition of the skin was assessed before and at 4 and 12 weeks after the intervention. The participants receiving 1500 mg/day exhibited significantly lower sebum at 12 weeks than those in the placebo group (P < .05).
Glycemic index
There were two publications reporting the glycemic index of cooked coix seed. In the first study, Lin et al reported a glycemic index of 55.3, although the cooking method was not described. 16 In the second study, Zhu et al examined the glycemic index of steamed coix seed after steaming for 30 and 60 min, which were 91 ± 10 and 100 ± 11, respectively. 17
Specific Substances in Coix Seed
Coixenolide
Coixenolide, (11E)-11-octadecenoic acid (1S,2R)-1-methyl-2-[[(9Z)-1-oxo-9-hexadecenyl]oxy]propyl ester (Figure 3A), was discovered as a substance responsible for antitumor activity in Ehrlich ascites sarcoma in mice.31,32 However, a study published in 1997 explored fat-soluble substances in coix seed to identify TG, free fatty acids, and phospholipids, but could not identify coixenolide. 33 The authors also synthesized coixenolide and found no corresponding substances in the ethanol extract of coix seed. 33

Structures of coix (Coix lacryma-jobi Linné. var. mayuen Stapf) seed-specific substances. (A) Coixenolide; (B) Coixol; (C) Coixide A; (D) trans-coniferyl aldehyde. Images A and D were obtained from KEGG COMPOUND (https://www.kegg.jp/kegg/compound/) and B and C were drawn using the online tool MolView (https://molview.org/).
Among the other studies, only one examined the effect of coixenolide and reported that it suppressed the decrease in Foxp3 + CD4 + CD25 + Treg levels in a mouse arthritis model. 34
Coixol
Coixol, 6-methoxy-2-benzoxazolinone (Figure 3B), was first isolated from coix root 35 and its structure was confirmed by organic synthesis. 36 Coixol was determined in a fraction of the methanol extract of coix seed that inhibited the growth of HeLa cells. 37 Cytotoxicity was also reported in B16-F10 mouse melanoma cells, 38 and in the human breast cancer cell lines MCF-7, MDA-MB-231, and T-47D. 39
In 1981, Sanders et al reported that coixol worked as a reproductive trigger in rodents. 39 Since then, 26 reports have been published, but the studies were limited to rodents.
Coixol was demonstrated to be an insulin secretagogue for isolated mice islets and the aMIN-6 pancreatic β-cell line. 40 Coixol enhanced insulin secretion in isolated and perfused mouse islets at a high glucose level, despite that it did not affect the basal glucose level. 41 The effect was inhibited by a protein kinase A inhibitor H-89 and an exchange protein directly activated by cAMP 2 (EPAC2) inhibitor MAY0132. 41 Therefore, coixol enhanced insulin secretion via the cAMP-mediated pathway. 41 However, the effect has not been tested and confirmed in humans.
Coixol has also been reported to affect airway mucin production and secretion in the human mucoepidermoid pulmonary carcinoma cell line NCI-H292, 42 behavioral and EEG effects in mice and rats, 43 and endoplasmic reticulum stress in Chang Liver (HeLa derivative) cells. 44
Recently, coixol has been reported to modulate the immune response. Hu et al showed that coixol suppressed the expression of interleukin (IL)-1β, IL-6, IL-18, tumor necrosis factor-α (TNF-α), nitric oxide (NO), inducible NO synthase, and cyclogenase (COX)-2, but not L-10 in lipopolysaccharide (LPS)-stimulated RAW264.7 cells. 45
Other Substances
A novel compound Coixide A, (7R,8S)-3’-demethyl-9′-methoxy-dehydrodiconiferyl alcohol-3'-O-β-glucopyranoside (Figure 3C), was identified in coix root by Kim et al 46 However, its physiological effect on mammals has not been reported. The same study also identified coixol and 14 other known compounds. 46
Trans-coniferyl aldehyde (Figure 3D) was identified from the acetone extract of the hull of coix seed as a potent antimutagenic compound. 44 The same study also identified coixol, p-hydroxybenzaldehyde, vanillin, syringaldehyde, and sinapaldehyde. 44
Discussion
Traditional Chinese herbal medicines have been empirically determined to be effective and thus have been used throughout history. Most traditional Chinese medical regimens are combinations of various ingredients, including from both plant and animal sources. The mechanism of action has been clarified for a few of these regimens. In contrast, coix seed is often used as a single source drug. According to a dictionary of Chinese medicine, coix seed has the actions of diuretic infiltration, detoxification and drainage against lung tumors, relief of pain caused by muscle cramps, and enhanced healing of flat warts. 1 Despite its use as a single source drug, the effectiveness and mechanism of coix seed effects have not been clarified. Therefore, this review comprehensively collected and summarized literature written in English and Japanese describing human trials with controls that assessed the effect of coix seed from PubMed, CiNii, ClinicalTrials.gov, and UMIN-CTR.
Many studies on kanglite, a purified oil extract from coix seed, were found. Meta-analyses of kanglaite emphasized its effectiveness in cancer therapy,18–30 and four studies described an increase in the CD3 + CD4 + subset of peripheral T cells.24,27,28,30 However, the mechanism or substances responsible for this effect were unknown because the chemical composition has not been published. In addition, we could not analyze its composition because kanglite is not available in Japan. Therefore, we did not discuss kanglite further in this study. The chemical composition should be clarified in a future study.
In Japan, coix seed is approved as the ethical drug Yokuinin. The Japanese Pharmacopeia lists Yokuinin, powdered Yokuinin, and its extract as approved ethical drugs. The typical adult dose is 3.0 to 6.0 g/day orally in three divided doses adjusted according to the patient's age and symptoms. Coix seed and its extract are generally used to treat verrucae planae juveniles (plane warts) and verruca vulgaris (common warts).
Plane warts and common warts are localized infections of the papillomavirus. Human papillomavirus infection is relatively common in young women. Spontaneous recovery is observed in about 90% of infections within 3 years, whereas long-term infection causes cervical cancer. 47
Human papillomavirus infects skin epithelial cells, but does not induce lysis of the infected cells. The viral antigen has little opportunity to encounter antigen-presenting cells, such as dendritic cells. Spontaneous regression is considered to occur when antigen exposure or inflammation triggers an immune response. 47 Molluscum contagiosum is also a skin infection caused by the virus Molluscum contagiosum. Similar to human papillomavirus infection, most cases of molluscum contagiosum regress without medical intervention.
Fushiki et al reported that coix seed extract enhances recovery from human papillomavirus infection. 5 A multicenter, randomized, double-blind, placebo-controlled parallel-group comparative study of molluscum contagiosum conducted in Japan also observed the possible enhancement of spontaneous regression of the lesion. 12 These results consistently suggest that coix seed extract enhances the spontaneous regression of viral infections of the skin.
In Japan, coix seed is believed to improve the condition of the skin, eg, dry skin, seborrheic dermatitis, and pigmentation. Thereby, cosmetics and dietary supplements are popular. Sugiura et al conducted two trials to confirm that coix seed improved pruritus in patients with dry skin.13,14 However, changes were not observed in the moisture, sebum, or pH of the skin.13,14 Goto et al reported that coix seed extract did not affect the moisture, melanin, and elasticity of the skin, while it lowered the sebum level in healthy women. 6 Only one study reported that coix seed extract significantly suppressed facial pigmentation and erythema in healthy women, 15 but the method used to prepare the extract was not clarified. Therefore, coix seed could affect the condition of the skin, but indices used to confirm the effect have not been determined. As immune dysfunction underlies the skin barrier and chronic pruritus, 48 it may be worthwhile to evaluate these changes in terms of immune indices.
Two studies reported improvements in the systemic conditions of patients with renal dysfunction and hyperlipidemia with ingestion of coix seed.10,11 However, each of these studies was unique and had no additional supportive studies.
Coix seed is a type of grain that is traditionally ingested. Two studies reported the glycemic index of cooked coix seed as 55.3 16 and 91 to 100, 17 which showed that its digestibility depends on the cooking method.16,17
As described above, a comprehensive review of human studies indicates that coix seed may enhance the spontaneous regression of viral skin infections and may also reduce pruritus. Additionally, both effects could be exerted via alteration of the immune system. However, immune effects have not been thoroughly examined due to difficulties in quantifying the effects of coix seed on skin symptoms and the lack of clear indicators of its effects.
When we look at the effects of coix seed on immune parameters, meta-analyses of kanglaite studies consistently support an increase in peripheral CD4 + T cells in cancer patients,24,27,28,30 although the composition of kanglite has not been described. Furthermore, coixol has been reported to modulate cytokine expression, 45 although the effects on immune cells in vivo have not been examined. Therefore, the effect of coix seed on immune cells also remains to be elucidated.
In this review, literature on clinical trials on coix seed was comprehensively summarized. However, there are limitations. First, the vast majority of studies originated from Eastern countries; thus, extrapolation of these results to Western populations warrants further research. Second, coix seed lacks a standard regimen, except for Kotaro in Japan. There is significant heterogeneity in regimens, doses, duration, center settings, and populations enrolled in the literature listed in this review, and so the results need to be interpreted cautiously. In addition, many of the studies had a significant risk of bias, and the effects were assessed by limited studies. Therefore, the evidence to support the effects remains low and needs further study.
Conclusion
Unlike many herbs used in Chinese traditional medicines, coix seed is often used as a single agent. This comprehensive review of human trials suggests that coix seed promotes the spontaneous regression of viral infections of the skin and may have a cognitive improvement on skin conditions. Meanwhile, kanglite, a purified oil agent used for cancer therapy, has been shown to increase the ratio of CD4 + T cells in the peripheral blood of cancer patients undergoing treatment. The above studies seem to indicate that coix seed may affect cellular immune function. However, there are limitations in the individual studies on which the above consideration is based, thereby it is not conclusive what specific immune or clinical indices coix seed affects. In addition, although substances such as coixol have been reported to be specific to coix seed, their effects on human immune function have not been clarified. Therefore, further research is required to clarify these issues. Thus, coix seed seems promising for health promotion and presents a challenging and attractive theme for natural product chemists, pharmacologists, immunologists, and clinicians.
Footnotes
Acknowledgments
Authors’ contributions
YS conceived and drafted the manuscript. YS conducted the database search and collected the candidate literature. YS and YK selected the articles involved in this review. YK evaluated the risk of bias. All authors (YS and YK) read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the grant from the Project of the National Agricultural Research Organisation (NARO) Bio-oriented Technology Research Advancement Institution (Research program on the development of innovative technology: 30032C).
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
