Abstract
Cryptomeria japonica biomass was obtained monthly from forest lumbering operations at São Miguel, Terceira and Pico (Azores), between May 2018 and June 2020. C japonica stands with different ages (2 to ±50 years), grown in different soil types (lithosol and andosol) and at different altitudes (200 to >800 m) were used. The chemical composition of 118 C japonica essential oil (EO) samples, plus 1 sample from an EO solid deposit, were analyzed by gas chromatography and gas chromatography–mass spectrometry. Chemical composition cluster analysis showed 2 clusters, cluster I and II, with no chemical correlation. Cluster I grouped all 117 EO samples that resulted from extraction of branches and foliage with, or without, strobili, from strobili, and from the solid deposit sample. Cluster II grouped the 2 samples of EO isolated from wood, with a markedly different composition. Cluster I showed 5 subclusters (Ia-Ie). The degree of chemical correlation between the samples in these subclusters varied between moderate (subclusters Id and Ie), high (subclusters Ib and Ic), and very high (subcluster Ia). Of the 119 samples, 94 (79% of the total) were included in subcluster Ia, with 66 samples from São Miguel, 2 from Terceira, and 26 from Pico, showing a yield range from 0.1% to 0.4% (v/w). All these samples, with a high chemical correlation, were obtained by steam distillation from branches and foliage with, or without, female or male strobili, or both. This subcluster did not show any grouping pattern regarding the collection month, the altitude (200 to >800 m), the soil type (lithosol, andosol, or its subtypes), or the wood age (±30 to ±50 years). Subcluster Ia was characterized by the dominance of α-pinene (13%-43%), sabinene (5%-25%), phyllocladene (2%-22%), limonene (2%-16%), kaurene (traces-13%), elemol (1%-11%), and terpinen-4-ol (1%-7%). The determination of the concentration limits of the EOs representative components confirmed the homogeneity of C japonica EO, obtained from plant material collected in the 3 Azores islands, despite the variability in the range of some diterpenes that deserves further study. This study allowed for the determination of the concentration limits of the EOs representative components, with the purpose of adding value to C japonica EO, obtained from forest lumbering.
Introduction
The evergreen pyramidal, narrow, and dense canopy of Cryptomeria japonica (Thunb. ex L.f.) D.Don (Cupressaceae, formerly in Taxodiaceae), reshaped the Azores islands landscape. The name of the genus derives from the Greek Kryptos (hidden) and meroe (parts). 1 It is commonly known as Japanese cedar and in the Azores as criptoméria, clica or clipa, among other common names. 2
The Japanese cedar was first reported in Japan, under the common name sugi, where it has a long history of being used as an ornamental, beautifying gardens, and boulevards near temples and tombs. Due to its high value, plus-tree breeding programs and natural stands it has been exploited for timber. 3
C japonica was introduced in the middle of the nineteenth century in the Azores, together with many other exotic trees, to embellish parks and gardens.2,4,5 Later, mainly due to the good adaptation to the Azorean conditions, fast growth, rooting rate, resistance to winds, and ease to grow in nurseries it was also used in hedges, shelters, and in the afforestation of wasteland. 5 C japonica became also a highly valued tree, cultivated in a monoculture system, due to the aromatic all-purpose wood used in construction. 1 Despite being a foreign element, C japonica shows some well adapted features to the Azorean landscape, such as contributing to preserve some biodiversity, being low acidophile and evergreen. 6
Official inventories show that about 25% of the total Azores area is occupied with forest, of which about 33% is of production, that is, of cultivated stands, mainly from C japonica (98%), but also from Acacia melanoxylon, Pinus pinaster, and Eucalyptus globulus. 6 C japonica lumbering generates large amounts of forest leftovers, such as branches, foliage, cones (strobili), and wood, which are left neglected and represent an environmental problem. The Direção Regional dos Recursos Florestais current management policies, 7 view a sustainable use of the forest, and include, among others, finding added value to the biomass resulting from forest lumbering. 8 Production of C japonica essential oil (EO) along with other extracts, would contribute to the valorization of this biomass, since this EO has shown some biological properties, namely whitening, antioxidant, antimicrobial, antimelanogenesis and acaricidal activities, among others.9-12
Integrated in a wider work of assessing the potential from C japonica grown in the Azores, 13 this work aimed at characterizing the composition and variability of the EO obtained monthly, for 2 years, from forest lumbering operations at São Miguel, Terceira and Pico (Azores).
Materials and Methods
C japonica Analyzed Samples
Between May 2018 and June 2020, C japonica biomass was collected monthly from forest lumbering operations at 3 of the Azorean islands, São Miguel, Terceira, and Pico. The number of samples, island of origin, sample types, and isolation procedures are detailed in Figure 1. Biomass was collected in C japonica woods with different ages (2 to ±50 years), grown in different soil types (lithosol and andosol subtypes) and at different altitudes (200 to >800 m). Plant biomass was heterogeneous composed mainly by branches and foliage, with or without female and/or male strobili (cones), tree buds or just wood (Figure 1). A voucher specimen has been deposited in the Herbarium of the Departamento de Ciências Agrárias da Universidade dos Açores under number 6401.

EOs samples isolated from Cryptomeria japonica between May 2018 and June 2020, island of plant origin (São Miguel, Terceira and Pico), type of plant material and isolation procedure. The total number of samples includes the 118 EO samples + 1 sample of solid deposit which was also analyzed.
The full data per sample, on sample type, island of collection, island location with global positioning system data and altitude of collection, wood age, soil type, month and year of collection, the extraction procedure, distillation time, EO yield, and sample code, are available on monthly, yearly, and final reports accessible in section Relatórios e Resultados of AZORINA. 8
EO Isolation
The EOs were obtained either by hydrodistillation (HD) or mainly by steam-distillation (SD; Figure 1). HD was performed in lab at Terceira and run for 2 h, using a Clevenger type apparatus according to the European Pharmacopoeia. 14 SD s were performed in Marques Ambiente and in Destillazores® at Pico, in stainless-steel distillers (1100 L, Vieirinox®,), during 2 h in average, at 0.4 bar.
Of the 121 isolated samples of C japonica EO, just 119 (118 EO samples plus 1 sample from an OE solid deposit) were further analyzed. Due to the low amount of EO, 2 samples from September 2018 were not analyzed.
EO Composition Analysis
EO were analyzed by gas chromatography–mass spectrometry (GC–MS) for component identification, and by GC for component quantification. Additionally, all the EO samples were also analyzed by carbon-13 nuclear magnetic resonance (13C NMR) as a tool for unequivocal identification of individual constituents of C japonica EOs.
Gas Chromatography
Gas chromatographic (GC) analyzes were performed using a Perkin Elmer Clarus 400 gas chromatograph equipped with 2 flame ionization detectors, a data handling system and a vaporizing injector port into which 2 columns of different polarities were installed: a DB-1 fused-silica column (polydimethylsiloxane, 30 m × 0.25 mm i.d., film thickness 0.25 μm; J & W Scientific Inc.) and a DB-17HT fused-silica column ([50% phenyl]-methylpolysiloxane, 30 m × 0.25 mm i.d., film thickness 0.15 μm; J & W Scientific Inc.). Oven temperature was programed, 45 °C to 175 °C, at 3 °C/min, subsequently at 15 °C/min up to 300 °C, and then held isothermal for 10 min; injector and detector temperatures, 280 °C and 300 °C, respectively; carrier gas, hydrogen, adjusted to a linear velocity of 30 cm/s. The samples were injected using split sampling technique, ratio 1:50. The volume of injection was 0.1 μL of a n to pentane-EO solution (1:1). The percentage composition of the volatiles was computed, by the normalization method from the GC peak areas, calculated as mean values of 2 injections, from each sample, without using the response factors, in accordance with ISO 7609. 15
Gas Chromatography–Mass Spectrometry
The GC–MS unit consisted on a Perkin Elmer Clarus 600 gas chromatograph, equipped with DB-1 fused-silica column (30 m × 0.25 mm i.d., film thickness 0.25 μm; J & W Scientific, Inc.), and interfaced with a Perkin Elmer 600 T mass spectrometer (software version 5.4.2.1617, Perkin Elmer). Injector and oven temperatures were as above; transfer line temperature, 280 °C; ion source temperature, 220 °C; carrier gas, helium, adjusted to a linear velocity of 30 cm/s; split ratio, 1:40; ionization energy, 70 eV; scan range, 40 to 300 u; scan time, 1 s. The identity of the components was assigned by comparison of their retention indices, calculated in accordance with ISO 7609, 15 relative to C9−C23 n-alkane indices and GC-MS spectra from a lab-made library, created with reference EO, laboratory to synthesized components, laboratory isolated compounds and commercially available standards.
Carbon-13 Nuclear Magnetic Resonance
3C NMR spectra were recorded, as previously detailed, 10 on a Bruker Avance 400 spectrometer operating at 100.61 MHz, equipped with a 5 mm QNP probe at room temperature. The EOs (65-70 mg) was dissolved in deuterochloroform (0.5 mL) and all chemical shift values given in ppm (δ) are referenced to that of tetramethylsilane added as an internal standard and taken as 0.0 ppm. 13C NMR spectra were recorded with the following parameters: spectral width 23.98 kHz under low-power proton decoupling and 13C pulse width of 9.0 μs, 2.0 s relaxation delay (D1) between scans, acquisition time 1.366 s, line broadening 1.0 Hz, for 64k data points. An APT experiment from the Bruker library was used with 4k accumulated scans and the plot limits were +219 to −19 ppm. Chemical shifts and peak assignment of 13C NMR spectra were made according to the literature data,16,17 and with our database containing, α-pinene, sabinene, limonene, terpinen-4-ol, elemol, α-, β-eudesmol, (+)-phyllocladene, and ent-kaur-16-ene compounds spectra, among others.
Statistical Analysis
The percentage composition of the EO components was used to determine the relationship between the different samples by cluster analysis using numerical taxonomy multivariate analysis System (PC software, version 2.2, Exeter Software). 18 For cluster analysis, correlation coefficient was selected as a measure of similarity among all samples and the unweighted pair group method with arithmetical averages was used for cluster definition. The degree of correlation was evaluated according to Pestana and Gageiro 19 as very high (0.90-1.0), high (0.70-0.89), moderate (0.40-0.69), low (0.20-0.39), and very low (<0.20).
The identification and selection of the representative and characteristic components of C japonica EO was calculated in accordance with ISO 11024 to 1, 20 defining the confidence interval at 95%, and using the equation: m1 ±1.96σ1, where m1 is the mean of the concentrations and σ1 is the standard deviation for each component.
Results and Discussion
EO Composition and Cluster Analysis
Of the 119 EOs samples isolated from C japonica, between May 2018 and June 2020, 73 samples resulted from plant material collected in São Miguel, 13 from Terceira and 32 from Pico (Figure 1). The isolated plant material was mainly composed by branches and foliage with female and/or male strobili (62 samples), just branches and foliage (52 samples), just strobili (2 samples), or just wood (2 samples). In 105 cases the EOs were obtained by steam distillation and in 13 cases, by HD (Figure 1). With storage at −4 °C, 13 samples developed a whitish solid deposit, 1 of which was analyzed for its chemical composition determination.
Excluding the wood samples, whose EO yield was <0.05% (v/w), the yield range of the remaining samples was 0.1% to 0.4%.
All 119 C japonica EO samples were fully chemically characterized. The relative amounts of all the identified components are listed in Table 1, following their elution order on the DB-1 column, and arranged according to the minimum and maximum percentage range for each component in the 2 groups defined by agglomerative cluster analysis.
Minimum and Maximum Percentage Range of Cryptomeria japonica Essential Oil Components.

Note: For samples grouped by each of the clusters and subclusters (see Figure 2).
Identification based on mass spectra only. t, traces (<0.05%).
Abbreviations: RI, retention index relative to C9-C23 n-alkanes on the DB-1 column; UI, unidentified compounds.
The determination of correlation between samples using cluster analysis, showed 2 chemically uncorrelated clusters, cluster I and II (Scor < 0; Figure 2). While cluster I (with blue border in Figure 2) grouped all 117 EO samples that resulted from extraction of branches and foliage with, or without, strobili, from strobili, and from the solid deposit, cluster II (with brown border in Figure 2) grouped the 2 samples of EO isolated from wood, with a markedly different composition. Cluster I showed 5 subclusters (Ia-Ie). The degree of chemical correlation between the samples in these subclusters varied between moderate (subclusters Id and Ie, Scor > 0.4), high (subclusters Ib and Ic, Scor > 0.8) and very high (subcluster Ia, Scor > 0.9). Figure 3 shows representative GC profiles, of C japonica EOs, from each cluster and subclusters.

Dendrogram obtained by cluster analysis of the percentage composition of the EO isolated from Cryptomeria japonica, based on correlation and using the unweighted pair-group method with arithmetic average (UPGMA). In samples codes, first 2 numbers represent collection year, 2 following numbers represent collection month, fifth number represent the altitude.

Gas chromatography profiles, taken on the DB-1 column, of representative Cryptomeria japonica EO from each cluster and subclusters. In samples codes, first 2 numbers represent collection year, 2 following numbers represent collection month, fifth number represent the altitude.
Of the 119 samples, 94 (79% of the total) were included in subcluster Ia, with 66 samples from São Miguel, 2 from Terceira and 26 from Pico. All these samples, with a high chemical correlation, were obtained by steam distillation from branches and foliage with, or without, female or male strobili, or both. In this subcluster, the EO yield ranged between 0.1% and 0.3% (v/w). There was no clustering of the samples by month or year, since there was a homogeneous distribution of the samples along the 2 years. This subcluster did not also show any grouping pattern regarding the altitude of the collection site (200 to >800 m), the soil type (lithosol, andosol, or its subtypes), or the wood age (±30 to ±50 years). This subcluster Ia was characterized by the dominance (at least once ≥10%) of α-pinene (13%-43%), sabinene (5%-25%), phyllocladene (2%-22%), limonene (2%-16%), kaurene (traces-13%), and elemol (1%-11%). Terpinen-4-ol (1%-7%) that in other subclusters attained percentage s ≥ 10%, ranged, in this case, between 1% and 7%.
Subcluster Ib grouped 8 samples (7% of the total) with a high correlation (Scor ≥ 0.8); Figure 2). This group included 3 samples from São Miguel, 1 from Terceira and 4 from Pico. Five of these samples were isolated by steam distillation from branches and foliage with, or without, strobili. The remaining 3 EOs samples were isolated by HD, in 1 case from branches and foliage, and in the other 2, from strobili (Table 1). The EO yield ranged, in this case, between 0.1% and 0.2%. These samples were isolated from plant material collected in 6 months of the year, without evidencing a specific month. The altitude of plant collection ranged from 200 to 500 m, with andosol soil type, with its subtypes, and the wood age ranged from ±30 to ±50 years. The samples of this subcluster were characterized by the dominance of α-pinene (27%-49%), sabinene (1%-11%), phyllocladene (3%-10%), limonene (1%-17%), elemol (2%-11%), kaurene (traces-9%), and terpinen-4-ol (1%-3%).
Like subcluster Ib, the subcluster Ic included samples with a high correlation (Scor ≥ 0.8; Figure 2), but less related to the samples from previous subclusters. The 10 samples of this clusters (8% in total) included 5 samples from São Miguel, among which the sample analyzed of solid deposit, 3 samples from Terceira and 2 from Pico. Six samples were isolated by steam distillation from branches and foliage with, or without, strobili. The 3 samples from Terceira were obtained by HD from similar material. This subcluster showed a yield range of 0.1% to 0.4%. As in the former subcluster, these samples were obtained in over 6 months, with no particular grouping. The collection altitude ranged from 400 to >700 m, in both soil types. Except for 1 sample from Terceira, in which the age of the trees was 2 to 3 years (18114L3_T3), the age of the remaining trees ranged from ±30 a±50 years. In this group the EOs main components were phyllocladene (17%-56%), followed by α-pinene (8%-20%), sabinene (5%-14%), elemol (3%-11%), terpinen-4-ol (1%-10%), kaurene (0.1%-10%), and limonene (2%-8%).
Subcluster Id, with just 2 samples from Terceira (2% of the total of samples), showed a moderate correlation with previous clusters. Both EO samples were isolated by HD from branches and foliage, but in 1 case from a 2-year old tree and, in the other case, from a 40-year old tree. The EO yield range was from 0.1% to 0.3%. Both samples were collected in the same month, at a similar altitude, 400 m, but in 1 case from andosol soil type and the other a lithosol. These samples were dominated by elemol (8%-19%), α-pinene (6%-14%), terpinen-4-ol (9%-13%), phyllocladene (4%-13%), sabinene (6%-8%), kaurene (7%), and limonene (6%).
The subcluster Ie included 3 samples (3% of the total) that although with a good correlation within the group, showed a moderate correlation with the samples of previous subclusters. All the EOs was obtained by HD from branches and foliage from material collected in Terceira. Two of the samples were isolated from 2- to 3-year-old trees, and the other sample from a 40-year old tree. The EO yield ranged between not determined to 0.4%. The samples were collected in different months and the altitude of collection ranged between 400 and 500 m, in 1 case with andosol soil type and the other 2, lithosol. Kaurene (21%-31%) was the main component of the EOs in this case, followed by α-pinene (4%-14%), sabinene (1%-10%), terpinen-4-ol (5%-13%), elemol (7%-10%), limonene (3%-4%), and phyllocladene (0.1%-5%).
The cluster II did not show chemical correlation with the cluster I (Scor < 0; Figures 2 and 3), and included the 2 EOs isolated by HD from wood, in the same month. The EO yield ranged from <0.05% to 0.4%. The main identified compounds of these OEs were γ-cadinene (13%-21%), germacrene D (7%-11%), and α-muurolol ( = δ-cadinol) and α-eudesmol (7%-9%). This data confirms the importance of sample type, namely the amount of branches versus wood, on the EO chemical composition.
C japonica EO samples showed no grouping according to the month, the year, the altitude of the collection site, or the soil type. The ratio between the EOs main compounds seemed to be mainly determined by the type of plant material (branches and foliage with more, or less, strobili vs wood), trees age (very young trees showing higher terpinen-4-ol content), and by the isolation procedure (HD or steam distillation).
EO Representative and Characteristic Components
C japonica EO representative and characteristic components, as well as their concentration limits, were determined in accordance with ISO 11024 to 1, 20 using only the 105 EO samples obtained by steam distillation, from plant material collected in the 3 Azores islands, over the 2-year period. Of the 76 components present in all EO samples obtained by SD, 20 were selected. By applying data analysis statistical methods, their minimum and maximum range were determined (Table 2). Except for α-pinene and kaurene, that showed from 7 to 8 values, in the 105 samples, which were outside this confidence interval, and were considered as outliers, the remaining 18 compounds range fitted within the confidence interval in over 100 of the 105 samples. This confirms the homogeneity of the EO composition obtained by steam distillation from plant material (branches and foliage with more, or less, strobili) collected in the 3 Azores islands.
Minimum and Maximum Percentage Range of Components Selected Among the Representative and Characteristic Components of Cryptomeria japonica Essential Oil.

Identification based on mass spectra only. ; t, traces (<0.05%).
Abbreviations: CL, confidence interval; UI, unidentified compounds.
EO Composition Comparison With Literature Data
GC, GC–MS, and 13C NMR analysis of C japonica EOs allowed to confirm the complexity of these EOs dominated by monoterpene hydrocarbons, but also with considerable amounts of oxygen-containing sesquiterpenes and diterpene hydrocarbons. To the best of our knowledge only 2 studies have previously addressed the EO composition of C japonica grown in the Azores (Table 3). The comparison of the present data, with recent studies using similar plant material (foliage with, or without, strobili -cones-, or just cones), collected either in Azores, in Corsica where C japonica was also introduced, and in Japan, showed, in general, a similar profile and components range, despite different time and distillation procedures (Table 3).
Range of Main Components ( ≥ 10%) Identified in the Essential Oils Isolated From Cryptomeria japonica Collected in the Azores, China, Corsica, and Japan.
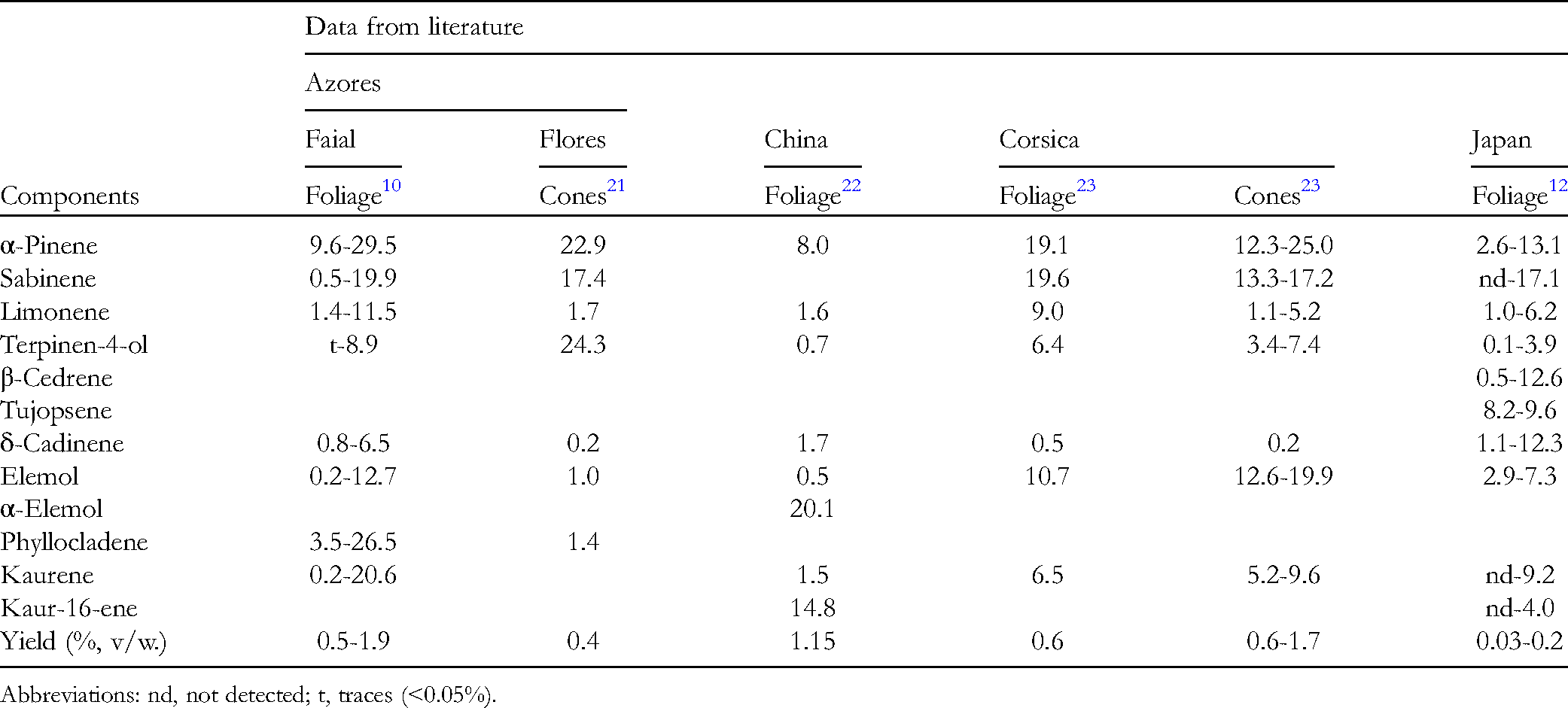
Abbreviations: nd, not detected; t, traces (<0.05%).
Interestingly, 1 of the main differences between studies was in the presence and relative amounts of some diterpenes (Table 3). The presence of diterpenes in EOs and other types of extracts of C japonica has been the subject of several studies. Appleton et al, 24 based on leaf light petroleum extracts, suggested the alternative occurrence of kaurene or phyllocladene in 2 varieties of C japonica. Later Yasue et al,3,25 proposed the existence of 8 chemically distinct types based on the relative amounts and proportions of sclarene, kaurene and phyllocladene. In the present study, all these diterpene hydrocarbons could be found, although in different ratios. Whether this reflects the existence or different chemotypes, or the existence of different C japonica varieties, requires further chemical and morphological studies with individual trees.
Footnotes
Acknowledgments
Thanks are due to Fundação para a Ciência e a Tecnologia through national funds, under CESAM UIDB/50017/2020 + UIDP/50017/2020, CQB UID/MULTI/0062/2019, CQE UIDB/00100/2020, FEDER, and PT2020 PACompete 2020. Project SAI-AZOR/2018/392 co-financed by Operational Program - PO Açores 2020 - FEDER and Regional Government of Azores - AÇORES-03-0853-FEDER-000027
Author Contributions
ACF, CM, MCSMR, and AJRMA conceived and designed the research, conducted the experiments and wrote the manuscript. AJRMA and MCSMR supervised the work. All authors read and approved the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Governo Regional dos Açores (grant number Project SAI-AZOR/2018/392, AÇORES-03-0853-FEDER-0, CESAM UIDB/50017/2020 + UIDP/50017/2020, CQB UID/MULTI/0062/2019, CQE UIDB/00100/2020).
