Abstract
This study investigated the chemical composition, and antimicrobial activity against food-borne pathogens of the essential oil isolated from the heartwood of Calocedrus formosana from Taiwan. The oil, isolated by hydrodistillation in a Clevenger-type apparatus, was characterized using GC–FID and GC–MS. The major constituents were τ-muurolol (16.1%), α-cadinol (11.1%), α-terpineol (10.6%), thymol (8.5%), and β-thujaplicin (4.5%). The oil demonstrated strong activity against food-borne bacterial and fungal pathogens, and, to determine the source compounds responsible for this activity, the main components were individually evaluated. The most active source compounds were determined to be τ-muurolol, α-cadinol, thymol, and β-thujaplicin.
Numerous essential oil compounds have antibacterial, antifungal, insecticidal, and medicinal functionality. 1 -7 There has been a recent worldwide increase in the number of reported cases of infectious diseases, especially those that are caused by microbial contamination of foods. 8 During the processes for the production and sale of food, it can be infected with different microorganisms, such as Escherichia coli, Pseudomonas aeruginosa, Staphylococcus aureus, S. epidermidis, Bacillus subtilis, Vibrio parahaemolyticus, Aspergillus niger, and Chaetomium globosum, which can cause food spoilage, food poisoning, and other problems. 9 Therefore, nowadays, synthetic antimicrobial agents are used to control microbial growth in food and to reduce the incidence of food poisoning and spoilage. However, synthetic chemicals produce residual compounds so, instead of their use, plant natural products have been utilized. Plant essential oils are used as antibacterial agents to inhibit food-borne pathogens. These products are thought to be safer, non-toxic, well tolerated in the human body, and the demand for these is increasingly accepted. 9,10
Calocedrus formosana Florin (=C. macrolepis var. formosana) is an indigenous coniferous tree in Taiwan. 11 It is one of the 5 “precious” conifers of Taiwan. 12 Many previous studies have shown that the leaf, bark and seed essential oils and extracts of C. formosana have antimicrobial, antioxidant, anti-inflammatory, and anti-mosquito larvicide properties, 13 -21 but no study has determined the chemical composition and biological activity of the essential oil of C. formosana heartwood.
This study used hydrodistillation to obtain the essential oil of C. formosana heartwood and its chemical composition was determined using gas chromatography-flame ionization detection (GC–FID) and gas chromatography-mass spectrometry (GC–MS). The second part of this study determined the activity of the oil against food-borne bacterial and fungal pathogens. The purpose of this study was to establish a chemical basis for the effective multipurpose utilization of the species.
Results and Discussion
The essential oil obtained from the heartwood of C. formosana was a highly aromatic, dark-yellow-colored liquid. The yield of oil was 3.34 ± 0.08 ml/100 g, calculated on a moisture free basis. Table 1 shows the constituents, the percentage composition and the retention index (RI) values, which are listed in order of elution from the DB-5MS capillary column. A total of 65 compounds were identified from the hydrodistilled heartwood oil (Table 1). Among these, 2.0% were monoterpene hydrocarbons, 33.8% oxygenated monoterpenes, 17.1% sesquiterpene hydrocarbons, 35.0% oxygenated sesquiterpenes, 0.8% one diterpene, and 10.3% two other compounds. τ-Muurolol (16.1%) and α-cadinol (11.1%) were the major oxygenated sesquiterpenes, and α-terpineol (10.6%) and β-thujaplicin (4.5%) the main oxygenated monoterpenes. Of the others, thymol (8.5%) was the major compound. This is the first report of the heartwood oil composition of C. formosana.
Chemical Composition of the Heartwood Essential Oil of Calocedrus Formosana.
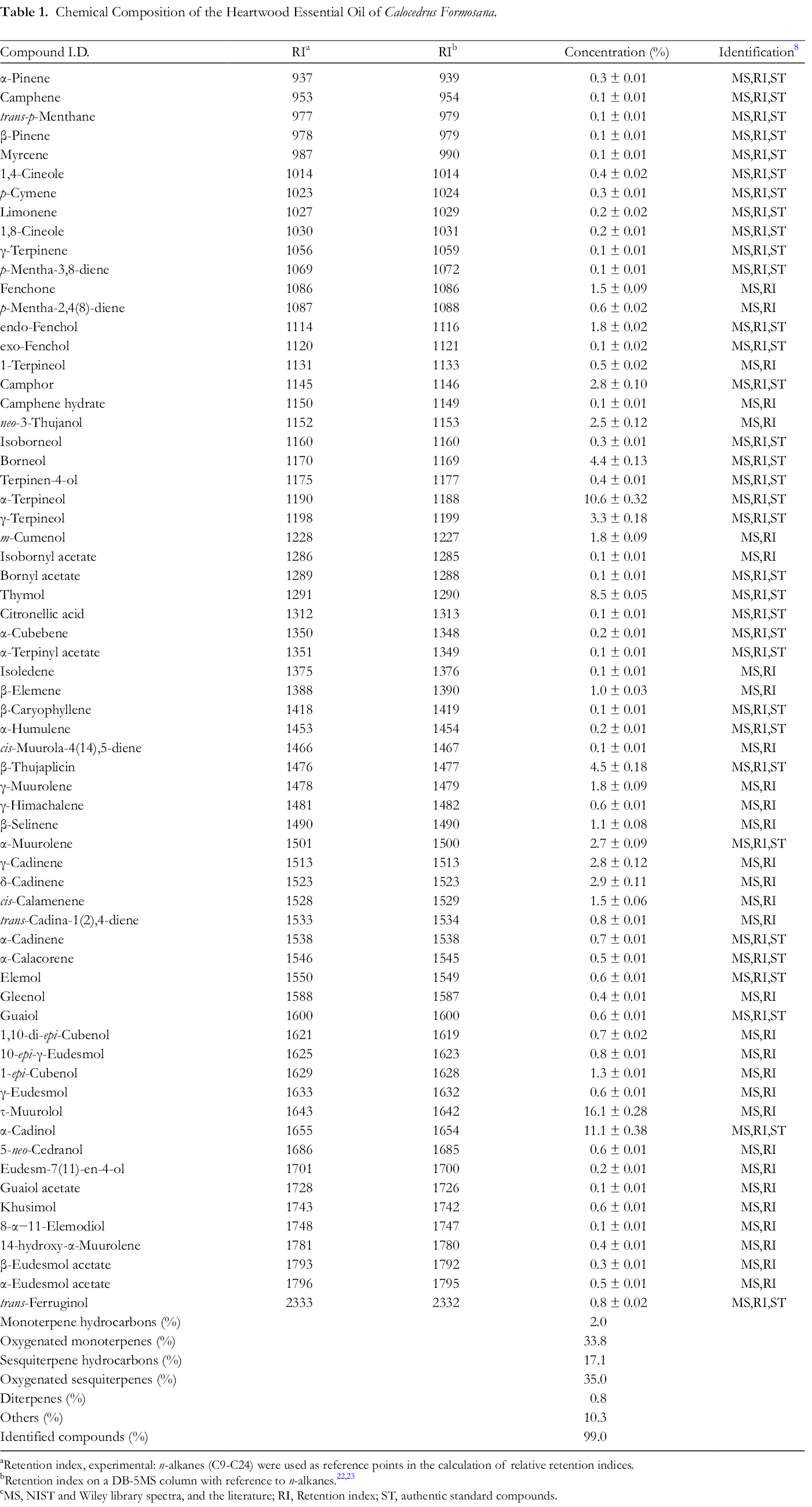
aRetention index, experimental: n-alkanes (C9-C24) were used as reference points in the calculation of relative retention indices.
cMS, NIST and Wiley library spectra, and the literature; RI, Retention index; ST, authentic standard compounds.
The heartwood oil of C. formosana was tested against 7 food-borne bacterial pathogens, including the 3 Gram-positive bacteria, Bacillus cereus, Staphylococcus aureus and S. epidermidis, and 4 Gram-negative bacteria, Escherichia coli, Enterobacter aerogenes, Pseudomonas aeruginosa and Vibrio parahaemolyticus. The inhibition zone, minimum inhibitory concentration (MIC), and minimum bactericidal concentration (MBC) values for the essential oil against the tested bacteria are listed in Table 2. The results show that the essential oil had good antibacterial activity against all tested microorganisms. The most sensitive were B. cereus, S. aureus, S. epidermidis, and E. coli, with inhibition zones of 53‐65 mm, MIC values of 31.25‐62.5 μg/mL, and MBC values of 62.5‐125 μg/mL.
Antibacterial Activity of the Heartwood Essential Oil From Calocedrus formosana.
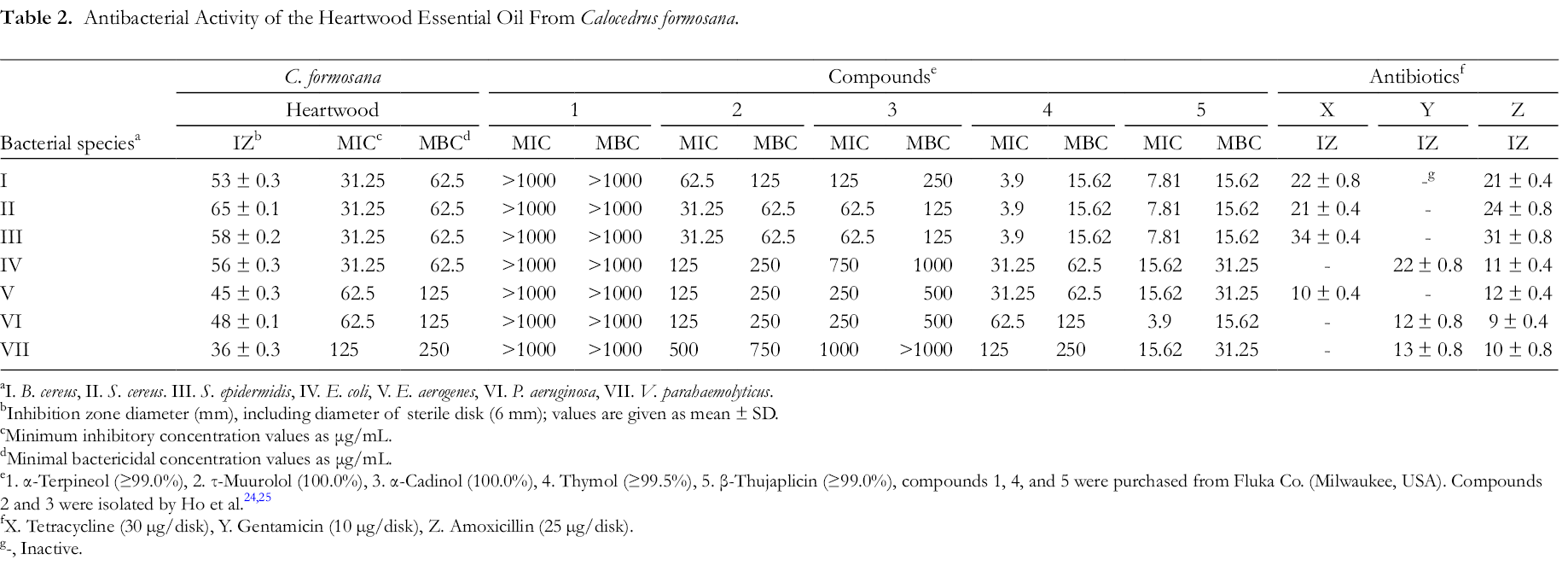
aI. B. cereus, II. S. cereus. III. S. epidermidis, IV. E. coli, V. E. aerogenes, VI. P. aeruginosa, VII. V. parahaemolyticus.
bInhibition zone diameter (mm), including diameter of sterile disk (6 mm); values are given as mean ± SD.
cMinimum inhibitory concentration values as μg/mL.
dMinimal bactericidal concentration values as μg/mL.
e1. α-Terpineol (≥99.0%), 2. τ-Muurolol (100.0%), 3. α-Cadinol (100.0%), 4. Thymol (≥99.5%), 5. β-Thujaplicin (≥99.0%), compounds 1, 4, and 5 were purchased from Fluka Co. (Milwaukee, USA). Compounds 2 and 3 were isolated by Ho et al. 24,25
fX. Tetracycline (30 µg/disk), Y. Gentamicin (10 µg/disk), Z. Amoxicillin (25 µg/disk).
g-, Inactive.
Kalemba and Kunicka 26 deemed that the majority of oxygenated essential oil compounds have antibacterial activity that is usually stronger than that of the oils, and always stronger than that of hydrocarbons. The reason for this is that terpenoids are lipid soluble, and the oxygen-containing compounds with phenol-, alcohol-, and aldehyde-containing functional groups can interfere with enzymatic activities, for instance, effecting, for example, respiratory function. As for inhibition of bacterial growth, these terpenoids can disrupt transduction of protons and affect phosphorylation of ADP, causing inhibition of bacterial growth. 27 In our experiments, the essential oil extracted from C. formosana heartwood was mainly composed of oxygenated compounds (oxygenated sesquiterpenes and oxygenated monoterpenes accounted for 68.8% of the total). Hence, the heartwood oil showed excellent anti-bacterial activity.
To determine the compounds that are responsible for the antibacterial activity of C. formosana heartwood oil, the main components were individually tested for antibacterial activity. The results showed that the active compounds are τ-muurolol, α-cadinol, thymol, and β-thujaplicin. τ-Muurolol and α-cadinol are cadinane-type compounds, and the literature reports that this type of compound possesses extensive antibacterial activity. 28,29 Thymol is well known to possess excellent anti-bacterial activity. 26 β-Thujaplicin, a tropolone-type compound, is known to possess multiple properties, including anti-oxidation, anti-inflammation, and antibacterial activities. 30 -32 Numerous literature supports that the 4 compounds, τ-muurolol, α-cadinol, thymol, and β-thujaplicin, possess excellent antibacterial activities. 33 -40
The heartwood essential oil was also tested against the 4 food-borne fungal pathogens, Aspergillus niger, Chaetomium globosum, Penicillium citrinum, and Trichoderma viride. Figure 1 shows the antifungal indices for the essential oil of the heartwood oil (at a concentration of 100 µg/mL) against the 4 food-borne fungi. At this concentration, the oil totally inhibited the growth of all 4 fungi. The MIC and IC50 values for the heartwood oil against these pathogens are shown in Table 3. The results clearly demonstrate that the heartwood essential oil of C. formosana exhibits excellent anti-fungal activity. The growth of A. niger, C. globosum, P. citrinum, and T. viride was completely inhibited at concentrations of 75, 50, 50, and 75 µg/mL, respectively.

Anti-food-borne fungal pathogen indices for the heartwood essential oil (100 µg/mL) from Calocedrus formosana.
MIC and IC50 Values (Μg/mL) for 5 Major Compounds From Calocedrus Formosana Heartwood Oil Against 4 Food-Borne Pathogenic Fungi.

Note: Nystatin (50 µg/mL) was used as a positive control.
To determine the compounds of C. formosana heartwood essential oil responsible for the anti-fungal activity, the major components were measured. The anti-fungal indices increased in the order: β-thujaplicin > thymol > α-cadinol> τ-muurolol> α-terpineol. At a concentration of 100 µg/mL, β-thujaplicin, thymol and α-cadinol totally inhibited the growth of the food-borne fungal pathogens. τ-Muurolol, at a concentration of 100 µg/mL, completely inhibited C. globosum and partially inhibited A. niger, P. citrinum, and T. viride; α-terpineol, at 100 µg/mL, slightly inhibited the fungal pathogens (Figure 2).

Anti-food-borne fungal pathogen indices for the 5 main compounds (100 µg/mL) of heartwood essential oil from Calocedrus formosana.
The MIC and IC50 values for α-terpineol, τ-muurolol, α-cadinol, thymol, and β-thujaplicin are shown in Table 3. The results show that the active source compounds are τ-muurolol, α-cadinol, thymol, and β-thujaplicin. Of these, β-thujaplicin has the best antifungal activity. The main mechanism of β-thujaplicin’s antifungal activity may lie in the change wrought by the compound in the permeability of the cell wall, causing leakage of the cell plasma and disrupting chelation of metallic ions in the biosynthesis of fungal enzymes. 41 Previous studies have shown that these compounds are very effective in suppressing fungal growth. 15,38,39,42 -45 The results show that τ-muurolol, α-cadinol, thymol, and β-thujaplicin of C. formosana heartwood oil are natural fungicides that can be used to control food-borne fungal pathogens.
Conclusion
This study reports that the essential oil extracted from the heartwood of Calocedrus formosana, with its main components of τ-muurolol, α-cadinol, α-terpineol, thymol, and β-thujaplicin, possesses pronounced antimicrobial activity against food-borne pathogens. The essential oil is composed mainly of oxygenated sesquiterpenes and oxygenated monoterpenes. Among these components, τ-muurolol, α-cadinol, thymol, and β-thujaplicin were proven to possess excellent antibacterial and antifungal activities. In the near future, our study should focus on developing a variety of natural anti-pathogen products.
Experimental
Plant Materials
Fresh heartwoods of C. formosana were collected in October 2020 from Chilan Mt in northeast Taiwan (Yilan County, altitude 850 m, latitude (N) 24°40’050’’, longitude (E) 121°39’10’’). The samples were compared with specimen no. ou 5886 from the Herbarium of National Chung-Hsing University and positively identified by Yu Hang-Ming of TFRI. A voucher specimen (CLH- 010) was deposited in the NCHU herbarium. Heartwood of the species was collected for subsequent extraction of essential oil and analysis.
Isolation of Heartwood Essential Oil
Heartwood of C. formosana (1 kg) was hydrodistilled for 3 hours using a Clevenger-type apparatus. The essential oil was then dried with anhydrous sodium sulfate. The oil yields and all test data are shown as the average of triplicate analyses.
Essential Oil Analysis
A Hewlett-Packard HP 6890 gas chromatograph equipped with a DB-5 fused silica capillary column (30 m x 0.25 mm x 0.25 μm film thickness, J&W Scientific) and a FID detector were used for the quantitative determination of oil components. The oven temperature was maintained at 50 °C for 2 minutes and increased to 250 °C at 5 °C/min. The injector temperature was 270 °C. The carrier gas was helium with a flow rate of 1 mL/min. The detector temperature was 250 °C and the split ratio was 1:10. Diluted samples (1.0 µL, 1/100, v/v, in ethyl acetate) were injected manually in split mode. The oil components were identified in terms of retention index (RI) 22 and mass spectra, which were obtained using GC/MS analysis on a Hewlett-Packard HP 6890/HP5973 equipped with a DB-5MS fused silica capillary column (30 m x 0.25 mm x 0.25 μm film thickness, J&W Scientific). The GC analysis parameters are listed above and the mass spectra were obtained (full scan mode: scan time: 0.3 s, mass range was m/z 30‐500) in EI mode at 70 eV. All data are shown as the average of triplicate analyses.
Component Identification
The heartwood oil constituents were identified by comparisons of retention index (RI), 22 retention time (RT) and mass spectra with those of authentic standards and/or the NIST and Wiley libraries spectra, and literature. 22,23
Antibacterial Activity
The antibacterial properties of the heartwood essential oil and the oil’s main compounds were analyzed using the paper disc diffusion method, minimum inhibition concentration (MIC), and minimum bactericidal concentration (MBC). Discs containing 15 µL and 30 µL of the oil dissolved in dimethylsulfoxide (DMSO) were placed on inoculated plates with test microorganisms. Growth inhibition zones (including a disc diameter of 6 mm) were measured after 24 hours of incubation at 37 °C. Gentamicin, tetracycline, and amoxicillin were used as positive controls. Microbial strains were obtained from the Culture Collection and Research Center of the Food Industry Research and Development Institute, Hsinchu City, Taiwan. The microbial strains included 4 Gram-negative bacteria, E. coli (IFO 3301), E. aerogenes (ATCC 13048), P. aeruginosa (IFO 3080), and V. parahaemolyticus (ATCC 17803), and 3 Gram-positive bacteria, B. cereus (ATCC 11778), S. aureus (ATCC 6538P), and S. epidermidis (ATCC 12228).
The minimum inhibitory concentration (MIC) values were measured using a microdilution broth susceptibility assay, as recommended by NCCLS, 46 and as reported earlier. 24 To determine the minimal bactericidal concentration (MBC), 100 µL of each dilution with no evident growth was spread on Mueller-Hinton agar (MHA). The inoculated Petri dishes and the controls were incubated at 37 °C for 24 hours. The number of colony forming units was counted and compared with that for the control dishes. MBC is defined as the lowest concentration that kills >99.9% of the initial inoculum. 47
Anti- Food-Borne Fungal Pathogen Assays
The method of Su et al. 48 was used. The fungi were A. niger (ATCC 6275), Ch. globosum (ATCC 6205), P. citrinum (ATCC 9849), and T. viride (ATCC8678). Microbial strains were obtained from the Culture Collection and Research Center of the Food Industry Research and Development Institute, Hsinchu City, Taiwan. Anti- food-borne fungal assays were performed in triplicate and the data were averaged. Different concentrations of the essential oil (6.25 ~ 250 µg/mL) were added to sterilized potato dextrose agar (PDA). The test plates were incubated at 27 °C. When the mycelium of the fungi reached the edge of the control plate, the anti-food-borne fungal pathogen index was calculated as:

where Da is the diameter of the growth zone in the experimental dish (cm) and Db is the diameter of the growth zone in the control dish (cm).
Nystatin was used as a positive control. Each test was repeated 5 times and the data were averaged. The IC50 values (the concentration in mg per mL that inhibit 50% of mycelium growth) were calculated using probit analysis.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
