Abstract
Curcumae Radix is an efficacious ingredient with various medicinal properties empirically used in traditional Chinese medicine (TCM) formula for the treatment of cancer, depression, chest pain, dysmenorrhea, epilepsy, and jaundice. However, either phytochemical or pharmacological information of Curcumae Radix underlying its traditionally medicinal uses is rarely summarized and systematically analyzed. To provide evidence for clinical trials, a comprehensive literature review has been prepared of the phytochemicals, and ethnopharmacological and pharmacological mechanisms of this herb. The review approach consisted of searching several web-based scientific databases, including PubMed, Web of Science, and Elsevier. The keywords included “Curcumae Radix,” “Curcuma wenyujin,” “Curcuma longa,” “Curcuma kwangsiensis,” and “Curcuma phaeocaulis.” Based on the proposed criteria, 57 articles were evaluated in detail. The accumulated data indicate that Curcumae Radix contains a number of bioactive phytochemicals, mainly sesquiterpenes, diarylheptanoids, and diarylpentanoids, which account for a variety of medicinal values, such as anticancer, anti-inflammation, anti-hepatic fibrosis, and antioxidant. A wide range of apoptotic proteins, cell adhesion molecules, inflammatory cytokines, and enzymic and nonenzymic antioxidants could be modulated by either Curcumae Radix or its bioactive compounds, thus underpinning a fundamental understanding for the pharmacological effects of this herb. This review highlights the therapeutic potential of Curcumae Radix to progress the development of versatile adjuvants or therapeutic agents in the future.
Curcumae Radix, named Yu Jin (in Chinese), is the dry root tuber of Curcuma wenyujin Y. H. Chen et C. Ling, C. longa L., C. kwangsiensis S. G. Lee et C. F. Liang and C. phaeocaulis Val., family Zingiberaceae. 1 These plants are widely distributed in the southern provinces of China, including Jiangsu, Zhejiang, Fujian, Guangdong, Guangxi, Sichuan and Yunnan. As shown in Figure 1, Curcumae Radix has an oblong, ovate or spindle shape with slightly pointed ends and a full central section. Its length and central diameter are about 1.5 ~ 7 cm and 1 ~ 2.5 cm, respectively. The herb smells slightly fragrant, but tastes spicy and bitter. The dry rhizomes, derived from the above-mentioned species, except Curcuma longa, also produce an herbal medicine, Curcumae Rhizoma (named as Ezhu in Chinese), recorded in the China Pharmacopoeia. On the basis of Traditional Chinese Medicine (TCM) theory, Curcumae Radix and Curcumae Rhizoma behave respective priorities in drug nature and traditional uses. Curcumae Radix, cold-natured and functioning through liver, heart and lung passages, is mainly used to treat mental or gastrointestinal symptoms caused by dampness-heat of liver and gallbladder. Curcumae Rhizoma, warm-natured and functioning through liver and spleen passages, is good at treating the syndrome of food retention and accumulation of extravasated blood. 1,2 Therefore, they need be specially authenticated during research and application.

Curcumae Radix.
According to TCM theory, the medicinal functions of Curcumae Radix include blood and qi circulation promotion, pain release, melancholy dispelling, dysphoria alleviation, and jaundice elimination. Traditionally, Curcumae Radix is commonly used as an efficacious ingredient in the TCM formula for the treatment of chest pain, dysmenorrhea, epilepsy, and jaundice. 1 However, the knowledge of either phytochemistry or pharmacology of Curcumae Radix underlying its traditionally medicinal uses is rarely summarized and systematically analyzed. Meanwhile, an increasing number of studies have been reported recently regarding the active metabolites and pharmacological activities of Curcumae Radix. To support further phytopharmacological research, drug development and clinical use of this herbal medicine, the present work herein aims to perform a systematic literature review on the traditional uses, phytochemistry, and pharmacological aspects of Curcumae Radix. The review will underpin the fundamental understanding and inspire Curcumae Radix-based drug development.
The work searched a number of electronic databases, including PubMed, Web of Science, and Elsevier, up to t December 21, 2020. The keywords included Curcumae Radix, Curcuma wenyujin, Curcuma longa, Curcuma kwangsiensis, and Curcuma phaeocaulis. Searching was limited to articles only in English. The irrelevant papers (especially studies on Curcumae Rhizoma or other parts of Curcumae plants), reviews, patents, abstracts, case reports, and abstracts in symposiums and congresses were excluded. The articles were reviewed by 2 authors independently to determine compatibility with the inclusion criteria above, and 57 articles were eligible to be evaluated in this work.
Ethnopharmacology
The TCM Yu Jin (Curcumae Radix) was first recorded in «Yao Xing Lun» by Quan Zhen in about 600 AD. According to «Compendium of Materia Medica», the most authoritative and comprehensive TCM encyclopedias, Curcumae Radix is able to disperse blood stasis, regenerate tissue and arrest bleeding, alleviate sores and skin ulcers, and dispel melancholy. 3 To activate blood circulation, promote tissue regeneration and arrest bleeding, patients are typically instructed by TCM doctors to take 7‐31 g powder of Curcumae Radix 3 times per day either alone or combined with other herbs, such as radices rehmanniae, cattail pollen or bezoar. 4 The powder of Curcumae Radix mixed with water could also be directly applied on the surface to alleviate sores and skin ulcers to relieve the local pain and swelling. For the treatment of anxious or depressed psychoses, the medicinal pills are made of Curcumae Radix and alum with a mass ratio of 7:3. The patients then self-administer 50 pills (12, 16 g) each time with water until the symptoms disappear. 3 The application experience of Curcumae Radix has been handed down from generation to generation for thousands of years. Though there is no evidence-based scientific reports, the abstracted information from the ancient TCM records has highlighted the potentials of Curcumae Radix as an effective medicinal candidate for certain diseases, thus providing a valuable natural resource for further scientific study.
In the current medicine system of China, Curcumae Radix is not only used by TCM doctors for treating depression, hematemesis, hematuria, gastrorrhagia, and hemorrhoids, but also applied in the clinical formula for treating symptoms associated with cancer, angina pectoris, hepatitis, cholelithiasis, jaundice, and ileac passion. Clearly, the bioactive secondary metabolites of Curcumae Radix are the basic functional units for these clinical applications.
Phytochemistry
The bioactive metabolites of Curcumae Radix are responsible for the exhibited medicinal activities. Sixty compounds, mainly sesquiterpenes, diarylheptanoids and diarylpentanoids, but also including alkaloids, polysaccharides, and some other types, have been directly isolated and elucidated from Curcumae Radix.
Sesquiterpenes
Sesquiterpenes, with 15 carbon atoms and 3 isoprene units, are widely distributed in plants, insects, microorganisms, and other natural products. In plants, they are major components of volatile oils existed in the form of alcohols, ketones and lactones. From the roots of Curcuma herbal plants, 39 sesquiterpenes have been reported; their structures are shown in Figure 2. Among them, curcumenone, curcumenol, zederone, neocurdione, curdione, curzerenone, β-elemene, curcumol, and germacrone were reported from the roots of Curcuma wenyujin, C. longa, C. kwangsiensis, and C. phaeocaulis. 5,6 Curcumolide, kwangsiensis A, kwangsiensis B, curcujinone A, curcujinone B, 4-epi-curcumenol, curcumadiol, gweicurculactone, (1R,4R,5S,7S)-curwenyujinone, zedoalactone E, zedoalactone H, zedoarolide B, and curdionolide A were identified from the root tuber of C. wenyujin, 7 -11 and turmeronol A, bisacurone, bisacurone A-C, dehydrozingerone, zingerone, 2, 5-dihydroxybisabola-3, 10-diene, and 4, 5-dihydroxybisabola-2,10-diene from the root tuber of C. longa. 12 Moreover, β-caryophyllene, ar-curcumene, zingiberene, β-bisabolene, β-sesquiplellendrene, ar-turmerone, α-turmerone, and β-turmerone were found in the tuberous root of C. longa. 13
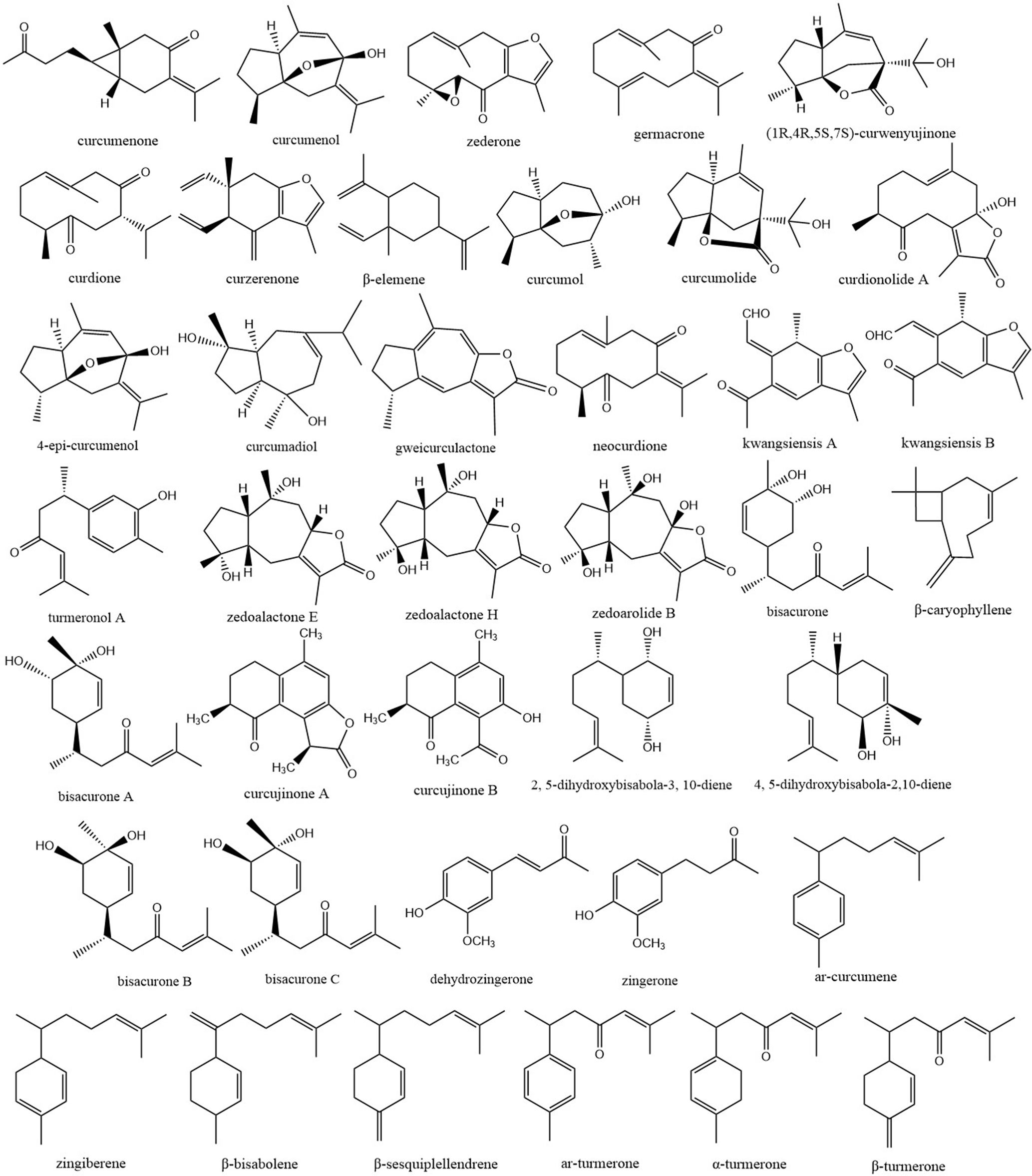
Sesquiterpene compounds of Curcumae Radix.
Diarylheptanoids and Diarylpentanoids
Diarylheptanoids are common and characteristic constituents of the genus Curcuma. To date, 9 diarylheptanoids or diarylpentanoids have been isolated from Curcumae Radix and their structures are shown in Figure 3. From the root tuber of C. longa, 6 diarylheptanoids, curcumin, dihydrocurcumin, demethoxycurcumin, bisdemethoxycurcumin, 1,5-epoxy-3-carbonyl-1,7-bis(4-hydroxylphenyl)−4,6-heptadiene, and 1,7-bis(4-hydroxyphenyl)−1,4,6-heptatrien-3-one, and 2 diarylpentanoids, 1,5-bis(4-hydroxyphenyl)-penta-(1E,4E)−1,4-dien-3-one and 1,5-bis(4-hydroxy-3-methoxyphenyl)-penta-(1E,4E)−1,4-dien-3-one, have been identified. 14 Meanwhile, curcumin, demethoxycurcumin, tetrahydrocurcumin and bisdemethoxycurcumin were reported from the root tuber of C. wenyujin. 8,15

Diarylheptanoid and diarylpentanoid compounds of Curcumae Radix.
Others
Besides the above mentioned compounds, 12 others have been reported for Curcumae Radix as well; their structures are shown in Figure 4. Among them, curcumrinols A-C were isolated from the root tuber of Curcuma wenyujin and curcumrinol A presented strong cytotoxicity to 2 leukemia cell lines in vitro. 16 Zhang et al., reported that curcumrinols D and E, from Curcuma wenyujin root, were also cytotoxic against cancer cells. 17 Diterpenoid C, curcuminol F, curcuminol G, wenyujinoside, and aurantiamide were then isolated from the roots of Curcuma wenyujin in the following year. 18 One hoursexane derivative, crotepoxide, and one alkaloid, 2-(2'-methyl-1'-propenyl)−4, 6-dimethyl-7-hydroxyquinoline, were reported from the root tuber of C. wenyujin and C. longa, respectively. 8,12

Other compounds of Curcumae Radix.
Pharmacology
The extracts or active compounds of Curcumae Radix exhibit a wide spectrum of pharmacological activities, mainly including anticancer (Table 1), anti-inflammatory, anti-hepatic fibrosis and antioxidant, as follows.
Anticancer Effects and Pharmacological Mechanisms of Curcumae Radix.

Anticancer Activities
Using N-methyl-N-nitro-N-nitrosoguanidine (MNNG)-induced gastric cancer rats, both tumor incidence and tumor volume were found to be lowered in the groups treated with the steam distilled extract of Curcumae radix. 19 In a following study, this effect was found to be related to the down-regulated expressions of vascular endothelial growth factor (VEGF), cyclooxygenase-2 (COX-2), and proliferating cell nuclear antigen (PCNA) in the gastric mucosa of rats under treatment with Curcumae radix. 20 n-Butyl alcohol extracts of Curcuma wenyujin were found to be able to down-regulate zincfifi nger ebox binding homeobox 1 (ZEB1) transcription, and caudal type homeobox transcription factor (CDX-2) and claudin-2 expression, thus preventing the invasiveness of human gastric cancer (GC) AGS cells cultured with Helicobacter pylori (HP) of positive cytotoxin-associated gene A (CagA) and vacuolating cytotoxin gene A (VacA) expression (CagA+VacA+). 21 Moreover, the 70% (v/v) ethanol extract of Curcumae radix was reported to significantly increase the survival of mouse models with breast cancer metastasis. In the MCF7 cell model, the mechanisms of its anti-metastatic activity were identified to regulate the expression of metastasis markers, including Matrix Metalloproteinase 9 (MMP 9) and the proto-oncogenes AP1 Complex (c-FOS and c-JUN), through suppressing the C-C Chemokine Receptor Type 7 (CCR7) expression. 22
β-Elemene (1-methyl-1-vinyl-2,4-diisopropenyl-cyclohexane), extracted from the root of Curcuma wenyujin, presents a broad-spectrum, moderate, antitumor effect and is widely used as an adjunctive drug to enhance the efficacy, reduce the toxicity of chemoradiotherapy, and reverse drug resistance in cancer treatment. 43 Previous studies have indicated that β‐elemene could inhibit cell proliferation and induce apoptosis in various types of cancers, including melanoma, 44 hepatocellular, 23 lymphoma, 26 glioma, 29 esophageal, 31 gastric, 45 glioblastoma multiforme, 32 nonsmall-cell lung, 33 pancreatic, 39 and nasopharyngeal. 34 Recent studies have reported that β‐elemene significantly inhibited the migration and invasive capacity of BGC823, SGC7901 and multidrug resistant (MDR) SGC7901/ADR gastric cancer cells, TE-1 and KYSE-150 esophageal cancer cells in vitro and inhibited the capacity of BGC823 cells to diffuse peritoneally and metastasize in vivo. 24,25,31 Moreover, the radio sensitivity of cancers, such as glioblastoma multiforme U87-MG, T98G, and U251 cells, 32 melanoma A375 cells, 44 and A549 lung adenocarcinoma xenograft 27,28 were also reported to be significantly enhanced by β-elemene.
The pharmacological studies demonstrated that β-elemene exerted its effects by modulating multiple molecular targets. It modulated Ki67, PCNA, mitogen-activated protein kinase kinase-3 (MKK3), MKK-6, and cyclin proteins to arrest the cell cycle 31,35,38 and regulated the expression of relative proteins such as the long non-coding RNA (lncRNA) highly up-regulated in liver cancer (HULC), survivin, caspase-3,‐8 and −9, PERK, IRE1α, ATF6, Hsp90/Raf-1, Fas/FasL, and Bcl-2/BAX proteins to induce cell apoptotic death of cancer cells. 26,27,29,33,35,37 In addition, β-elemene performed synergistic or directly suppressive effects through targeting histone H1, 23 copper transporter 1 (CTR1), 30 E3 ubiquitin ligase Cbl, 36 signal transducer, and activator of transcription 3 (Stat3), DNA methyltransferase 1 (DNMT1), enhancer of zeste homolog 2 (EZH2), 34 hypoxia inducible factor 1 subunit α (HIF1α), vascular endothelial growth factor A (VEGFA), 39 PTEN, 31 and ataxia telangiectasia mutated (ATM) signaling pathways. 32 Furthermore, β-elemene inhibited tumor angiogenesis and metastasis through regulation of crucial molecules, such as Claudin-1 by downregulating FAK phosphorylation, 24 MMP-2/9 via modulating Cbl-b/EGFR/ERK/AKT signaling 25 and programmed cell death 1-ligand 1 (PD-L1) by regulating p-AKT. 31 β-Elemene was also found to reverse epithelial-mesenchymal transition (EMT) in MDR gastric cancer cells by inhibiting the transcription factors ZEB1 and ZEB2 that contribute to antitumor metastatic activities. 25 Moreover, the radiation-induced HIF1α and peroxiredoxin-1 (Prx-1) expression were significantly suppressed by β-elemene, which reversed the radio-resistance of the human lung cancer. 27,28
Besides β-elemene, curcumol, diterpenoid C and tetrahydrocurcumin, identified from Curcumae radix, were also reported to demonstrate anticancer effects in vitro and in vivo. Curcumol could inhibit the growth of MDA–MB–231 and Hep3B cells in vitro and tumor growth of the xenograft model in vivo. The underlying mechanisms of its anticancer effects were related to triggering p53–independent apoptosis through the p73–PUMA/Bak signaling pathway in breast cancer cells, or inhibiting the activation of programmed cell death-ligand 1 (PD-L1) through crosstalk between inhibited hypoxia-inducible factor-1α (HIF-1α) and phosphorylated signal transducer and activator of transcription 3 (p-STAT3) signaling pathways in hepatic cancer. 40,41 Shen et al. reported that diterpenoid C induced apoptosis and arrested cell cycle through suppressing the phosphorylation of extracellular signal-regulated kinase (ERK), c-Jun N-terminal kinase (JNK), p38 mitogen-activated protein kinase (p38 MAPK), and the cleavage of caspase 3 on human colon adenocarcinoma SW620 cells. 42 Moreover, tetrahydrocurcumin was reported to induce the autophagic death of non-small cell lung carcinoma cells. The expression of autophagy-associated proteins, including p62, phosphorylated (P)-mechanistic target of rapamycin (mTOR), phosphoinositide 3–kinase (PI3K), p–PI3K, protein kinase B (Akt) and p–Akt, was reduced in cancer cells under tetrahydrocurcumin treatment. 15
Anti-inflammatory Activities
In the 7,12‐dimethylbenz(a)anthracene (DMBA)/12‐tetradecanoylphorbol‐13‐acetate (TPA)‐promoted skin of mice models, oral treatment of β‐elemene (50 mg/kg bwt, 21 days) could drastically reduce the expression levels of the inflammatory protein markers, including nuclear factor‐κB (NF‐κB), tumor necrosis factor‐α (TNF‐α), interleukin‐6 (IL‐6), nitric oxide synthase (iNOS), and cyclooxygenase (COX‐2), thus effectively inhibiting the inflammation. 35 The protein expression of proinflammatory cytokines and cell adhesion molecules, including IL-1β, TNF-α, MCP-1, ICAM-1, and INF-γ, could also be remarkably reduced under β-elemene treatment in the arteries of experimental mice. 46 Demethoxycurcumin and bisdemethoxycurcumin, isolated from Curcumae longae Radix, were reported to exhibit anti-inflammatory effects in RAW 264.7 macrophage cells. In addition, carrageenan-induced paw edema in mice models could significantly be controlled by demethoxycurcumin and bisdemethoxycurcumin. Their anti-inflammation activities were attributed to the reduced expression of inflammatory mediators iNOS and COX-2, as initiated by the inhibition of NF‐κB activity. 47 In Helicobacter pylori-induced gastric epithelial GES-1 cell inflammation, Curcumae Radix derived diterpenoid C was found to block the NF-κB signal pathway as well. The mechanisms underlying these effects were claimed to be associated with the inhibited secretion of proinflammatory cytokines IL-8 and IL-6, and the promoted secretion of anti-inflammatory cytokine IL-4. 48 Besides these, curcumolide, a sesquiterpenoid isolated from Curcuma wenyujin, was also reported to exhibit significant anti-inflammatory effects in LPS-induced RAW 264.7 macrophages, the streptozotocin-induced diabetic rat model, and TNF-α stimulated human umbilical vein endothelial cells (HUVECs). 7,49 Using macrophage models, curcumolide could suppress lipopolysaccharides (LPS)-induced NF-κB activation, and reduce TNF-α, IL-6, IL-1β, nitric oxide (NO) and reactive oxygen species (ROS) production in vitro. 49 Meanwhile, it could alleviate retinal vascular permeability and leukostasis and attenuate the overexpression of TNF-α and ICAM-1 in diabetic retinas either in vitro or in vivo. 7 On mouse RAW 264.7 cells, 2 guaiane-type sesquiterpenes, kwangsiensis A and B, isolated from the roots of Curcuma kwangsiensis, were also reported to exhibit moderate anti-inflammatory activities on the basis of their inhibition of LPS-induced NO production with IC50 values of 27.4 and 35.1 µM, respectively. It could be inferred that their guaiane-type structure contributed to the anti-inflammatory effects via suppressing the activation of NF-κB or protein tyrosine phosphorylation. 10
Anti-Hepatic Fibrosis Activities
In hepatic stellate cell-T6 (HSC-T6), Curcuma kwangsiensis root extract was found to improve MMPs-13 expression by inhibiting the tissue inhibitor of metalloproteinase-1 (TIMP-1) expression to accelerate the decomposition of extracellular matrix (ECM). Meanwhile, the expression of aI-collagen was also inhibited to reduce the production of ECM. 50 Further pharmacological studies indicated that the medicated serum of Curcumae Radix extracts could inhibit expression of Tβ-RⅠ mRNA and prevent transmission of the downstream signal to block the action of TGF-β1, thus significantly reducing the development of hepatic fibrosis. 51
Antioxidant Activities
Curcumae Radix has been reported to present obvious protective effects against oxidative damage in experimental models. Extracts of Curcuma kwangsiensis roots, including the 95% ethanol, 75% ethanol, methanol, dichloromethane, and petroleum ether extracts, could protect against H2O2-induced PC12 cell damage in vitro. Treatment with Curcumae Radix extracts could remarkably inhibit lactate dehydrogenase (LDH) leakage and the accumulation of intracellular ROS. Therefore, the activities of superoxide dismutase (SOD), catalase (CAT), and the contents of glutathione (GSH) would be further enhanced. The possible antioxidant mechanisms might be related to the regulation of Ca2+ levels and mitochondrial membrane potential within the oxidative damaged cells. 52 Oral administration of β‐elemene has been reported to restore the levels of lipid peroxidation and antioxidants status in DMBA/TPA‐induced animals. It could enhance the formation of thiobarbituric acid‐reactive substances (TBARS) and increase the levels of nonenzymatic antioxidants GSH, vitamin E and vitamin C, and enzymatic antioxidants SOD, CAT, and glutathione peroxidase [GPx]. 35 In rheumatoid arthritis fibroblast-like synoviocytes, β‐elemene caused an approximately 9-fold increase in intracellular ROS production, consequently leading to the activation of p38 mitogen-activated protein kinase (MAPK). 53 Moreover, β-elemene was also found to inhibit atherosclerotic lesion size and increase stability of plaques in animals by alleviating levels of vascular oxidative stress, which was indicated through the elevated SOD activity and the lowered malondialdehyde (MDA) level in the aorta. The activities of CAT, GPx and GSH were increased under this treatment as well. 46
Other Pharmacological Activities
Many Curcumae Radix-containing TCM formulas are empirically claimed to be used for Alzheimer’s disease (AD) and depression treatment in China. The aqueous extract of Curcuma wenyujin was reported to improve the cognitive function and rescue learning and memory deficits in amyloidal beta (Aβ)-induced AD mice. The possible mechanisms were related to the reduced phosphorylation level of tau proteins in the hippocampus (Thr-181, Thr-231, and Ser-404), which was induced by the elevated phosphorylation level of PI3K, Akt, and GSK-3β under Curcumae Radix treatment. 54
As a herb used for blood circulation promotion in TCM, several studies have provided evidence that Curcumae Radix could improve the hemorheological state in animal models. 55,56 Intragastric administration of Curcuma longa root decoction (0.9 g·kg-1, 2 weeks) was found to significantly reduce the serum levels of Epo, EpoR, and CD62p, thus reducing the formation of physiological thrombosis in experimental rats. 55 Moreover, Chen et al. reported that the same extract of Curcumae Radix could significantly improve the abnormality of 5-HT in plasma, β-EP in brain, and inhibit the expression of c-fos, which led to an analgesic effect in rats. 57
Besides the anti-inflammation effect described above, curcumolide derived from Curcuma wenyujin was also reported to have an anti-angiogenic effect in HUVECs and in a murine oxygen induced retinopathy model of ischemia-induced retinal neovascularization. The mechanisms underlying this effect might be linked to the prohibited retinal neovascularization and direct suppression of the tyrosine kinase activation of VEGFR2 by inhibiting phosphorylation of downstream protein kinases Src, FAK, ERK, AKT, mTOR, and p70S6K, and promoted endothelial cell apoptosis through a caspase-3 dependent pathway. 58
Concluding Remarks
As an important herbal medicine traditionally used for centuries in China, an increasing number of studies of Curcumae Radix have been reported regarding its chemical constituents and pharmacological activities in recent decades. According to the data reviewed in this work, it is quite evident that Curcumae Radix contains many bioactive components, mainly sesquiterpenes, diarylheptanoids, and diarylpentanoids, which account for a variety of medicinal properties, such as anticancer, anti-inflammation, anti-hepatic fibrosis, antioxidant and so on. Curcumae Radix and its bioactive compounds can affect various signaling pathways through regulation of a wide range of apoptotic proteins, cell adhesion molecules, inflammatory cytokines, and enzymic and nonenzymic antioxidants. The development of a versatile adjuvant or even pharmaceutical agents from Curcumae Radix thus could be considered in the treatment of diseases. However, the major shortfalls of this development have been limited to the few studies assessing the treatment effects of Curcumae Radix against diseases related to its main pharmacological activities and drug metabolism in human subjects to confirm its therapeutic outcomes. This should be further complemented with comprehensive well-controlled and double-blind clinical trials to provide meaningful data in determining the efficacy and safety of a given Curcumae Radix treatment. Moreover, although phytochemical and pharmacological studies have received great interest, the identity, pharmacokinetics, bioavailability, physiological and pharmacological pathways of most functionally bioactive constituents in Curcumae Radix remain unknown and need further investigation.
Overall, the ethnopharmacological, phytochemical and pharmacological evidence accumulated herein strongly underpin a fundamental understanding of herbal Curcumae Radix and support its traditional and ongoing clinical applications in China. Further safety evaluation and clinical trials are expected to finally transform the traditional TCM herb Curcumae Radix to a valuable medicinal resource with promising therapeutic and industrial applications.
Supplemental Material
Online supplementary file 1 - Supplemental material for A Comprehensive Mini-Review of Curcumae Radix: Ethnopharmacology, Phytochemistry, and Pharmacology
Supplemental material, Online supplementary file 1, for A Comprehensive Mini-Review of Curcumae Radix: Ethnopharmacology, Phytochemistry, and Pharmacology by Dongyi Hu, Jiayu Gao, Xiao Yang and Ying Liang in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
