Abstract
Endophytic fungi, especially those found in medicinal plants, are widely studied as producers of secondary metabolites of biotechnological interest. In this study, on the basis of an activity-directed isolation method and spectroscopic analysis, two active polyketides, citrinin (
Keywords
Polyketides are distributed widely in organisms and constitute a large family of natural products with various biological activities. These possess great commercial value. 1 Fungi are one of the main sources for polyketides with a wealth of chemical diversity. 2 Endophytic fungi, microorganisms that reside in the tissues of living plants without causing any apparent symptoms of disease, especially those found in medicinal plants, are an outstanding resource for searching for biologically active products. 3-12 In this study, we isolated and identified the active polyketide natural products from endophytic Penicillium citrinum DBR-9, which was isolated from the healthy root tubers of the traditional Chinese medicinal plant Stephania kwangsiensis Lo. In previous studies, S. kwangsiensis was found to be a potential source of antimicrobial active endophytic fungi, and the extract of the endophytic fungus P. citrinum DBR-9 showed excellent antifungal activity. 13-15 Besides, the species of Penicillium had been reported to be an important source of polyketides. 2 In this paper, the purpose was to discover excellent biocontrol agents as alternatives to synthetic chemical fungicides for the control of plant pathogenic fungi in agriculture.
Using bioassay-guided fractionation, two active polyketide compounds, citrinin (
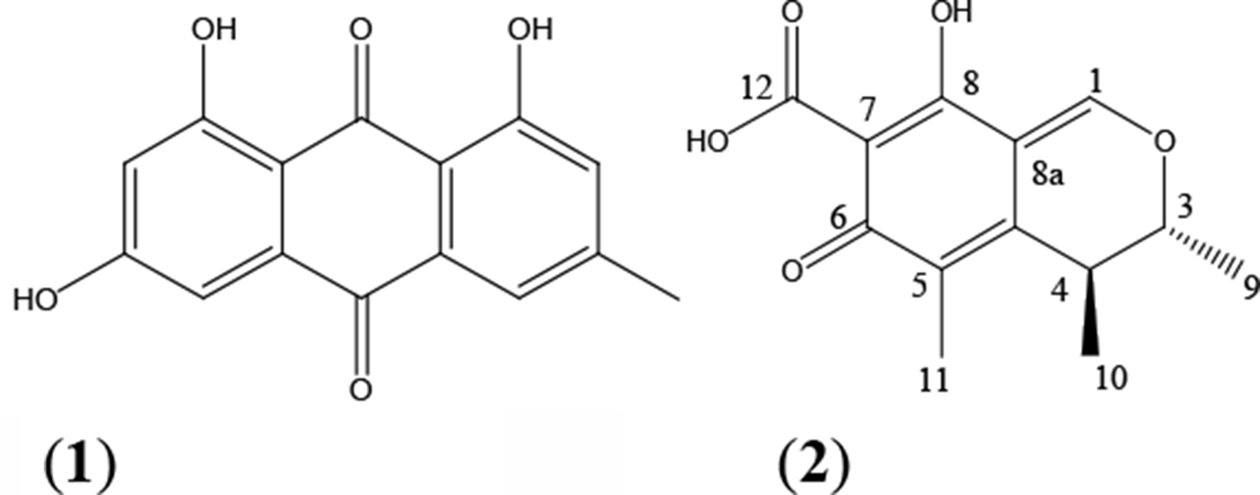
Structures of citrinin (1) and emodin (2).
The Antifungal Activity of the Two Polyketides to Plant Pathogenic Fungi.

aThe values are expressed as means±standard deviations (n = 3). Carbendazim was used as the positive control, referred to the data in our previous study. 14
b“—” means that the IC50 values are not calculated, because the antifungal activities of the compounds toward these pathogenic fungi were too low in preliminary experiment.
Based on clear observations in preliminary experiments, Phytophthora parasitica var. nicotianae was chosen for further study of the mechanism of effects of emodin (

The effects of emodin on the mycelial morphology of P. parasitica var. nicotianae (40×) ((a) control group, added the mixture of acetone and water (1:1 by volume) instead of the emodin solution; (b) emodin-treated group, treated with 100 µg/mL emodin).
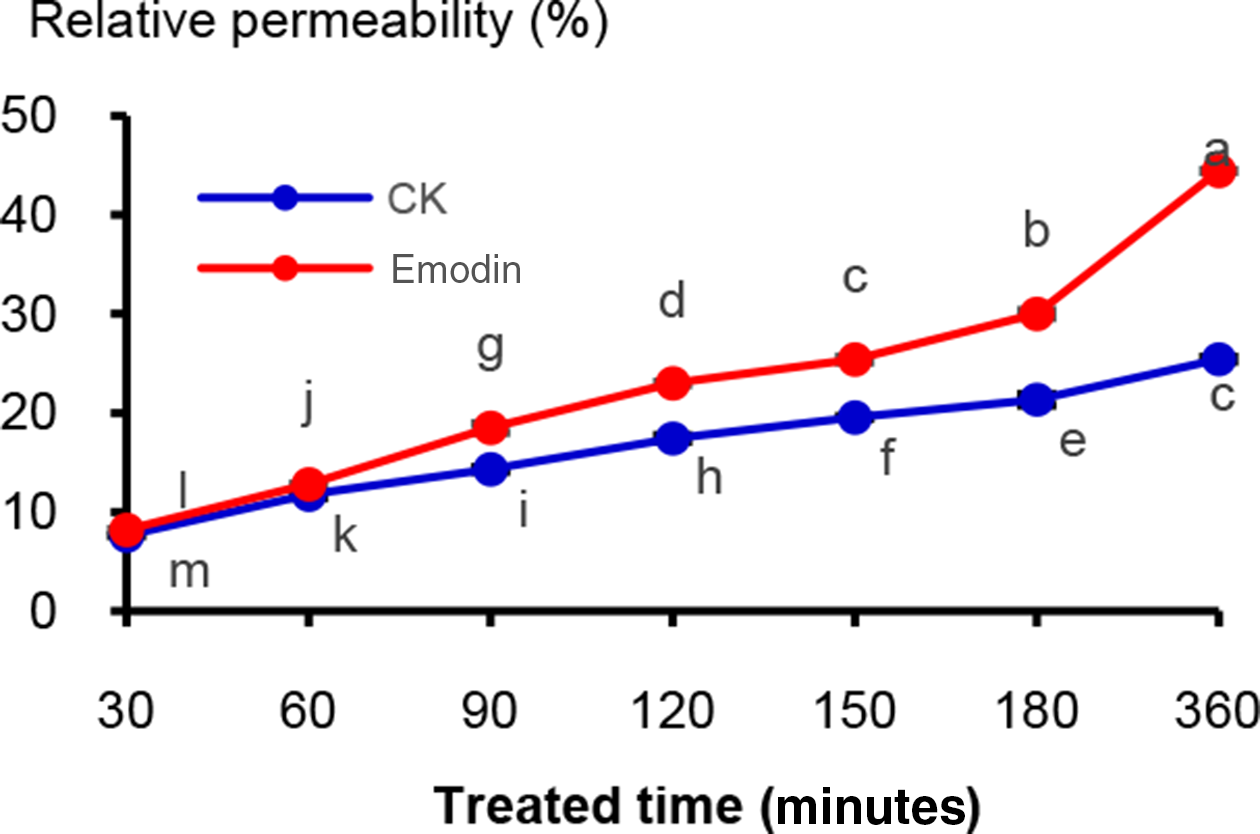
The effects of emodin on the membrane permeability of P. parasitica var. nicotianae. (The concentration of emodin was 100 µg/mL. “CK” is the control group, the mixture of acetone and water was added (1:1 by volume) instead of the emodin solution. Different letters, i.e., a, b, c, …, indicate significant differences among the treatments at P = 0.05 level.)
The effects of emodin on the protein concentration of P. parasitica var. nicotianae. (The concentration of emodin was 100 µg/mL. “CK” is the control group, without emodin, and the mixture of acetone and water was added (1:1 by volume) instead of the emodin solution. Different letters, i.e., a, b, c, …, indicate significant differences among the treatments at P = 0.05 level.)

The changes of relative membrane permeability of P. parasitica var. nicotianae (Figure 3) were positively correlated with the treatment time. The relative permeability of the emodin-treated group had a sharp increase and ranged from 8.21% to 44.44% within 30 to 360 minutes, significantly higher than the control group (without emodin) (7.63%-25.44%) with an increase by 7.60%-74.69%. The cell membrane plays a primary role in protecting the cellular components from the extracellular environment and encountering antimicrobial agents. 20,21 The leakage of cellular components could indicate damage of the fungal membrane. 18 Most of the antifungal peptides also primarily target the fungal cell wall and membrane. 20,22–24 The sharp increase of the relative permeability in the emodin-treated group indicated that emodin could induce the destruction of cytoplasmic membranes and cause efflux of the intercellular content, which could lead to the death of the fungi.
The effects of emodin on the concentration of soluble protein in the mycelia of P. parasitica var. nicotianae are shown in Figure 4. The concentration of soluble protein of the emodin-treated group was significantly lower than the control group. In addition, as the treatment time increased, the concentration of soluble protein in the mycelia decreased gradually. After treatment for 48 hours, the concentration of mycelial soluble protein was about 0.50 mg/mL, decreased by 20.63% compared with the control group (without emodin) (0.63 mg/mL). The results indicated that emodin could influence protein synthesis of the fungal cell and then influence the main components of cell membranes.
The above results demonstrated that the antifungal effects of emodin occurred by causing changes to the cell membrane and structure, based on the results of the morphological aberrations including swelling, distortion, visible loss of pigmentation, the sharp increase of the relative membrane permeability, and decrease of the total cellular protein content. These results indicate that the phytopathogenic fungi are susceptible to emodin under laboratorial conditions. Perhaps further tests using plants infected with the fungi could give further clarifications of emodin’s potential use in agriculture.
Although citrinin (
Emodin (
The findings of our study provide a new opportunity to know the antifungal activity of citrinin (
Experimental
Endophytic Fungus and Their Fermentation Culture
The endophytic fungus DBR-9 identified as Penicillium citrinum by morphological and molecular taxonomical parameters was isolated from the healthy root tubers of the Chinese medicinal plant S. kwangsiensis, which was collected from Guangxi Institute of Botany, Guilin, Guangxi, China, in February 2014. The plant was identified according to its morphological features by Prof. Shuiyuan Jiang, a botanist from Guangxi Institute of Botany. The voucher specimen (No. 201400012) of this plant was deposited in the Herbarium of the College of Life Science, Guangxi Normal University (GNU). For the fermentation culture, three plugs of agar medium (0.4 × 0.4 cm) were inoculated, respectively, in each 1000 mL Erlenmeyer flask containing 400 mL potato dextrose broth (PDB) medium, supplemented with 200 g/L of potato and 20 g/L of dextrose. The flasks were incubated in the condition of stilling culture for 30 days at 28°C.
Crude Extract Preparation
Four hundred milliliters of absolute ethyl acetate (EtOAc) was poured into the flask (contained 400 mL fermented liquid) and kept aside for 3 days. Then the fermented broth was first filtrated through two layers of gauze, and further filtered under vacuum to obtain the filtrate. The filtrate was evaporated to a concentrated liquor. The concentrated liquor was, respectively, extracted three times with an equal volume of EtOAc and n-butanol, and the three times filtrates were combined. The remaining concentrated liquor and the combined filtrates were, respectively, evaporated to dryness to obtain the residue of concentrated liquor, and the extracts of EtOAc and n-butanol of the fermented liquid, respectively.
Isolation and Identification of the Activity Compounds of Endophytic Fungus DBR-9
In this paper, the active compounds of the EtOAc extract of endophytic P. citrinum DBR-9, which was isolated from the root tubers of S. kwangsiensis, were isolated using an activity-guided isolation method and chromatography. The EtOAc extract (14.0 g) was dissolved in acetone and mixed with silica gel (20 g), dried, and then loaded onto a silica gel column; elution was performed with a linear gradient of petroleum ether/EtOAc (7:3→4:6→0:10), to EtOAc/methanol (7:3→4:6). Five fractions (F1-F5) of 2000 mL were collected. The antifungal activities of these fractions were evaluated with a growth rate method.
14
The bioactive fractions F1 (1.8 g) were combined and separated further by silica gel column chromatography, and elution was performed with a linear gradient of petroleum ether/EtOAc (95:5→90:10→85:15→80:20→75:25→70:30), and six subfractions (SF1.1-SF1.6) of 400 mL were collected. According to the determination of antifungal activities, SF1.1 (0.29 g) and SF1.2 (0.1 g) were the bioactive subfractions. After evaporation of solvent in SF1.2, a yellow needle crystal emerged. By recrystallization, a pure compound was obtained and numbered as compound
Determination of the Inhibitory Activity of the Two Compounds Against Mycelial Growth of Plant Pathogenic Fungi
The inhibition of samples on mycelial growth of plant pathogenic fungi was determined according to the previous report. 14 The required concentration of samples dissolved in acetone:water (1:1 by volume) was evenly mixed with molten potato dextrose agar (PDA) medium and poured into a Petri dish (sample solution:PDA medium = 1:9, v/v, mL). The control was added to the solvent of acetone:water (1:1 by volume) instead of samples solution. Each treatment had three replicates. For determination of inhibitory activities, three discs of agar medium (0.4 × 0.4 cm) with fungal cultures of pathogens were inoculated on PDA medium containing sample with different concentration, and incubated at 28°C for 72 hours. And the diameters of the colony were then measured at decussation and the inhibitory rates were calculated. Finally, the regression equations, IC50, and 95% confidence intervals were calculated by the least-squares method. Ten economically significant plant pathogenic fungi Bipolaris maydis, P. parasitica var. nicotianae, Exserohilum turcicum, Colletotrichum capsici, Alternaria oleracea, Diaporthe citri, Ceratocystis paradoxa, Pestalotiopsis theae, A. citri, and Cochliobolus miyabeanus were used for bioassay. Among them, D. citri and A. citri were provided by Guangxi Academy of Specialty Crop, and the other eight were provided by the Laboratory of Phytopathology, the College of Agriculture, Guangxi University.
Observation of the Morphology Changes of P. parasitica Var. nicotianae
The tested pathogen cultured on the PDA medium containing emodin sample with required concentration. Cultures without emodin were used as control. After incubated at 28°C for 72 hours, morphological alterations in hyphae were observed by macro and microscopic methods. For the microscopic observation, mycelia samples were viewed by using a DM3000 microscope (Leica Microsystems Instrument Incorporated Company, Germany) with a 40× objective and photographed with attached digital-coupled device camera and capture software (Leica Application Suite).
Determination of the Cell Membrane Permeability
The determination of the cell membrane permeability of P. parasitica var. nicotianae was performed according to previous reports with slight modifications. 34 After incubated in a rotary shaker at 120 rpm and 28°C for 7 days, the hyphae were collected by filtering with double gauze, and washed 3 times with double distilled water. One thousand milligrams of hyphae was resuspended in 10 mL antimicrobials (100 µg/mL emodin) and the initial electrical conductivity (EC0) of each sample was measured. Then samples were incubated at 28°C with agitation of 120 rpm for 30, 60, 90, 120, 180 and 360 minutes. The supernatants were collected and centrifuged (4000 rpm for 5 minutes) and the electrical conductivities (ECn) were measured by each collected point. After 360 minutes, samples were then boiled for 15 minutes, cooled to room temperature, and final electrical conductivity (ECf) was measured. Cultures without emodin were used as controls. Each treatment had three replicates. The relative permeability of cell membranes was calculated by the following equation

Determination of the Soluble Protein Content of Mycelia
Soluble protein content in the mycelia of P. parasitica var. nicotianae was performed according to the previous report with some modifications. 35 A standard curve was established according to the method described by Jiao. 35 The protein standard curve equation was obtained as follows, where x was the absorption value OD, and y was the protein concentration.

For the tested pathogenic fungus, 5 mycelial plugs taken from 3-day-old colonies of the strain were inoculated in a 250 mL flask containing 100 mL PDB. The flasks were shaken at 120 rpm and 28°C for 5 days. Partial flasks were treated with emodin at the concentration of 100 µg/mL. Flasks without emodin were prepared as a control. Then flasks were shaken for additional 4, 8, 16, 24, and 48 hours, respectively, and then mycelia were collected by filtering with double gauze and washed three times with double distilled water. Each sample (0.5 g mycelia) was ground to a paste in an ice bath and then centrifuged at 10 000 rpm for 15 minutes before supernatants were collected. Into the test tubes, 0.1 mL centrifugal supernatant was added (control group was added 0.1 mL acetone:water [1:1 by volume]), then 0.9 mL distilled water and 5 mL coomassie bright blue solution were added and mixed uniformly, and quantified with the standard curve. Each treatment had three replicates. OD values were measured at 595 nm.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was co-funded by grants from the Natural Science Foundation of Guangxi, China (Project Nos. 2016GXNSFAA380207; 2016GXNSFBA380049; 2018GXNSFAA281013), Key Laboratory of Ecology of Rare and Endangered Species and Environmental Protection (Guangxi Normal University) (ERESEP2018Z04), Ministry of Education, Guangxi Normal University, Guangxi Key Laboratory of Rare and Endangered Animal Ecology, Guangxi Normal University (Project Nos. GKN. 17-A-02-02), the Ecology Doctoral Program Foundation (EDPC 2018002), and Youth and Middle-aged Basic Ability Promotion Project (2017KY0080), Guangxi Normal University, Guangxi, China.
