Abstract
Atractylodes macrocephala (AM) is a famous traditional Chinese medicine for intestinal epithelial restitution through activating Ca2+ channels. However, the roles of specific AM compositions in intestinal epithelial restitution are sparse. Therefore, this study aimed to compare the concrete effects of the 4 active ingredients (atractylon, β-eudesmol, atractylenolide II, atractylenolide III) of AM and their combination on intestinal epithelial repair and the Ca2+ pathway in intestinal epithelial cell (IEC-6) cells. First, the best combination of the 4 ingredients with an optimal mixing ratio of atractylon: β-eudesmol: atractylenolide II: atractylenolide III = 1:2:2:2 was demonstrated by a 3-[4,5-dimethylthiazol-2-yl]-2,5 diphenyl tetrazolium bromide orthogonal experiment. Subsequently, enzyme-linked immunosorbent assay was used to measure anti-inflammatory cytokine levels, the migratory ability was evaluated by cell scratch experiments, cell cycle analysis and [Ca2+]cyt concentration in cells were detected by flow cytometry, and the expression of the Ca2+ pathway-related genes was detected by immunofluorescence staining, quantitative polymerase chain reaction and whole blood assays. Our result showed that atractylon, β-Eudesmol, atractylenolide II, and atractylenolide III showed different abilities to promote the IEC-6 cells proliferation, migration, and the expression of anti-inflammatory cytokines interleukin (IL)-2, IL-10, and ornithine decarboxylase, as well as the intracellular [Ca2+]cyt concentration through stromal interaction molecule 1 transposition to activate Ca2+ pathway. Thereinto, atractylenolide III was the main active ingredient of AM for pro-proliferation and anti-inflammation, and the combination of 4 AM ingredients performed better beneficial effects on IEC-6 cells. Therefore, our study suggested that atractylenolide III was the active ingredient of AM for intestinal epithelial repair through activating the Ca2+ pathway, and the 4 ingredients of AM have a synergy in intestinal epithelial repair.
Introduction
The gastrointestinal (GI) tract acts as one of the greatest barriers between the external and internal environments and the integrity of this barrier plays a pivotal role in maintaining homeostasis.1-3 Intestinal mucosal is not only involved in the digestion and absorption of nutrients, mostly, provides a selectively permeable barrier against endogenous and exogenous antigens.4,5 Intestinal barrier defects have been shown in a broad range of diseases, containing inflammatory bowel disease (IBD), colon carcinoma, chronic liver disease, type 1 diabetes, obesity, and so on. 6 Remarkably the basic structure of barriers between the environment and the internal host milieu is formed by mucosal epithelial cells.7,8 During the early epithelial restitution of the GI mucosa, the functions of intestinal epithelial cells (IECs) including migration and metabolism are important.6,9-14
The cytosolic free Ca2+ ([Ca2+]cyt) plays a significant role in the regulation of epithelial restitution after injury.13,15,16 Experiments in vitro and in vivo have shown that [Ca2+]cyt promotes intestinal epithelial restitution after wounding. 17 [Ca2+]cyt transport is regulated by the Ca2+ channels at the cell. 18 Canonical transient receptor potential 1 (TRPC1) are store-operated Ca2+ channels that control the balance of [Ca2+]cyt to influence epithelial restitution. 15 Stromal interaction molecule 1 (STIM1) and stromal interaction molecule 2 (STIM2) are the sensors of Ca2+ within the store, acting as essential parts in regulating TRPC1-mediated Ca2+ influx after store depletion. 19
Atractylodes macrocephala (AM) is an important traditional Chinese medicine which is widely used to treat chronic intestinal disease. 20 Recently, there has been an increasing interest in exploring the pharmacological roles of this drug. Some studies have proved that AM promoted intestinal epithelial restitution through the Ca2+ channels.21,22 AM is comprised of many substances, however, the studies in the role of specific AM composition are sparse.
IECs play a key role in the initiation and perpetuation of intestinal inflammation. And the intestinal epithelial (enterocyte) cell line IEC-6 is often used as an intestinal inflammation model system in vitro. 23 Previous studies showed that atractylon, β-eudesmol, atractylenolide II, and atractylenolide III were the main compositions of AM,24-28 however, their impact on intestinal repair has been less studied. So, in this study, we aimed to compare the concrete effect of the 4 AM active ingredient atractylon, β-eudesmol, atractylenolide II, and atractylenolide III was as well as the combination on the intestinal epithelial repair through the Ca2+ pathway.
Result
The Pro-Proliferation of 4 AM Effective Ingredients for IEC-6 Cells
As shown in Figure 1, atractylon (10-160 μM), β-eudesmol (10-40 μM), atractylenolide II (10-40 μM), and atractylenolide III (10-20 μM) significantly increased the survival rate of IEC-6 cells after 24 h co-culture, while atractylon (10-160 μM), β-eudesmol (10-40 μM), atractylenolide II (10-20 μM) and atractylenolide III (10-20 μM) also showed a significant increase after 48 h co-culture. Notably, the excess β-eudesmol (≥ 80 μM), atractylenolide II (≥ 160 μM), and atractylenolide III (≥ 40 μM) were cytotoxic to normal cells.

The pro-proliferation of the 4 AM effective ingredients for IEC-6 cells. IEC-6 cells were incubated with atractylon, β-eudesmol, atractylenolide II, and atractylenolide III for 24 h or 48 h, respectively. After incubation, the survival rate was determined by MTT assay. n = 6. * denote P < .05 versus the control group (0 μM of active ingredients of AM).
Analysis of the Optimal Mixing Ratio to Promote the IEC-6 Cells Proliferation
According to the results of 3-[4,5-dimethylthiazol-2-yl]-2,5 diphenyl tetrazolium bromide (MTT) (Figure 2), the concentrations from 10 to 40 μM were employed for the L9 (34) orthogonal test to choose the optimal mixing ratio of the 4 active ingredients. The result of the L9 (34) orthogonal test showed that the best combination to promote the IEC-6 cells proliferation was consisted by 10 μM atractylon, 20 μM β-eudesmol, 20 μM atractylenolide II, and 20 μM atractylenolid III. Thus, the optimal mixing ratio was atractylon: β-eudesmol: atractylenolide II: atractylenolide III = 1:2:2:2.

Analysis for the optimal mixing ratio to promote the IEC-6 cells proliferation
The Anti-Inflammatory Effect of 4 AM Effective Ingredients and Their Combination for IEC-6 Cells
The results of enzyme-linked immunosorbent (ELISA) for anti-inflammatory cytokines interleukin (IL)-2, IL-10, and ornithine decarboxylase (ODC), as shown in Figure 3, atractylon could only increase the ODC expression, and β-eudesmol only increased the IL-2 expression. While, both atractylenolide III and the combination with 1:2:2:2 mixing ratio significantly increased the expressions of anti-inflammatory cytokines IL-2, IL-10, and ODC. Moreover, the combination with 1:2:2:2 mixing ratio had a higher expression of IL-10 and ODC compared to atractylenolide III.

The anti-inflammatory effect of 4 AM effective ingredients and their combination for IEC-6 cells. IEC-6 cells were incubated with atractylon, β-eudesmol, atractylenolide II, atractylenolide III, and the combination of 1:2:2:2 mixing ratio treatments in a final concentration of 20 μM for 24 h, respectively. Then, the cell culture supernatants were collected to detect the anti-inflammatory cytokines IL-2, IL-10, and ODC by ELISA kits. *, **, *** denote P < .05, P < .01, and P < .001 versus the control group. #, ##, ### denote P < .05, P < .01, and P < .001 versus combination group.
The Pro-Cell Motility and Cell Cycle of 4 AM Effective Ingredients and Their Combination for IEC-6 Cells
The results of the wound healing assay showed that the cell motility of IEC-6 cells was promoted by atractylon, β-eudesmol, Atractylenolide II, Atractylenolide III, and their combination. Thereinto, atractylenolide III performed a better pro-motility on ICE-6 cells in the 4 ingredients, while, the promotive effect of their combination was better than all the ingredients (Figure 4A). The results of the cell cycles as shown in Figure 4B, the 4 ingredients of AM and their combination could significantly increase the cells %age of G1 phase, while atractylon, atractylenolide III, and their combination decreased the cells %age of S phase in IEC-6 cells. Of them, the pro-cell cycle progression effects of atractylenolide III and atractylon were more obvious than the other 2 ingredients, and the pro-cell cycle progression effects of their combination were the most prominent in all groups. Those suggested that atractylon, atractylenolide III, and their combination promoted the proliferation of IEC-6 cells through advancing the cell cycle progression.

The effects on cell motility and cell cycle. (A) Wound healing assay. The scratch-wound was drawn and IEC-6 cells were incubated with ingredients (total 20 μM per group) or not for 24 h, n = 6. (B) Cell cycle analysis. After 24 h incubation with ingredients (20 μM) or not, PI was used as a stain, and the fluorescence was acquired with an Accuri C6 flow cytometer, n = 3. *, **, *** denote P < .05, P < .01, and P < .001 versus the control group. #, ##, ### denote P < .05, P < .01, and P < .001 versus combination group.
The Effects on STIM1 and STIM2 Expression in IEC-6 Cells
The result in Figure 5A showed that atractylon, β-eudesmol, atractylenolide II, atractylenolide III, and the combination all promoted the translocation of STIM1 to the plasma membrane, and the effect of atractylenolide III on the translocation of STIM1 was better than the other 3 ingredients, while the combination had a better effect than all ingredients. The results of quantitative polymerase chain reaction (q-PCR) and whole blood (WB) showed that atractylon, β-eudesmol, atractylenolide II, atractylenolide III, and the combination all had a significant impact to up-regulate STIM1 expression and down-regulate STIM2 expression. Similarly, the effect of atractylenolide III on STIM1 and STIM2 expressions was more obviously than the other 3 ingredients, and the combination had the largest effect in all groups (Figure 5B).

The effects of 4 AM effective ingredients and their combination on STIM1 and STIM2 expression. IEC-6 cells were incubated with ingredients (total 20 μM per group) or not for 24 h. After incubation, IEC-6 cells were determined by immunofluorescence staining WB and q-PCR, n = 3. (A) The immunofluorescence staining results of STIM1. (B) The WB results of STIM1 and STIM2 protein expression. (C) The q-PCR results of STIM1 and STIM2 mRNA expression. *, **, *** denoted P < .05, P < .01, and P < .001 versus the control group. #, ##, ### denote P < .05, P < .01, and P < .001 versus combination group.
The Effects of 4 AM Effective Ingredients and Their Combination on Ca2+ Pathways in IEC-6 Cells
As shown in Figure 6A, atractylon, atractylenolide III, and the combination significantly increased the [Ca2+]cyt concentration of IEC-6 cells, and the effect of atractylenolide III on [Ca2+]cyt concentration of IEC-6 cells was better than atractylon, while the [Ca2+]cyt cell concentration of the combination was highest in all the groups. However, there was no difference in the [Ca2+]cyt concentration after β-eudesmol and atractylenolide II treatments. As shown in Figure 6B, atractylon and atractylenolide II only significantly increased the expression of TRPC1 and RhoA protein, and β-eudesmol and atractylenolide III significantly enhanced the expression of TRPC1, PLC-γ1, and RhoA proteins, while atractylenolide III demonstrated a higher protein expression level than β-eudesmol. Moreover, the combination exhibited the strongest promoting effect on TRPC1, PLC-γ1, and RhoA proteins.
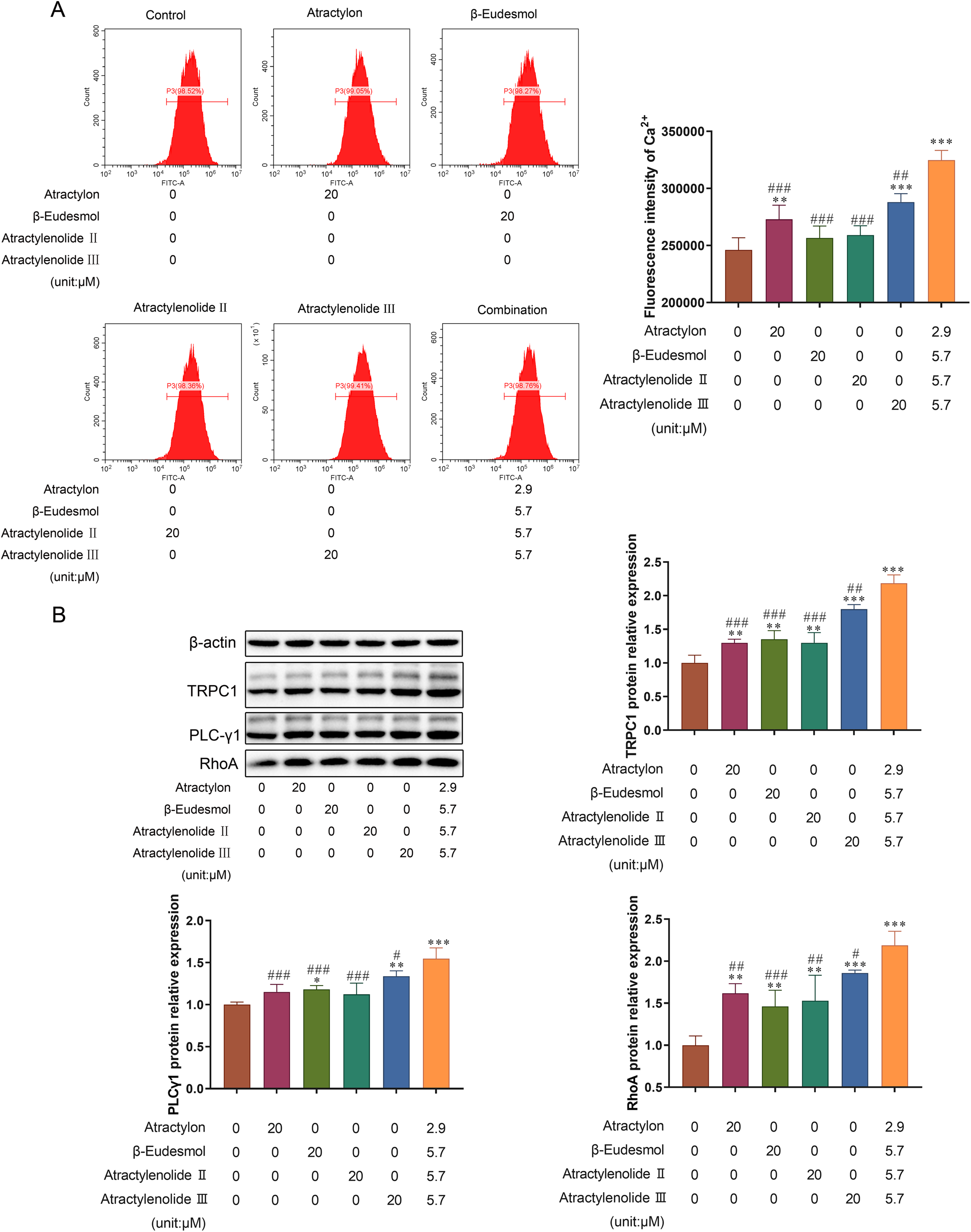
The effect on Ca2+ pathways. IEC-6 cells were incubated with ingredients (total 20 μM per group) or not for 24 h. After incubation, IEC-6 cells were determined by flow cytometry and WB, n = 3. (A) The result of [Ca2+]cyt concentration in cells. (B) The effects on TRPC1, PLC-γ1 and RhoA protein expressions in Ca2+ pathways. *, **, *** denote P < .05, P < .01, and P < .001 versus the control group. #, ##, ### denote P < .05, P < .01, and P < .001 versus combination group.
Discussion
AM, as a famous Chinese medicine used to treat chronic intestinal disease, has been known to repair intestinal epithelium through the Ca2+ channels.21,22 However, there were fewer studies to elucidate the specific roles of concrete ingredients involved in this process. In this study, we investigated the role of the 4 ingredients atractylon, β-Eudesmol, atractylenolide II, and atractylenolide III of AM and their best combination on the intestinal epithelium repairment. Based on the results, we found that the 4 ingredients of AM could promote the proliferation of IEC-6 cells, but the excess β-eudesmol (≥ 80 μM), atractylenolide II (≥ 160 μM), and atractylenolide III (≥ 40 μM) had cytotoxicity to normal cells. Furthermore, we found that the 4 ingredients all had anti-inflammatory, pro-cell motility, and pro-cell cycle effects through activating Ca2+ pathways, while atractylenolide III exerted the most prominent effect in them. Additionally, we confirmed the best combination of the 4 AM ingredients to promote the proliferation of IEC-6 cells, which was formed by a 1:2:2:2 mixing ratio of the 4 AM ingredients, and the promotive effect of the 4 ingredients combination on proliferation, was better than they used separately.
The GI tract is the most susceptible target to inflammatory responses, 29 and IBD are more likely to cause intestinal epithelial injury. 30 Thus, the anti-inflammatory activity of intestinal epithelium is particularly important to intestinal epithelial repair. It has been reported that atractylenolide I to III had an anti-inflammatory activity for the GI tract. 31 ODC, as a critical regulator of M1 macrophage activation, could inhibit M1 macrophage activation, 32 and IL-2 and IL-10 are anti-inflammatory cytokines, which play a vital role in the repair of intestinal epithelial cells. In this study, we found that atractylon, β-eudesmol, atractylenolide II, atractylenolide III, and the combination all had an anti-inflammatory effect for IEC-6 cells, but with different benefits in the anti-inflammatory cytokines IL-2, IL-10, and ODC. Only atractylenolide III and the combination of the 4 AM ingredients could significantly increase the expression of IL-2, IL-10, and ODC, while the combination of 4 AM ingredients was slightly better than all the individual AM ingredients. Therefore our result suggested that atractylenolide III had the best anti-inflammatory capacity in the 4 compositions, and the combination enhanced the anti-inflammatory activity.
Epithelial repair is the tight coordination of processes including migration, proliferation, and differentiation. 33 So, proliferation and migration are key events during the epithelial repair. In the previous study, it has been shown that AM Koidz could promote IEC-6 cell migration, which was a dose-dependently process. 21 Our study showed that atractylenolide III performed the best ability of IEC-6 cell migration in the 4 AM compositions and the combination of the 4 AM ingredients showed a better pro-cell motility and cell cycle for IEC-6 cells than any individual AM ingredients. In addition, cell proliferation depends on an orderly cell cycle process, 34 our study pointed out that the 4 compositions and the combination not only increased the number of G1 phase cells, but also reduced the number of S phase cells. Thereinto, atractylon, and atractylenolide III had a stronger promotive effect on IEC-6 cells and the combination of the 4 AM ingredients intensified the change in the cell cycle process. Therefore, we believed that atractylenolide III played a major role in cell proliferation and migration, and the 4 ingredients had synergistic effects. Apart from that, polyamines play an essential role in the regulation of cell differentiation and proliferation and tissue repair,35,36 and the intracellular levels of polyamines are highly regulated by the activity of ODC. 36 Song et al 21 found a significant increase of polyamines in IEC-6 cells with AM Koidz treatment, and thought the increase of polyamines was related to the promotional effect of AM Koidzs on IEC-6 cell migration. In our study, we observed the level of ODC in IEC-6 cells was increased, so it suggested that the contribution of atractylenolide III and their combination to IEC-6 cells proliferation and migration was connected to polyamine.
It has been reported that AM and its some active ingredients can repair intestinal epithelium through the Ca2+ pathway. For example, the study of Song et al 21 revealed that the treatment with AM Koidz significantly stimulated the migration of intestinal epithelial cells through polyamine-Kv1.1 channel signaling pathway, when intestinal epithelium got an injury. Zeng et al 22 found that the polysaccharide of AM Koidz extracts stimulated the migration of intestinal epithelial cells via a polyamine-Kv1.1 channel activated signaling pathway, which facilitated intestinal injury healing. Further, another study provided additional evidence suggesting that atractylenolide I promoted cell migration and proliferation, increased polyamines content, raised cytosolic free Ca2+ concentration, and enhanced TRPC1 and PLC-γ1 expression in IEC-6 cells. 18 However, there were few studies comparing the effects of the active ingredients of AM on the Ca2+ pathway. Therefore, we detected the effects of atractylon, β-eudesmol, atractylenolide II, atractylenolide III, and their combination to the Ca2 + pathway. As we expected, atractylenolide III significantly increased [Ca2+]cyt cell concentration and STIM1 expression and the up-regulated TRPC1, PLC-γ1, and RhoA levels, and their combination strengthened the effect.
Our data suggested that the 4 compositions of AM promoted intestinal epithelial repair by promoting Ca2+ influx and up-regulating the cell [Ca2+]cyt concentration, in particular, atractylenolide III exerted the most prominent effect. Moreover, our results highlighted a mutual facilitative interaction on the Ca2+ pathway among the 4 compositions and their combination was the most promising. However, the same type of research is still scarce, there is a lack of comparison of the research conclusions. In the future, we will take account of some aspects to research the effects of active components of AM and their combined use on intestinal epithelial repair.
In conclusion, our results demonstrated that atractylon, β-eudesmol, atractylenolide II, and atractylenolide III all promoted the IEC-6 cells’ abilities of proliferation, migration, and anti-inflammatory functions, and increased the intracellular [Ca2+]cyt concentration through the Ca2+ pathway, promoting intestinal epithelial recovery. Of these, atractylenolide III exerted the most prominent effect relatively and the improvement of the 4 ingredients combination use was better than using them alone. Therefore, we emphasized the contribution and treatment value of their combined use.
Methods
Reagents
The standards of the 4 active ingredients (atractylon, β-eudesmol, atractylenolide II, and atractylenolide III) were obtained from Chengdu Push Bio-Technology Co., Ltd Atractylon standard, CAS No 6989 to 21 to 5, purity≥98%, white or light yellow crystal; β-eudesmol, CAS No 473 to 15 to 4, purity≥98%, white acicular crystal; atractylenolide II, CAS No 73069 to 14 to 4, purity≥98%, white crystal; atractylenolide III, CAS No 73030 to 71 to 4, purity≥98%, white acicular crystal. The chemical structures of those compounds are provided in Figure 7.

Chemical structures of atractylon, β-eudesmol, atractylenolide II, and atractylenolide III.
Preparation
The IEC-6 cell was purchased from the American Type Culture Collection (NO. 63139935). The cells were cultured with high-glucose Dulbecco’s modified Eagle's medium (high-glucose DMEM) containing 100 units/mL penicillin, 100 μg/mL streptomycin, which facilitated intestinal injuries of 95% air-5% CO2 at 37°C. Cell subculturing was performed when the cells reached 80% to 90% coverage. The 16th to 19th passages of IEC-6 cells were used for this experiment.
The compound standards were weighed accurately and added to a volumetric flask (100 mL). Then the compound standards were dissolved in DMEM, sonicated, and the final volume was made up to 100 mL with DMEM. Finally, the solution was filtered through a 0.22 µm filter. The weight of the compound standards was determined by the required concentration of the test, and the formula is:
The compound standard weight (g) = C × V × M × 1 × 10−6, where C: experimental concentration (μM); V: the volume of a volumetric flask (0.1 L); M: the molar mass of the compound (g/mol).
3-[4,5-dimethylthiazol-2-yl]-2,5 diphenyl tetrazolium bromide (MTT)
IEC-6 cells in the logarithmic phase were enzymatically digested to a single-cell suspension. The cells were plated at 2 × 105 cells per well to 6-well plates and allowed to attach. The complete DMEM separately containing various concentrations (0-160 μM) of β-eudesmol, atractylenolide II, atractylenolide III, and atractylon were added and incubated with IEC-6 cells for 24 h and 48 h. After culture, cell proliferation was assessed using an MTT assay, performed as previously described. 23 Briefly, added 20 μL MTT (5 mg/mL) for 4 h. Thereafter, 150 μL dimethyl sulfoxide (DMSO) was used to solubilize the dark blue crystals for 10 min. The results were monitored at 490 nm in a microplate reader (Bio-Rad). Cell growth proliferation rate was calculated as [A490 nm (treated cells)/A490 nm (control cells)−1] × 100%.
Orthogonal Experiment
According to the results of MTT, an L9 (34) orthogonal test was employed to choose the best combination of the 4 active ingredients. Each factor level is shown in Table 1 and the L9 (34) orthogonal table shown in Table 2. The control group was not given any intervention of the 4 active ingredients. Cell treatment was identical to the MTT assay described above and cultured for 24 h.
Factors and Levels for the L9 (34) Orthogonal Table.

The L9 (34) Orthogonal Table.

Following Experimental Groups
The following experiments were divided into 6 groups, containing the control group, atractylon group, β-eudesmol group, atractylenolide II group, atractylenolide III group, and the combination group (depended on the result of the orthogonal experiment) and the drug concentration was 20 μM.
Enzyme-Linked Immunosorbent
IEC-6 cells in the logarithmic phase were seeded in 6-well plates at a concentration of 2 × 105 cells per well and allowed to grow for 24 h. Then, 4 active ingredients and their combination were respectively added to the 6-well plate and cultured for 24 h. After this step, cell culture supernatants were collected for ELISA. ELISA assays were determined for IL-2, IL-10, and ODC by ELISA kits (Abcam) according to instructions. 37 Briefly, samples were added to 96-well plates precoated with antibodies. After several incubation steps, washing procedures, and a final incubation with a chromogenic substrate, the result was measured with a microplate reader (Bio-Rad).
Wound Healing Assay
Wound healing assay migration was tested by a wound-healing assay. 38 IEC-6 cells in the logarithmic phase were seeded in 6-well plates at a concentration of 2 × 105 cells per well and allowed to grow for 24 h, reaching a cover of 80%. The scratch-wound was drawn perpendicularly with a 10 μL pipette tip. PBS was used to wash off damaged cells in plates. Subsequently, the complete medium (including 4 μg/mL mitomycin) with different active ingredients based on the above grouping information was treated for 24 h. Photomicrographs of the cell migration were taken with an Olympus inverted phase-contrast microscope (Japan). The relative wound recovery ratio of the cell was calculated as follows: relative wound recovery ratio (%) = [distance within scratch (0 h)−distance within scratch (24 h)]/distance within scratch (0 h).
Cell Cycle Analysis
Cell cycle analysis was detected by the BD Cycletest Plus DNA kit. 39 IEC-6 cells in the logarithmic phase were seeded in 6-well plates at a concentration of 2 × 105 cells per well and allowed to grow for 24 h. The respective medium was treated for 24 h with complete DMEM separately containing different active ingredients based on the above grouping information. After digestion with trypsin, 200 µL solution of ice-cold PI stain or PBS was added into 6-well plates for 10 min at the temperature 37°C in the dark. Finally, the cells were washed and the fluorescence was acquired with an Accuri C6 flow cytometer. The %ages of cells in the G1, S, and G2 phases were determined.
Flow Cytometry for Ca2+
IEC-6 cells in the logarithmic phase were seeded in 6-well plates at a concentration of 2 × 105 cells per well and allowed to grow for 24 h. The respective medium was treated for 24 h with complete DMEM separately containing different active ingredients based on the above grouping information. Ca2+ concentration in cells was tested by flow cytometry. 40 Brief, the upper culture solution was discarded carefully and the remaining cells were washed twice with PBS. Then 5 μL fluo-3 AM (2 mmol/L) was added into 6-well plates for 45 min at the temperature 37°C. Finally, the cells were washed and the fluorescence was acquired with an Accuri C6 flow cytometer.
Quantitative Polymerase Chain Reaction (q-PCR)
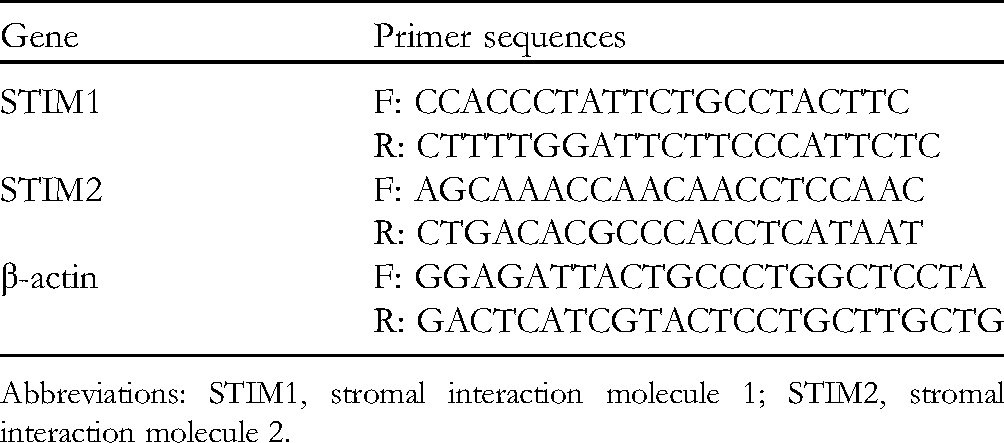
The cell culture and the treatments were the same as that of flow cytometry. Total RNA was collected by TRIzol RNA extraction kit (Invitrogen) and RNA integrity and purity were determined by gel electrophoresis and NanoDrop 2000 spectrophotometry (Thermo). Reverse transcription was conducted using a reverse transcription kit (Invitrogen) as described by the manufacturer. Then the resulting cDNA was used for quantitative analysis by q-PCR, assessed by a Qubit spectrofluorimeter (Invitrogen). The primer sequences are shown in Table 3. Gene expression was normalized to β-actin.
Primer Sequences.
Abbreviations: STIM1, stromal interaction molecule 1; STIM2, stromal interaction molecule 2.
Whole Blood
The cell culture and the treatments were the same as that of flow cytometry. The total protein was extracted from the cells. Proteins were separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis and transferred electrostatically to polyvinylidene difluoride membranes. The membranes were blocked with 5% nonfat milk in TBS (50 mM Tris, pH 7.6, 0.9% NaCl, and 0.1% Tween-20) at room temperature, and then incubated overnight at 4°C with primary antibody against. The membranes were washed with TBS and then a second antibody with HRP labeled was combined. After washing again, the protein bands were visualized by a Western ECL Substrate (Bio-Rad) and the result was determined by Image Lab software (Bio-Rad).
Immunofluorescence Staining
The cell culture and the treatments were the same as that of flow cytometry. At the end of the culture time, cells were fixed using 3.7% formaldehyde. The slides then were blocked with 2% BSA for 30 min. A primary antibody against STIM1 (1:300) was added and incubated overnight. After 3 washes, a secondary antibody Alexa Fluor-594 (Invitrogen, USA) was added for 2 h. Finally, DAPI nuclei staining was performed. The slides were viewed and photographed under a DM2000 fluorescence microscope (Leica).
Statistical Analysis
All values were expressed as means ± SD, and SPSS 19.0 software was used for analysis. Differences in mean values were determined by one-way analysis of variance test. Then, multiple comparisons between the groups were determined using the least significant difference’s multiple range test. P < .05 was accepted as statistical significance.
Footnotes
Acknowledgments
This study was supported by the Joint Fund Project of Science and Technology Department of Guizhou Province (LH 2017, Qian).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Joint Fund Project of Science and Technology Department of Guizhou Province (grant no. LH 2017).
Supplemental Material
Supplemental material for this article is available online.
