Abstract
A series of calycanthaceous alkaloid analogs have been synthesized in excellent yields. All the target compounds were evaluated in vitro for biological activity against a broad range of plant pathogen fungi, bacteria and human pathogenic fungi, and some of the designed compounds exhibited potential activity in the primary assays. Notably, Compound
Introduction
The use of synthetic pesticides in agriculture worldwide is still the most widespread method for the control of plant diseases. However, the extensive application of agrochemicals over the years has led to the development of resistance in pest populations and environmental problems. Consequently, the discovery of new agrochemicals with high efficacy and selectivity against target species directly or indirectly from natural product has recently been crucial in the research and development of agrochemicals.
The hexahydropyrroloindole skeletons are very important moieties that are widespread in a large family of natural products with wide range of attractive bioactivities. Calycanthaceous alkaloids1-4 (Figure 1), which contain hexahydropyrroloindole skeletons, are an important class of alkaloid that can be isolated from roots, leaves, flowers, and fruits of chimonanthus praecox. 5 The Calycanthaceae plants have been used as traditional Chinese medicines for the treatment of fungal, 6 hypertension, tumor, and inflammatory.7,8
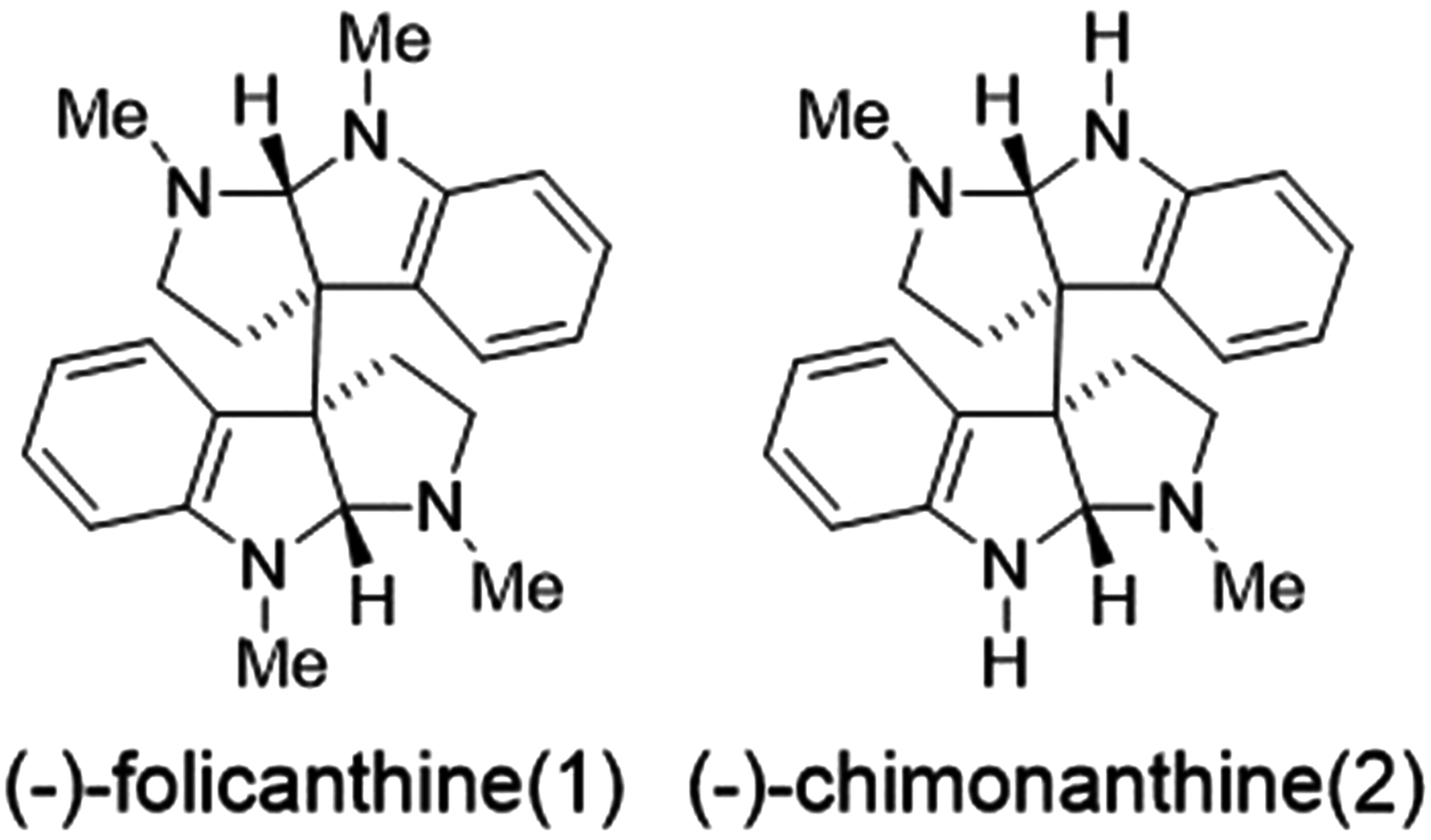
Structures of calycanthaceous alkaloids.
Our group has recently reported the preparation and potent antimicrobial activity of calycanthaceous alkaloid derivatives.9-15 These findings inspired us to further modify the structure of calycanthaceous alkaloids with functional motifs so as to acquire potential agrochemical leads for plant disease control.
As part of ongoing efforts to discover new natural-product-based antifungal agent, 2 novel series of N-substituted calycanthaceous alkaloid derivatives were designed and synthesized, and their structures were identified on the basis of satisfactory analytical and spectral (1H NMR, 13C NMR, and ESI-MS) data.
To the best of our knowledge, the antimicrobial activities of the synthetic derivatives are reported herein for the first time.
Design and Synthesis of Calycanthaceous Alkaloids Analogs
The synthetic route to the target compounds is shown in Figure 2. A total of 36 calycanthaceous analogs were prepared from indole-3-acetonitrile via acylation at the N-position according to a previously reported procedure in our group and the spectral data were characterized by 1H-NMR, 13C-NMR spectroscopy, and ESI-MS.16-25

Synthetic route to the title compounds a1 to a18 and b1 to b18.
Antimicrobial Acitivity
The results of the biological testing against a wide range of plant pathogen fungi, Gram-negative bacteria, Gram-positive bacteria, and human pathogenic fungi are listed in Table 1. The minimal inhibitory concentration (MIC) were evaluated with Carbendazim, Amphotericin B, Chlorothalonil, Gentamicin, Streptomycin, Penicillin, and Fluconazole as positive controls, to evaluate the activity of the synthesized calycanthaceous alkaloid analogs against Verticillium dahliae, Fusarium oxysperium sp vasinfectum, Cytospora juglandis, Aspergillu sflavu, Penicillium citrinum, Fusarium oxysporum, Colletotrichum orbiculare, Aspergillus niger, Bcinerea pers, Curvularia lunaia, Escherichia sp, Pseudomonas aeruginosa, Ralstonia solanacearum, Bacillus cereus, Staphylococcus aureus, Candida krusei, Crytococcus neofonmans, and C tropicalis.
MIC of Compounds Against Plant Pathogenic Fungi, Gram-Negative Bacteria, Gram-Positive Bacteria, and Human Pathogenic Fungi.

Note: The Carbendazim and chlorothalonil were used as the positive controls; “─” means no inhibition effect.
Abbreviations: MIC, minimal inhibitory concentration; V.d, Verticillium dahliae; F.v, Fusarium oxysperium sp. vasinfectum; C.j, Cytospora juglandis; A.s, Aspergillu sflavu; P.c, Penicillium citrinum; F.o, F oxysporum; C.o, Colletotrichum orbiculare; A.n, A niger; B.p, Bcinerea pers; C.l, Curvularia lunaia; E.s,Escherichia sp; P.a, Pseudomonas aeruginosa; R.s, Ralstonia solanacearum; B.c, Bacillus cereus; S.a, Staphylococcus aureus; C.k, C krolimus; C.n, Crytococcus neofonmans; C.t, Candida tropicalis; Ca, Carbendazim; A, amphotericin B; Ch, chlorothalonil; G, gentamicin; S, streptomycin; P, penicillin; F, fluconazole.
It is manifested that compounds
Materials and Methods
Instruments and Chemicals
All reagents and solvents were reagent grade or purified according to standard methods before use. Analytical thin-layer chromatography was performed with silica gel plates using silica gel 60 GF254 (Qingdao Haiyang Chemical Co., Ltd). Melting points were measured on an Electrothermal digital apparatus and were uncorrected. The 1H - NMR (500 MHz), and 13C-NMR (125 MHz) were obtained on an AM-500 FT-NMR spectrometer (Bruker Corporation) with CDCl3 as the solvent and tetramethylsilane as the internal standard. Mass spectrum (MS) were recorded under electrospray ionization (ESI) conditions using a LCQ Fleet instrument (Thermo Fisher). Yields were not optimized. The title compounds were synthesized under a nitrogen atmosphere.
Synthesis
Intermediates 1 to 3 Were Synthesized According to Our Previously Reported Procedure
12
Synthesis of Compounds a1 to a18 and b1 to b18
Compound
Biological Activity
The antimicrobial activity of calycanthaceous alkaloids analogs was measured according to the previously reported method.26,27
The tested compounds dissolved in 5% dimethyl sulfoxide, to a concentration of 1.02 mg/mL, 100 μL of the solutions were added to the first well and serially diluted from first well by taking 100 μL into second. This 2-fold dilution was continued down the plate and 100 μL from the eighth column of the plated discarded. The ninth column of the plate was reserved for negative control wells (without inocula) and the tenth column, for the positive growth control wells (without antibacterial agent). The antibacterial concentrations were 256, 128, 64, 32, 16, 8, 4, and 2 µg mL, respectively. The antibacterial test plates were incubated aerobically at 37 °C for 24 h, the antifungal test plates were incubated aerobically at 28 °C for 48 h. The MICs were examined. All tests were performed in triplicate and repeated if the results differed.
Conclusions
A total of 36 novel tetrahydropyrroloindole-based calycanthaceous alkaloid analogs were prepared using indole-3-acetonitrile as the starting material via acylation at the N-position and the activity against a wide range of plant pathogen fungi were screened. Notably, compound
Supplemental Material
sj-docx-1-npx-10.1177_1934578X211032611 - Supplemental material for Synthesis and Antimicrobial Activity of Calycanthaceous Derivatives
Supplemental material, sj-docx-1-npx-10.1177_1934578X211032611 for Synthesis and Antimicrobial Activity of Calycanthaceous Derivatives by Rui Zhu, Cheng Yang, Ke Han, Yongdong Gu, Jiwen Zhang, Shaojun Zheng and Hongjin Bai in Natural Product Communications
Footnotes
Author Contributions
SZ, HB and JZ contributed to designed research; CY KH and YG contributed to performed research; YC and RZ contributed to performed statistical analysis; SZ wrote the paper; and HB and JZ reviewed the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (21502073 and 31360079), the Natural Science Foundation of Jiangsu Province (Grants No BK 20150465 and BK20180978), the Key Research and Development Program (Modern Agriculture) of Zhenjiang City (NY2018002), Zhoushan Public Welfare Science and Technology Project (2019C31066), and Xinjiang Production Construction Corps Key Laboratory of Protection and Utilization of Biological Resources in Tarim Basin (BRYB1703).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
