Abstract
Tea tree hydrosol (TTH) is the aqueous phase of the distillate from the steam distillation process of the twigs and leaves of Melaleuca alternifolia. In this study, the chemical components of TTH were analyzed by gas chromatography–mass spectrometry (GC–MS) and gas chromatography–flame ionization detector (GC-FID). The major components were terpenoids (1.53 mg/mL), including monoterpenoids and sesquiterpenoids. The antibacterial activity of TTH was assessed by paper disc agar diffusion and broth dilution methods. Terpinen-4-ol (MIC: 0.31-0.62 mg/mL), 2-endo-hydroxy-1,8-cineole (MIC: 1.25-2.50 mg/mL) and p-menth-3-ene-1,2-diol (MIC: 0.62-2.50 mg/mL) were the main antibacterial components of TTH. The results showed that the major components of TTH and tea tree oil (TTO) had the same parent structures, but different degrees of oxidation. Terpinen-4-ol was the most abundant component in both TTH (624.2 μg/mL) and TTO (35-48%), and played a major role in antibacterial activities. This is the first report on the chemical components and antibacterial activities of TTH and their correlation with TTO, which demonstrates the potential application of TTH as a natural antimicrobial agent in health care products.
Keywords
Introduction
Melaleuca alternifolia (Maiden & Betche) Cheel, the plant used for the production of tea tree oil (TTO), belongs to the Myrtaceae family. It is native to Australia and can be found in coastal areas. It also distributed in Southeast Asia and the southern part of China, but most of the industrialized plantations are in southern Australia.1,2 TTO is obtained from the twigs and leaves of M alternifolia by steam distillation.3,4 The aqueous phase produced alongside TTO is called tea tree hydrosol. (TTH).5,6 TTH is claimed as being useful for treating acne, eczema, psoriasis, skin redness and swellings, sinus congestion, scratch wounds, dandruff, and skin fungal and bacterial infections. It is an excellent complement to TTO because it is gentle to be used directly on the skin and is more suitable for use for children and pets. In recent years, there have been more TTH-containing products coming to the market.
Although many functions of TTH have been reported, there are still no reports on its chemical composition and antibacterial activities, and no reports on the qualitative and quantitative analysis of the related products. In this work, we report the chemical components of TTH by gas chromatography–mass spectrometry (GC–MS) qualitative and gas chromatography–flame ionization detector (GC–FID) quantitative analyses and its antibacterial evaluation against 12 strains of bacteria. After we obtained all the analytical results, the chemical component features of TTH and their correlation with TTO were studied to help us understand the similarities and differences between TTH and TTO in their properties and application.
Result and Discussion
Chemical Composition of TTH
GC-MS was used to identify 13 components of TTH by retention time, molecular and fragment ions, and comparison with the National Institute of Standards and Technology (NIST) mass spectrometry database, literature data, and reference materials (Table 1). Most of the components were monoterpenoids with molecular weight <200. In our previous research,
7
TTH enriched by resins D101 and AB-8 and eluted with 50% and 95% ethanol was fractionated into 4 parts (M1-50, M1-95, M2-50, and M2-95); 16 compounds were isolated. The established quantitative method by GC–FID was applied to determine the contents of the 9 main compounds (Supplemental Table S1), including monocyclic monoterpenes and sesquiterpenes. Monocyclic monoterpenes containing one hydroxyl group presented the highest content (
Identification of the Compounds in TTH by GC-MS.

“A” = NIST; “B” = literature reference; “C” = reference compound.
Determined Retention Index (RId).
Literature Retention Index (RIl).
Abbreviations: TTH, tea tree hydrosol; GC-MS, gas chromatography-mass spectrometry.
Antibacterial Activity of TTH
Four TTH fractions (M1-50, M1-95, M2-50, and M2-95) and TTO were diluted with 10% Tween 80 − water to obtain 3 (high, medium, and low) concentrations of solution. The high concentration samples of all parts had stronger antibacterial activities than medium and low concentration samples (Table 2). The inhibition zones obtained by M2-50 and M2-95 were very small, which means that the inhibitory activities of these 2 fractions were very weak. For most strains, the inhibitory activities of M1-50 and TTO were basically the same, while the activity of M1-95 was higher than that of TTO. Of the 12 strains, Bacillus subtilis, Candida albicans, and Micrococcus luteus were very sensitive, Escherichia coli, Salmonella paratyphi B, Staphylococcus aureus, S saprophyticus, and Klebsiella pneumonia were sensitive, but Streptococcus pyogenes, S epidermidis, Shigella flexneri, and Pseudomonas aeruginosa were less sensitive. Therefore, we selected the first 8 strains for the investigation of the antibacterial activity of the compounds in TTH.
Diameter of Zone Inhibition (mm) of Four TTH Fractions and TTO.
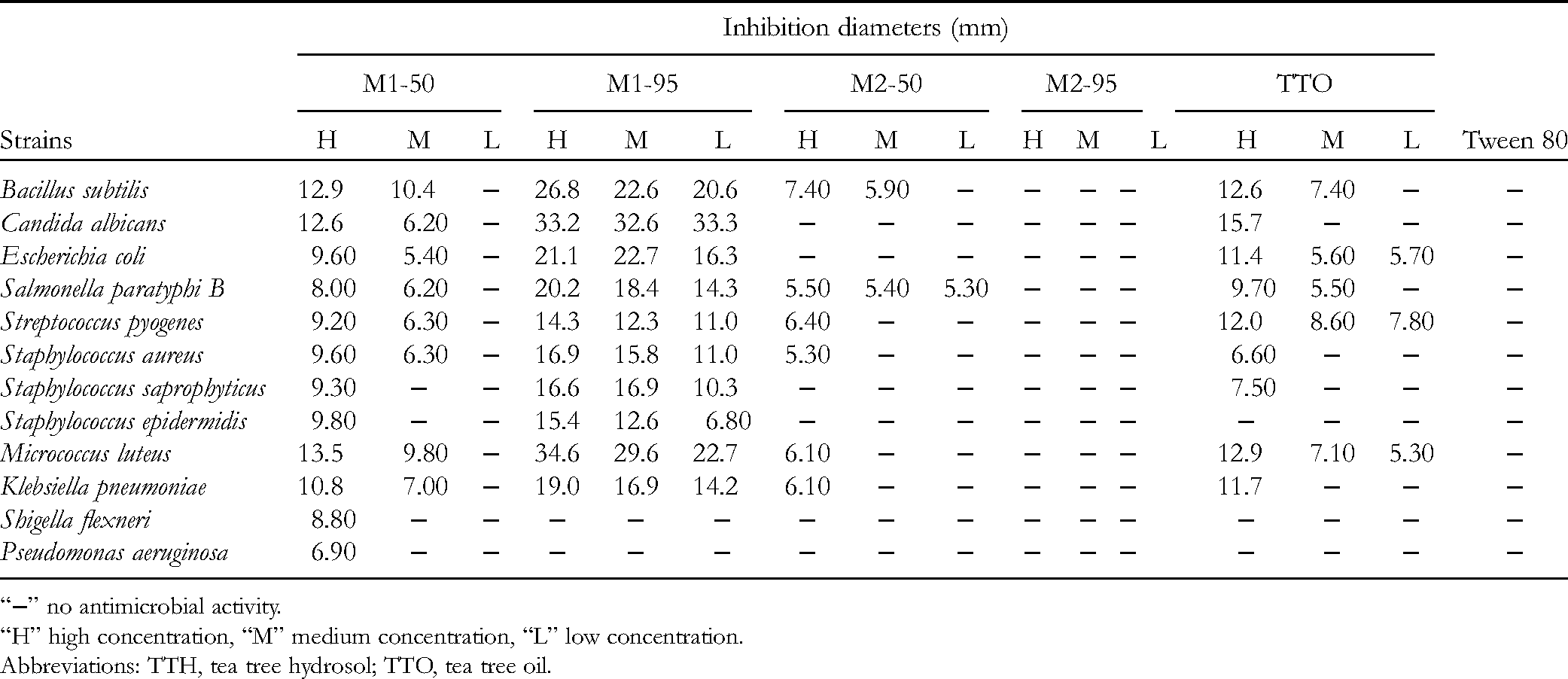
“−” no antimicrobial activity.
“H” high concentration, “M” medium concentration, “L” low concentration.
Abbreviations: TTH, tea tree hydrosol; TTO, tea tree oil.
The broth dilution results (Table 3) showed that compounds
Minimal Inhibitory Concentration of Compounds From tea tree hydrosol.

“−” no antimicrobial activity.
Correlation Between TTH and TTO
Comparison of Components in TTH and TTO by GC and GC/MS
Both TTH and TTO come from the distillate produced by the steam distillation of the twigs and leaves of M alternifolia; TTH is the aqueous phase and TTO is the oil phase. Therefore, there should be certain connections between the chemical components of TTH and TTO. The results of GC and GC/MS analyses showed that the components of TTO appeared almost within 15 min, while most of the components of TTH appeared after that time (Figure 1). The main common compounds between them were 1,8-cineole (tR = 4.85 min), terpinen-4-ol (tR = 7.16 min) and α-terpineol (tR = 7.35 min), with terpinen-4-ol being the most abundant component in both (Supplemental Table S2). The quantitative analysis of the main common components of TTH and TTO showed that the contents of 1,8-cineole (

The GC chromatogram of TTH and TTO.
Comparison of Antibacterial Components of TTH and TTO
Both TTO and terpinen-4-ol have antibacterial activities and terpinen-4-ol, as the major component of TTO, was considered to be the major contributor to the antimicrobial activities of TTO.
8
The results in this paper showed that TTH enrichments also had significant antimicrobial activities. Among them, the activity of M1-95 was stronger than that of TTO at the same concentration, and it had a significant inhibitory effect on S epidermidis. Although the activity of M1-50 was similar to that of TTO, it showed certain inhibitory effects on S flexneri, P aeruginosa, and S epidermidis, whereas, in contrast, TTO did not show inhibitory activity on these 3 strains. It should be pointed out that terpinen-4-ol (
Correlation Between the Structure of Components in TTH and TTO
Through comprehensive qualitative analysis of TTH and TTO using GC and GC/MS and examination of the chemical structures of the isolated compounds from TTH, as well as the literature reports, we found that the major chemical components of TTH and TTO had the same parent structure, namely the p-menthane skeleton of moncyclic monoterpenoids (Supplemental Figure S1A) and the aromadendrane skeleton of tricyclic sesquiterpenoids (Supplemental Figure S1B). The structural difference between them lies in the type, position, and number of functional groups in the parent structures. Nonpolar and weakly polar terpenoids, such as alkanes, alkenes, dienes, monohydric alcohols and ketones, were present in TTO, while dihydric and trihydric alcohols with higher polarity were found in the aqueous phase. Terpinen-4-ol, with a monohydroxy group, and 1,8-cineole, with a 1,8-epoxy group, possessed moderate polarity and are present in both oil and aqueous phases, with the majority in the oil phase. In other words, with the increase in the degree of oxidation of the parent structure, the polarity of terpenoids gradually increases and their solubility in water also gradually increases. Therefore, terpenoids with higher oxidation degrees were concentrated in the aqueous phase of the steam distillation, which formed the chemical component features of TTH. As to whether these terpenoids with higher oxidation degree are secondary metabolites of the plant or produced by steam distillation, there are no relevant research reports so far.
Materials and Method
Materials and Regents
TTH, produced by Main Camp Natural Extracts Pty Ltd in Australia, was provided by Tianjin Sino-Australia Casino Biotechnology Company. MCI Gel CHP20/P20 was provided by Mitsubishi Chemical Corporation. 1,4-Cineole (
Preparation of TTH and TTO Samples
The analytical samples of TTH were prepared using solid phase extraction (SPE). The SPE column (12.8 × 60 mm, LISUI SCIENCE Co., Ltd) containing 3 g of MCI gel was conditioned with 100.0 mL deionized water. A total of 200.0 mL of TTH was then loaded onto the SPE column at a flow rate of 10.0 mL/min and eluted with 8.0 mL ethyl acetate to a 10 mL volumetric flask. Subsequently, 0.1 g anhydrous sodium sulfate was added to the eluent solution and shaken well. After standing for 10 min, it was used for GC–MS/GC-FID analysis.
TTO was accurately weighed and dissolved in ethyl acetate to obtain a solution of 50 mg/mL for GC–MS analysis.
Qualitative Conditions
The TTH samples were analyzed using TRACE-DSQ GC–MS from Thermo Fisher Scientific, with a HP-5MS column. High-purity helium was used as the carrier gas at a flow rate of 1 mL/min. The sample injection volume was 1 µL, with a split ratio of 10:1. The column temperatures were as follows: first ascending from 60 °C to 120 °C at 10 °C/min, then raised to 180 °C at a rate of 5 °C/min, and finally to 250 °C at a rate of 10 °C/min. The injector temperature was 200 °C and the detector temperature was 300 °C. For MS, the samples were analyzed in electron impact (EI) mode (70 eV) in the mass range of m/z 33 to 350. Retention indices (RIs) were determined relative to the retention times of a series of n-alkanes (C9 -C17).
For the qualitative analysis of TTO, the injection volume was 0.2 μL, with a split ratio of 30:1. The column temperatures were as follows: first ascending from 60 °C to 120 °C at 10 °C/min, then raised to 140 °C at a rate of 2 °C/min, and finally to 250 °C at a rate of 10 °C/min. The other conditions were the same as for TTH.
Quantitative Conditions
All samples were analyzed with an Agilent 7890B gas chromatograph equipped with an Agilent G4567A auto injector, an FID, hydrogen as combustion gas, and an Agilent DB-624 column (30 m × 0.45 mm × 2.55 μm film thickness). Nitrogen was used as the carrier gas at a flow rate of 1 mL/min. The injection volume was 1 μL with a split ratio of 10:1. The column temperatures were as follows: initial temperature of 60 °C raised to 120 °C at a rate of 10 °C/min, and to 150 °C at a rate of 2 °C/min, then to 165 °C at a rate of 1 °C/min, next to 200 °C at a rate of 2 °C/min, and finally to 250 °C at a rate of 10 °C/min (total time = 58.5 min). The injection temperature was 200 °C and the detection temperature 300 °C. The quantitative conditions of TTO were the same as for TTH.
Antibacterial Bioassay of Isolated Fractions and Compounds
The antibacterial activity of TTH fractions and TTO against 12 strains of bacteria was tested using a paper disc agar diffusion method; 8 strains were used to screen the activity of isolated compounds. The fractions were dissolved and diluted with 10% Tween 80−water and prepared into high, medium, and low concentration solutions. The freshly grown bacterial suspension (with a concentration of 107 colony forming units [CFU]/mL) was spread on the culture medium. The sensitive papers (diameter: 6 mm) were immersed in the test solutions for 2 h and then removed with forceps. After freeze drying, they were attached to the medium and placed in an incubator maintained at 37 °C. Finally, the diameter of the zone of inhibition (ZOI) was determined using a vernier caliper. 9
Cefalexin and norfloxacin were used as positive controls for the antibacterial bioassay of the compounds isolated from TTH in vitro by the broth dilution method. The compounds were dissolved in 20% DMSO−water to prepare a solution with an initial concentration of 20 mg/mL, and then diluted into 10, 5, 2.5, 1.25, 0.625, 0.312 and 0.156 mg/mL test solutions in turn in a 96-well plate for the determination of minimum inhibitory concentrations (MICs). The 20% DMSO−water solution was used as a blank control. The bacterial suspension was then added to the 96-well plate at a concentration of 106 CFU/mL. Finally, the optical density (OD) at 600 nm was measured and the MIC calculated. 10 Each sample was tested 3 times in parallel.
Conclusions
In summary, the chemical components and antibacterial activities of TTH have been studied. The main compounds in TTH were terpenoids, of which terpinen-4-ol, trans-p-menthane-1,4-diol, cis-p-menthane-1,8-diol, and 2-endo-hydroxy-1,4-cineole were the main components by GC analysis. Antimicrobial assay showed that both TTH and TTO have obvious antimicrobial activities, and terpinen-4-ol, 2-endo-hydroxy-1,8-cineole and p-menth-3-ene-1,2-diol were the main antimicrobial components of TTH. As TTH and TTO are from the same steam distillation process, their major components have the same parental structure, but different degrees of oxidation. This work gives support for the wide applications of TTH as a natural antimicrobial agent.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X211038390 - Supplemental material for Chemical Components and Antimicrobial Activities of Tea Tree Hydrosol and Their Correlation With Tea Tree Oil
Supplemental material, sj-docx-1-npx-10.1177_1934578X211038390 for Chemical Components and Antimicrobial Activities of Tea Tree Hydrosol and Their Correlation With Tea Tree Oil by Xiaoxue Li, Dan Shen, Qiushuang Zang, Yan Qiu and Xuedong Yang in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
