Abstract
An asymmetric synthesis of 14-methyl-1-octadecene, the sex pheromone of the peach leafminer moth, has been efficiently achieved. A key chiral intermediate, (R)−4-(benzyloxy)−3-methylbutanal, which has been developed as the resource chemical, can be obtained in large quantities from the steroid industry. 1,11-Undecanediol is selectively masked, and then converted into a phosphonium salt derivative, thereby effectively constructing a carbon skeleton. The target molecule is synthesized in 8 linear steps, with a 42% yield. The key characteristic of our synthesis lies in its chiral-pool strategy.
Recently, there has been a sharp conflict between intense agricultural production and environmental issues. We all have to face the harmful environmental effects from traditional agricultural production. 1 Therefore, there is an urgent demand for the development of a “green” production mode, such as low-dosage sex pheromones to control pests without the negative side effects. The peach leafminer moth (Lyonetia clerkella) is one of the most destructive pests in East Asia, and also feeds on pear, plum, and apple. 2 It induces defoliation when the leaves are infested with only a few larvae. This has led a high demand for the use of sex pheromones to control its population by disrupting its mating process. Regarding chirality, the purity of the different enantiomers will change biological activity dramatically, but the provision of enantio-pure pheromones at low cost is still an unmet need in the industry.
Sugie et al. first isolated (S)−14-methyl-1-octadecene (

Chemical structure of the sex pheromone of the peach leafminer moth.
Our research group is committed to discovering a new method for the synthesis of the sex pheromone of the peach leafminer moth. 11 We are interested in using more economical, effective, and natural chemical substances to synthesize the sex pheromone. After oxidative degradation of the green and environmentally friendly diosgenin Figure 2, a steroidal sapogenin obtained from Dioscorea zingiberensis (Dioscorea oppositifolia), 12 (5R)−3-hydroxy-5-methyltetrahydro-2H-pyran-2-one, is obtained as a by-product. 13 In this article, we report our new synthetic method for (S)−1 through the chiral pool strategy. 14

Dioscorea oppositifolia and the resource chemicals.
The retrosynthetic analysis of
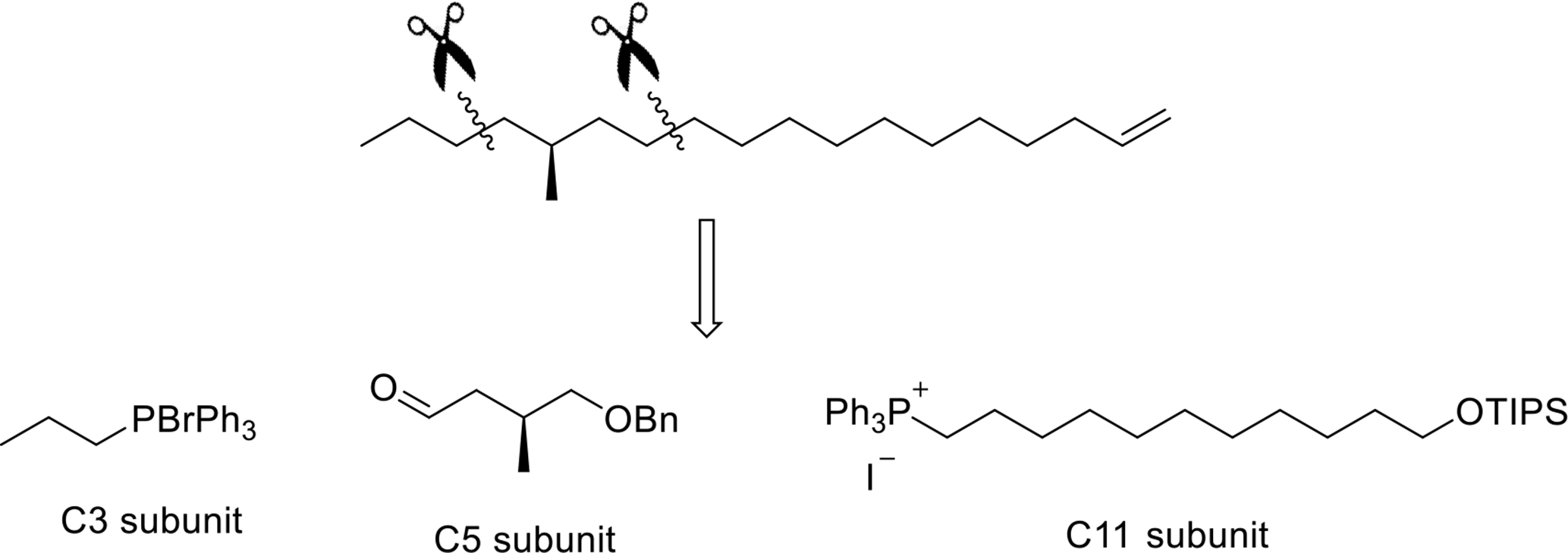
Retrosynthetic analysis of 1.
Results and Discussion
As shown in Scheme 1, our synthesis started from (R)−4-(benzyloxy)−3-methylbutanal (

Synthetic pathway for (S)−
With compound
Reagents and conditions: a) n-BuLi, THF, compound 9, -78°C~rt, 84%; b) H2, MeOH, Pd/C, rt, 95%; c) DMP, DCM, 0°C~rt, 93%; d) n-BuLi, THF, triphenyl(propyl)phosphonium iodide, -78°C ~rt, 83%; e) TBAF, DCM, 0°C ~rt, 93%; f) H2, MeOH, Pt/C, rt, 95%; g) CBr4, PPh3, DCM, 0°C~rt, 91%; h) KOBu-t, THF, 85%.
Conclusions
In summary, we have built the C-C skeleton of the pheromone of the peach leafminer moth through the ‘C3 +C5+C11’ strategy. The asymmetric total synthesis of the sex pheromone of peachleaf miner moth, (S)−14-methyl-1-octadecene was achieved with Wittig coupling and hydrogenation as the key steps in 42% overall yield, starting with (R)−4-(benzyloxy)−3-methylbutanal
Supplemental Material
Online supplementary file 1 - Supplemental material for A New Asymmetric Synthesis of (S)-14-Methyl-1-Octadecene, the Sex Pheromone of the Peach Leafminer Moth
Supplemental material, Online supplementary file 1, for A New Asymmetric Synthesis of (S)-14-Methyl-1-Octadecene, the Sex Pheromone of the Peach Leafminer Moth by Guo-Guo He, Si-Fan Liu, Bao-Qi Rao, Hong-Jin Bai and Zhen-Ting Du in Natural Product Communications
Footnotes
Acknowledgments
The authors thank Liang Zhu Huang for the hydrogenation steps, technician Hong-Li Zhang for the NMR analysis and Yao-Xiang Duan for HRMS detection.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by NSFC grants (numbers 31301712 and 31760639). Partial financial support from the Jiangxi Technological Normal University is greatly appreciated. Zhen-Ting Du thanks the Opening Funds of the Key Laboratory of Synthetic Chemistry of Natural Substances, Shanghai Institute of Organic Chemistry, and the Chinese Academy of Sciences for partial financial support.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
