Abstract
Two new α-methoxy-γ-pyrone analogs, 2-methoxy-3-methyl-5,6-diethyl-γ-pyrone (
Introduction
Mangrove forest, distributed in the transition between land and sea, possesses extensive microbial diversity and has the potential to discover new bioactive natural products, including those with potential medicinal application.1–3 A serious of natural products with novel structure and significant activity have been reported from the microbial community, isolated from the sediment, leaves, branches, and roots.4–6 Several studies have shown the uniqueness of mangrove sediments with respect to their microbialcomposition.7,8 Soil or sediment samples collected in mangroves forests showed a high diversity of associated microbes due to their unique ecosystem.9,10
During the course of our search for novel lead compounds, the mangrove sediment-derived strain Streptomyces psammoticus SCSIO NS126 was revealed with important potential medicinal value in our previous study. Twenty-seven natural piericidins were obtained in this strain with antirenal cell carcinoma activities. 11 In order to further explore the comprehensive secondary metabolites of this strain, careful chemical separation study was taken to obtain the different types of compounds except the piericidins. Herein, we report the isolation and structural elucidation of those compounds.
Results and Discussion
The strain was fermented and then harvested by extraction with EtOAc. The extract was subjected to repeated silica gel column chromatography (CC) followed by semipreparative high-performance liquid chromatography (HPLC). As a result, a α-methoxy-γ-pyrone compound firstly isolated from natural sources, 2-methoxy-3,5-dimethyl-6-ethyl-γ-pyrone (
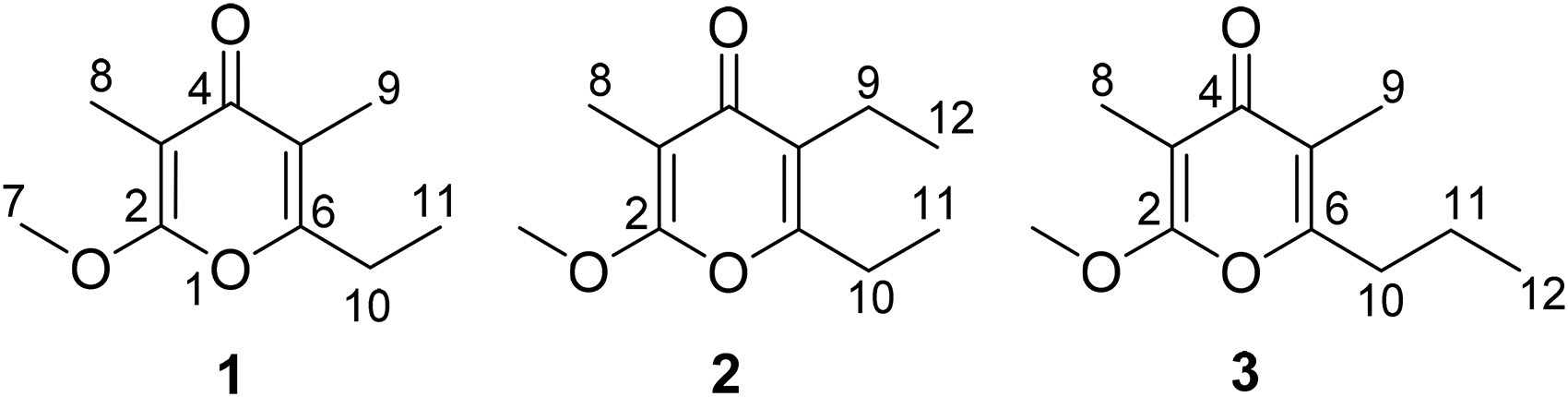
The structures of α-methoxy-γ-pyrones
Compound
1H (700 MHz) and 13C NMR (175 MHz) NMR Data of

Two α-methoxy-γ-pyrone analogs,

Key COSY, HMBC, and NOESY correlations of
Those α-methoxy-γ-pyrones (
Because this strain produces a rich variety of natural piericidins compounds, 11 the biosynthetic relationship between those α-methoxy-γ-pyrone analogs and piericidins is now discussed. The formation of the pyridone ring in piericidinsis dependent on the amidation and cyclization of the linear β,γ-diketo carboxylic acid, and ATP-dependent amidotransferase PieD plays a key role in introducing the nitrogen into the pyridone ring in piericidin. 14 The only difference of pyrone ring formation in the biosynthetic pathway is the absence of amidation. 15 Actinopyrones with α-methoxy-γ-pyrone ring, such as actinopyrones A-D,16,17 M050511,PM050463, PM060054, and PM060431, 18 were also discovered from Streptomyces strains. Not only the chemical structural similarity, actinopyrones showed resembling antibiotic/antitumor activities and related biosynthetic pathway with piericidins.18,19 It's interesting that 3 α-methoxy-γ-pyrone analogs have been discovered in this piericidins productive strain, without actinopyrones metabolites.
Experimental
General Experimental Procedures
UV spectra were recorded on a Shimadzu UV-2600 PC spectrometer (Shimadzu). IR spectra were measured on an IR Affinity-1spectrometer (Shimadzu). The NMR spectra were obtained on a Bruker Avance spectrometer (Bruker) operating at 700 MHz for 1H NMR and 175 MHz for 13C NMR, using tetramethylsilane as an internal standard. HRESIMS spectra were collected on a Bruker mix is TOF-QII mass spectrometer (Bruker). Thin layer chromatography (TLC) and CC were performed on plates precoated with silica gel GF254 (10-40 μm) and over silica gel (200-300 mesh) (Qingdao Marine Chemical Factory) and Sephadex LH-20 (Amersham Biosciences), respectively. All solvents employed were of analytical grade (Tianjin Fuyu Chemical and Industry Factory). The semipreparative HPLC was performed on an HPLC (Hitachi-L2130, diode array detector, Hitachi L-2455, Tokyo, Japan) using a Phenomenex Octadecylsilyl (ODS) column (250 mm × 10.0 mm i.d., 5 μm; Phenomenex). The artificial sea salt was a commercial product (Guangzhou Haili Aquarium Technology Company).
Bacteria Material
The strain information and the fermentation have been reported in literature. 11
Extraction and Isolation
The culture broth of this strain was extracted with an equal volume of EtOAc 3 times. The organic extract was then concentrated under vacuum to afford the EtOAc extract (38.2 g). The extract was subjected to silica gel vacuum liquid chromatography using step gradient elution of petroleum ether (PE)–CH2Cl2 (1:0, 2:1, 0:1), CH2Cl2–MeOH (200:1, 100:1, 50:1, 30:1, 0:1) to yield 8 fractions according to TLC profiles (Frs.B1–B8). Frs.B4 (205 mg) was separated into 4 subfractions (Frs.B4-1–B4-4) by ODS silica gel chromatography eluting with MeCN/H2O (5%-100%). Frs.B4 to 3 (20 mg) was directly separated by semipreparative HPLC (30% MeCN/H2O, 2 ml/min, 280 nm) to provide
2-Methoxy-3,5-dimethyl-6-ethyl-γ-pyrone (
2-Methoxy-3-methyl-5,6-diethyl-γ-pyrone (
2-Methoxy-3,5-dimethyl-6-propyl-γ- pyrone (
AChE Inhibitory Bioassay
AChE inhibitory bioassay was assayed using Ellman method and the enzyme was from Saccharomyces cerevisiae, Sigma Aldrich by a spectrophotometric method. Tacrine was used as a positive control. 13
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Special Funds for Promoting Economic Development (Marine Economic Development) of Guangdong Province (GDOE[2019]A28, GDNRC2021052), Natural Science Foundation of Guangdong Province (2021A1515011711), Key-Area Research and Development Program of Guangdong Province (2020B1111030005), National Natural Science Foundation of China (U20A20101, 31900286, 81973235).
